
Amgen Inc. is an American multinational biopharmaceutical company headquartered in Thousand Oaks, California. One of the world's largest independent biotechnology companies, As of 2022, Amgen has approximately 24,000 staff in total.
Panitumumab, sold under the brand name Vectibix, is a fully human monoclonal antibody specific to the epidermal growth factor receptor.

KRAS is a gene that provides instructions for making a protein called K-Ras, a part of the RAS/MAPK pathway. The protein relays signals from outside the cell to the cell's nucleus. These signals instruct the cell to grow and divide (proliferate) or to mature and take on specialized functions (differentiate). It is called KRAS because it was first identified as a viral oncogene in the KirstenRAt Sarcoma virus. The oncogene identified was derived from a cellular genome, so KRAS, when found in a cellular genome, is called a proto-oncogene.
Crizotinib, sold under the brand name Xalkori among others, is an anti-cancer medication used for the treatment of non-small cell lung carcinoma (NSCLC). Crizotinib inhibits the c-Met/Hepatocyte growth factor receptor (HGFR) tyrosine kinase, which is involved in the oncogenesis of a number of other histological forms of malignant neoplasms. It also acts as an ALK and ROS1 inhibitor.
Selumetinib (INN), sold under the brand name Koselugo, is a medication for the treatment of children, two years of age and older, with neurofibromatosis type I (NF-1), a genetic disorder of the nervous system causing tumors to grow on nerves. It is taken by mouth.
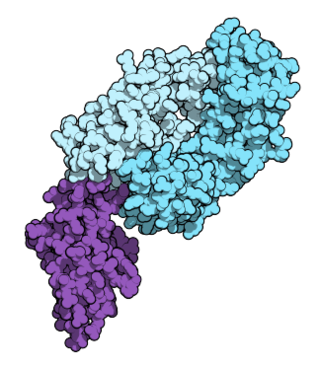
Nivolumab, sold under the brand name Opdivo, is an anti-cancer medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urothelial carcinoma, colon cancer, esophageal squamous cell carcinoma, liver cancer, gastric cancer, and esophageal or gastroesophageal junction cancer. It is administered intravenously.
A MEK inhibitor is a chemical or drug that inhibits the mitogen-activated protein kinase kinase enzymes MEK1 and/or MEK2. They can be used to affect the MAPK/ERK pathway which is often overactive in some cancers.

Pembrolizumab, sold under the brand name Keytruda, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer, cervical cancer, and certain types of breast cancer. It is administered by slow intravenous injection.
K-Ras(G12C) inhibitor 6 is an irreversible inhibitor of oncogenic K-Ras(G12C), subverting the native nucleotide preference to favour GDP over GTP. Its family of inhibitors allosterically control GTP affinity and effector interactions by fitting inside a "pocket", or binding site, of mutant K-Ras. It is the most frequently mutated oncogene.

Atezolizumab, sold under the brand name Tecentriq among others, is a monoclonal antibody medication used to treat urothelial carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC), hepatocellular carcinoma and alveolar soft part sarcoma, but discontinued for use in triple-negative breast cancer (TNBC). It is a fully humanized, engineered monoclonal antibody of IgG1 isotype against the protein programmed cell death-ligand 1 (PD-L1).

Osimertinib, sold under the brand name Tagrisso, is a medication used to treat non-small-cell lung carcinomas with specific mutations. It is a third-generation epidermal growth factor receptor tyrosine kinase inhibitor.

Capmatinib, sold under the brand name Tabrecta, is an anticancer medication used for the treatment of metastatic non-small cell lung cancer whose tumors have a mutation that leads to the exon 14 skipping of the MET gene, which codes for the membrane receptor HGFR.
Erdafitinib, sold under the brand name Balversa, is an anti-cancer medication. It is a small molecule inhibitor of fibroblast growth factor receptor (FGFR) used for the treatment of cancer. FGFRs are a subset of tyrosine kinases which are unregulated in some tumors and influence tumor cell differentiation, proliferation, angiogenesis, and cell survival. Astex Pharmaceuticals discovered the drug and licensed it to Janssen Pharmaceuticals for further development.
Cemiplimab, sold under the brand name Libtayo, is a monoclonal antibody medication for the treatment of squamous cell skin cancer. Cemiplimab belongs to a class of drugs that binds to the programmed death receptor-1 (PD-1), blocking the PD-1/PD-L1 pathway.

Tepotinib, sold under the brand name Tepmetko, is an anti-cancer medication used for the treatment of adults with non-small cell lung cancer (NSCLC).

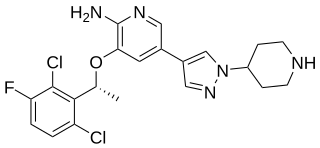
Selpercatinib, sold under the brand name Retevmo among others, is a medication for the treatment of cancers in people whose tumors have an alteration in a specific gene. It is taken by mouth.
Pralsetinib, sold under the brand name Gavreto, is a medication approved for RET mutation-positive medullary thyroid cancer (MTC) and RET fusion-positive differentiated thyroid cancer (DTC) refractory to radioactive iodine (RAI) therapy. Pralsetinib is a tyrosine kinase inhibitor. It is taken by mouth.

Mobocertinib, sold under the brand name Exkivity, is used for the treatment of non-small cell lung cancer.

Adagrasib, sold under the brand name Krazati, is an anticancer medication used to treat non-small cell lung cancer. Adagrasib is an inhibitor of the RAS GTPase family. It is taken by mouth. It is being developed by Mirati Therapeutics.
Amivantamab, sold under the brand name Rybrevant, is a bispecific monoclonal antibody used to treat non-small cell lung cancer. Amivantamab is a bispecific epidermal growth factor (EGF) receptor-directed and mesenchymal–epithelial transition (MET) receptor-directed antibody. It is the first treatment for adults with non-small cell lung cancer whose tumors have specific types of genetic mutations: epidermal growth factor receptor (EGFR) exon 20 insertion mutations.














