
PiHKAL: A Chemical Love Story is a book by Dr. Alexander Shulgin and Ann Shulgin, published in 1991. The subject of the work is psychoactive phenethylamine chemical derivatives, notably those that act as psychedelics and/or empathogen-entactogens. The main title, PiHKAL, is an acronym that stands for "Phenethylamines I Have Known and Loved."
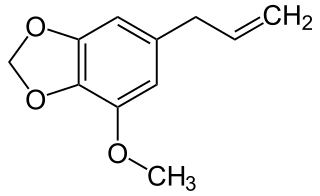
Myristicin is a naturally occurring compound found in common herbs and spices, such as nutmeg. It is an insecticide, and has been shown to enhance the effectiveness of other insecticides.

The MTT assay is a colorimetric assay for assessing cell metabolic activity. NAD(P)H-dependent cellular oxidoreductase enzymes may, under defined conditions, reflect the number of viable cells present. These enzymes are capable of reducing the tetrazolium dye MTT, which is chemically 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, to its insoluble formazan, which has a purple color. Other closely related tetrazolium dyes including XTT, MTS and the WSTs, are used in conjunction with the intermediate electron acceptor, 1-methoxy phenazine methosulfate (PMS). With WST-1, which is cell-impermeable, reduction occurs outside the cell via plasma membrane electron transport. However, this traditionally assumed explanation is currently contended as proof has also been found of MTT reduction to formazan in lipidic cellular structures without apparent involvement of oxidoreductases.

The Controlled Drugs and Substances Act is Canada's federal drug control statute. Passed in 1996 under Prime Minister Jean Chrétien's government, it repeals the Narcotic Control Act and Parts III and IV of the Food and Drugs Act, and establishes eight Schedules of controlled substances and two Classes of precursors. It provides that "The Governor in Council may, by order, amend any of Schedules I to VIII by adding to them or deleting from them any item or portion of an item, where the Governor in Council deems the amendment to be necessary in the public interest."

Lophophine is a putative psychedelic and entactogen drug of the methylenedioxyphenethylamine class. It is the α-demethylated homologue of MMDA, and is also closely related to mescaline.

MMDA is a psychedelic and entactogen drug of the amphetamine class. It is an analogue of lophophine, MDA, and MDMA.

MMDA-2 (2-methoxy-4,5-methylenedioxyamphetamine) is a psychedelic drug of the amphetamine class. It is closely related to MMDA and MDA.

Trimethyl phosphite is an organophosphorus compound with the formula P(OCH3)3, often abbreviated P(OMe)3. It is a colorless liquid with a highly pungent odor. It is the simplest phosphite ester and finds used as a ligand in organometallic chemistry and as a reagent in organic synthesis. The molecule features a pyramidal phosphorus(III) center bound to three methoxy groups.

5-MeO-MPMI (also known as 5-Methoxy-N-methyl-(α,N-trimethylene)tryptamine) is a tryptamine derivative that is a psychedelic drug. It was first developed by the team led by JE Macor in 1992, and subsequently investigated by the team led by David Nichols from Purdue University in the late 1990s. This compound produces psychedelic-appropriate responding in animal tests with a similar potency to the amphetamine-derived psychedelic DOI, and has two enantiomers, with only the (R)-enantiomer being active.

3-Methoxy-4-methylamphetamine (MMA) is an entactogen and psychedelic drug of the phenethylamine and amphetamine classes. It was first synthesized in 1970 and was encountered as a street drug in Italy in the same decade. MMA was largely forgotten until being reassayed by David E. Nichols as a non-neurotoxic MDMA analogue in 1991, and has subsequently been sold as a designer drug on the internet since the late 2000s (decade).
A serotonin releasing agent (SRA) is a type of drug that induces the release of serotonin into the neuronal synaptic cleft. A selective serotonin releasing agent (SSRA) is an SRA with less significant or no efficacy in producing neurotransmitter efflux at other types of monoamine neurons.

meta-Methoxyamphetamine (MMA), also known as 3-methoxyamphetamine (3-MA), is a stimulant drug from the amphetamine family. It has similar effects in animal drug discrimination tests to the more widely known derivative 4-methoxyamphetamine (PMA), although with a slightly different ratio of monoamine release, being a combined serotonin, dopamine, and norepinephrine releasing agent rather than a fairly selective serotonin releaser like PMA. 3-Methoxyamphetamine has similarly appeared on the illicit market as a designer drug alternative to MDMA, although far more rarely than its infamous positional isomer. It produces gepefrine, a cardiac stimulant, as one of its major metabolites.

A-836,339 is a drug developed by Abbott Laboratories that acts as a potent cannabinoid receptor full agonist. It is selective for CB2, with Ki values of 0.64 nM at CB2 vs 270 nM at the psychoactive CB1 receptor, but while it exhibits selective analgesic, anti-inflammatory and anti-hyperalgesic effects at low doses, its high efficacy at both targets results in typical cannabis-like effects appearing at higher doses, despite its low binding affinity for CB1. In 2012 A-836,339 was detected via X-ray crystallography in a "dubious product" sold in Japan, though the product was described as a white powder, not herbal incense, it was suggested to be for human consumption.
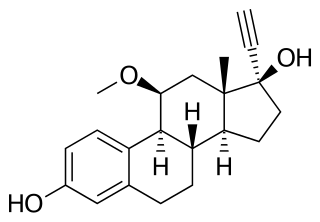
Moxestrol, sold under the brand name Surestryl, is an estrogen medication which has been used in Europe for the treatment of menopausal symptoms and menstrual disorders. It is taken by mouth. In addition to its use as a medication, moxestrol has been used in scientific research as a radioligand of the estrogen receptor.
The major drug laws of India are the Narcotic Drugs and Psychotropic Substances Act (1985) and the Prevention of Illicit Trafficking in Narcotic Drugs and Psychotropic Substances Act (1988).

5-MeO-NBpBrT is a N-substituted member of the methoxytryptamine family of compounds. Like other such compounds it acts as an antagonist for the 5-HT2A receptor, with a claimed 100x selectivity over the closely related 5-HT2C receptor. While N-benzyl substitution of psychedelic phenethylamines often results in potent 5-HT2A agonists, it had been thought that N-benzyl tryptamines show much lower efficacy and are either very weak partial agonists or antagonists at 5-HT2A, though more recent research has shown stronger agonist activity for 3-substituted benzyl derivatives. Extending the benzyl group to a substituted phenethyl can also recover agonist activity in certain cases.

Piper marginatum, the cake bush, Anesi wiwiri, marigold pepper, Ti Bombé in Creole or Hinojo, is a plant species in the genus Piper found in moist, shady spots in the Amazon rainforest in Surinam, French Guiana and Brazil.

Homoisoflavonoids (3-benzylidenechroman-4-ones) are a type of phenolic compounds occurring naturally in plants.

4'-Methoxy-α-pyrrolidinopentiophenone is a stimulant drug of the cathinone class that has been sold online as a designer drug.

















