
In molecular biology, CD4 is a glycoprotein that serves as a co-receptor for the T-cell receptor (TCR). CD4 is found on the surface of immune cells such as T helper cells, monocytes, macrophages, and dendritic cells. It was discovered in the late 1970s and was originally known as leu-3 and T4 before being named CD4 in 1984. In humans, the CD4 protein is encoded by the CD4 gene.

The T-cell receptor (TCR) is a protein complex found on the surface of T cells, or T lymphocytes, that is responsible for recognizing fragments of antigen as peptides bound to major histocompatibility complex (MHC) molecules. The binding between TCR and antigen peptides is of relatively low affinity and is degenerate: that is, many TCRs recognize the same antigen peptide and many antigen peptides are recognized by the same TCR.
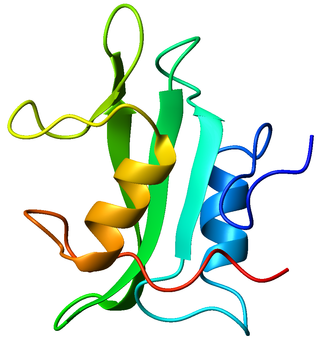
Lck is a 56 kDa protein that is found inside specialized cells of the immune system called lymphocytes. The Lck is a member of Src kinase family (SFK), it is important for the activation of the T-cell receptor signaling in both naive T cells and effector T cells. The role of the Lck is less prominent in the activation or in the maintenance of memory CD8 T cells in comparison to CD4 T cells. In addition, the role of the lck varies among the memory T cells subsets. It seems that in mice, in the effector memory T cells (TEM) population, more than 50% of lck is present in a constitutively active conformation, whereas, only less than 20% of lck is present as active form of lck. These differences are due to differential regulation by SH2 domain–containing phosphatase-1 (Shp-1) and C-terminal Src kinase.

Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit delta isoform also known as phosphoinositide 3-kinase (PI3K) delta isoform or p110δ is an enzyme that in humans is encoded by the PIK3CD gene.
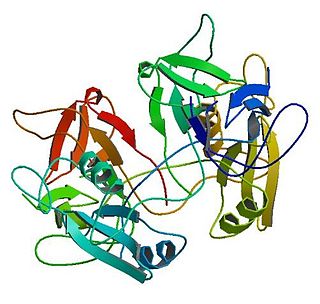
Growth factor receptor-bound protein 2, also known as Grb2, is an adaptor protein involved in signal transduction/cell communication. In humans, the GRB2 protein is encoded by the GRB2 gene.
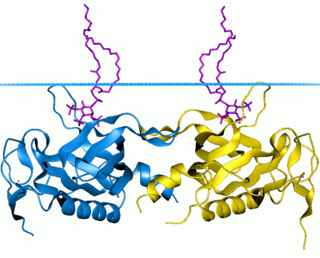
Bruton's tyrosine kinase, also known as tyrosine-protein kinase BTK, is a tyrosine kinase that is encoded by the BTK gene in humans. BTK plays a crucial role in B cell development.

Tyrosine-protein kinase SYK, also known as spleen tyrosine kinase, is an enzyme which in humans is encoded by the SYK gene.
An immunoreceptor tyrosine-based activation motif (ITAM) is a conserved sequence of four amino acids that is repeated twice in the cytoplasmic tails of non-catalytic tyrosine-phosphorylated receptors, cell-surface proteins found mainly on immune cells. Its major role is being an integral component for the initiation of a variety of signaling pathway and subsequently the activation of immune cells, although different functions have been described, for example an osteoclast maturation.
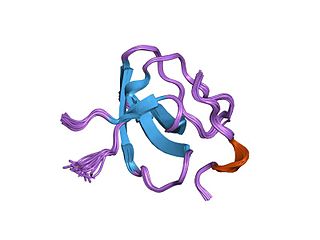
Lymphocyte cytosolic protein 2, also known as LCP2 or SLP-76, is a signal-transducing adaptor protein expressed in T cells and myeloid cells and is important in the signaling of T-cell receptors (TCRs). As an adaptor protein, SLP-76 does not have catalytic functions, primarily binding other signaling proteins to form larger signaling complexes. It is a key component of the signaling pathways of receptors with immunoreceptor tyrosine-based activation motifs (ITAMs) such as T-cell receptors, its precursors, and receptors for the Fc regions of certain antibodies. SLP-76 is expressed in T-cells and related lymphocytes like natural killer cells.

