Ion channels are pore-forming membrane proteins that allow ions to pass through the channel pore. Their functions include establishing a resting membrane potential, shaping action potentials and other electrical signals by gating the flow of ions across the cell membrane, controlling the flow of ions across secretory and epithelial cells, and regulating cell volume. Ion channels are present in the membranes of all cells. Ion channels are one of the two classes of ionophoric proteins, the other being ion transporters.

Voltage-gated ion channels are a class of transmembrane proteins that form ion channels that are activated by changes in the electrical membrane potential near the channel. The membrane potential alters the conformation of the channel proteins, regulating their opening and closing. Cell membranes are generally impermeable to ions, thus they must diffuse through the membrane through transmembrane protein channels. They have a crucial role in excitable cells such as neuronal and muscle tissues, allowing a rapid and co-ordinated depolarization in response to triggering voltage change. Found along the axon and at the synapse, voltage-gated ion channels directionally propagate electrical signals. Voltage-gated ion-channels are usually ion-specific, and channels specific to sodium (Na+), potassium (K+), calcium (Ca2+), and chloride (Cl−) ions have been identified. The opening and closing of the channels are triggered by changing ion concentration, and hence charge gradient, between the sides of the cell membrane.
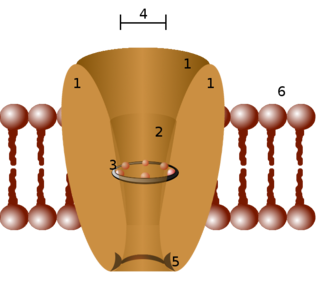
Two-pore channels (TPCs) are eukaryotic intracellular voltage-gated and ligand gated cation selective ion channels. There are two known paralogs in the human genome, TPC1s and TPC2s. In humans, TPC1s are sodium selective and TPC2s conduct sodium ions, calcium ions and possibly hydrogen ions. Plant TPC1s are non-selective channels. Expression of TPCs are found in both plant vacuoles and animal acidic organelles. These organelles consist of endosomes and lysosomes. TPCs are formed from two transmembrane non-equivalent tandem Shaker-like, pore-forming subunits, dimerized to form quasi-tetramers. Quasi-tetramers appear very similar to tetramers, but are not quite the same. Some key roles of TPCs include calcium dependent responses in muscle contraction(s), hormone secretion, fertilization, and differentiation. Disorders linked to TPCs include membrane trafficking, Parkinson's disease, Ebola, and fatty liver.

Nicotinic acid adenine dinucleotide phosphate (NAADP) is a Ca2+-mobilizing second messenger synthesised in response to extracellular stimuli. Like its mechanistic cousins, IP3 and cyclic adenosine diphosphoribose (Cyclic ADP-ribose), NAADP binds to and opens Ca2+ channels on intracellular organelles, thereby increasing the intracellular Ca2+ concentration which, in turn, modulates sundry cellular processes (see Calcium signalling). Structurally, it is a dinucleotide that only differs from the house-keeping enzyme cofactor, NADP by a hydroxyl group (replacing the nicotinamide amino group) and yet this minor modification converts it into the most potent Ca2+-mobilizing second messenger yet described. NAADP acts across phyla from plants to humans.

Sodium channel, voltage-gated, type XI, alpha subunit also known as SCN11A or Nav1.9 is a voltage-gated sodium ion channel protein which is encoded by the SCN11A gene on chromosome 3 in humans. Like Nav1.7 and Nav1.8, Nav1.9 plays a role in pain perception. This channel is largely expressed in small-diameter nociceptors of the dorsal root ganglion and trigeminal ganglion neurons, but is also found in intrinsic myenteric neurons.
Transient receptor potential cation channel, subfamily M, member 2, also known as TRPM2, is a protein that in humans is encoded by the TRPM2 gene.

Frizzled-8(Fz-8) is a protein that in humans is encoded by the FZD8 gene.

Potassium channel subfamily K member 1 is a protein that in humans is encoded by the KCNK1 gene.

Calcium channel, voltage-dependent, L type, alpha 1D subunit is a protein that in humans is encoded by the CACNA1D gene. Cav1.3 channels belong to the Cav1 family, which form L-type calcium currents and are sensitive to selective inhibition by dihydropyridines (DHP).
Potassium channel subfamily K member 4 is a protein that in humans is encoded by the KCNK4 gene. KCNK4 protein channels are also called TRAAK channels.

Structural maintenance of chromosomes protein 5 is a protein encoded by the SMC5 gene in human.

DNA replication licensing factor MCM8 is a protein that in humans is encoded by the MCM8 gene.

Potassium channel subfamily K member 17 is a protein that in humans is encoded by the KCNK17 gene.

Two pore segment channel 1 (TPC1) is a human protein encoded by the TPCN1 gene. The protein encoded by this gene is an ion channel. In contrast to other calcium and sodium channels which have four homologous domains, each containing six transmembrane segments, TPCN1 only contains two domains.

Potassium channel, subfamily K, member 10, also known as KCNK10 is a human gene. The protein encoded by this gene, K2P10.1, is a potassium channel containing two pore-forming P domains.

Potassium channel subfamily K member 16 is a protein that in humans is encoded by the KCNK16 gene. The protein encoded by this gene, K2P16.1, is a potassium channel containing two pore-forming P domains.

CatSper1, is a protein which in humans is encoded by the CATSPER1 gene. CatSper1 is a member of the cation channels of sperm family of protein. The four proteins in this family together form a Ca2+-permeant ion channel specific essential for the correct function of sperm cells.

CatSper2, is a protein which in humans is encoded by the CATSPER2 gene. CatSper2 is a member of the cation channels of sperm family of protein. The four proteins in this family together form a Ca2+-permeant ion channel specific essential for the correct function of sperm cells.

Chloroplast DNA (cpDNA) is the DNA located in chloroplasts, which are photosynthetic organelles located within the cells of some eukaryotic organisms. Chloroplasts, like other types of plastid, contain a genome separate from that in the cell nucleus. The existence of chloroplast DNA was identified biochemically in 1959, and confirmed by electron microscopy in 1962. The discoveries that the chloroplast contains ribosomes and performs protein synthesis revealed that the chloroplast is genetically semi-autonomous. The first complete chloroplast genome sequences were published in 1986, Nicotiana tabacum (tobacco) by Sugiura and colleagues and Marchantia polymorpha (liverwort) by Ozeki et al. Since then, a great number of chloroplast DNAs from various species have been sequenced.

Voltage-gated hydrogen channel 1 is a protein that in humans is encoded by the HVCN1 gene.



