Tyrosine-protein kinase ITK/TSK also known as interleukin-2-inducible T-cell kinase or simply ITK, is a protein that in humans is encoded by the ITK gene. ITK is a member of the TEC family of kinases and is highly expressed in T cells.
CD5 is a cluster of differentiation expressed on the surface of T cells and in a subset of murine B cells known as B-1a. The expression of this receptor in human B cells has been a controversial topic and to date there is no consensus regarding the role of this receptor as a marker of human B cells. B-1 cells have limited diversity of their B-cell receptor due to their lack of the enzyme terminal deoxynucleotidyl transferase (TdT) and are potentially self-reactive. CD5 serves to mitigate activating signals from the BCR so that the B-1 cells can only be activated by very strong stimuli and not by normal tissue proteins. CD5 was used as a T-cell marker until monoclonal antibodies against CD3 were developed.
The Linker for activation of T cells, also known as linker of activated T cells or LAT, is a protein involved in the T-cell antigen receptor signal transduction pathway which in humans is encoded by the LAT gene. Alternative splicing results in multiple transcript variants encoding different isoforms.

Phospholipase C, gamma 1, also known as PLCG1 and PLCgamma1, is a protein that in humans involved in cell growth, migration, apoptosis, and proliferation. It is encoded by the PLCG1 gene and is part of the PLC superfamily.
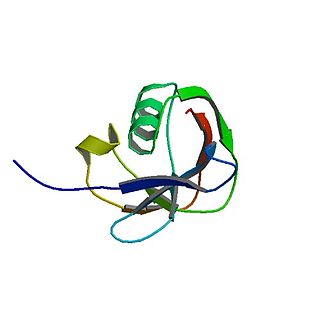
T-cell surface glycoprotein CD3 gamma chain is a protein that in humans is encoded by the CD3G gene.

B-cell linker (BLNK) protein is expressed in B cells and macrophages and plays a large role in B cell receptor signaling. Like all adaptor proteins, BLNK has no known intrinsic enzymatic activity. Its function is to temporally and spatially coordinate and regulate downstream signaling effectors in B cell receptor (BCR) signaling, which is important in B cell development. Binding of these downstream effectors is dependent on BLNK phosphorylation. BLNK is encoded by the BLNK gene and is also known as SLP-65, BASH, and BCA.

FYN binding protein (FYB-120/130), also known as FYB, ADAP, and SLAP-130 is a protein that is encoded by the FYB gene in humans. The protein is expressed in T cells, monocytes, mast cells, macrophages, NK cells, but not B cells. FYB is a multifunctional protein involved in post-activation T cell signaling, lymphocyte cytokine production, cell adhesion, and actin remodeling.

Tyrosine-protein kinase TXK is an enzyme that in humans is encoded by the TXK gene.
A non-receptor tyrosine kinase (nRTK) is a cytosolic enzyme that is responsible for catalysing the transfer of a phosphate group from a nucleoside triphosphate donor, such as ATP, to tyrosine residues in proteins. Non-receptor tyrosine kinases are a subgroup of protein family tyrosine kinases, enzymes that can transfer the phosphate group from ATP to a tyrosine residue of a protein (phosphorylation). These enzymes regulate many cellular functions by switching on or switching off other enzymes in a cell.
Kinetic-segregation is a model proposed for the mechanism of T-cell receptor (TCR) triggering. It offers an explanation for how TCR binding to its ligand triggers T-cell activation, based on size-sensitivity for the molecules involved. Simon J. Davis and Anton van der Merwe, University of Oxford, proposed this model in 1996. According to the model, TCR signalling is initiated by segregation of phosphatases with large extracellular domains from the TCR complex when binding to its ligand, allowing small kinases to phosphorylate intracellular domains of the TCR without inhibition. Its might also be applicable to other receptors of the Non-catalytic tyrosine-phosphorylated receptors family such as CD28.
Non-catalytic tyrosine-phosphorylated receptors (NTRs), also called immunoreceptors or Src-family kinase-dependent receptors, are a group of cell surface receptors expressed by leukocytes that are important for cell migration and the recognition of abnormal cells or structures and the initiation of an immune response. These transmembrane receptors are not grouped into the NTR family based on sequence homology, but because they share a conserved signalling pathway utilizing the same signalling motifs. A signaling cascade is initiated when the receptors bind their respective ligand resulting in cell activation. For that tyrosine residues in the cytoplasmic tail of the receptors have to be phosphorylated, hence the receptors are referred to as tyrosine-phosphorylated receptors. They are called non-catalytic receptors, as the receptors have no intrinsic tyrosine kinase activity and cannot phosphorylate their own tyrosine residues. Phosphorylation is mediated by additionally recruited kinases. A prominent member of this receptor family is the T-cell receptor.

























