Primary myelofibrosis (PMF) is a rare bone marrow blood cancer. It is classified by the World Health Organization (WHO) as a type of myeloproliferative neoplasm, a group of cancers in which there is activation and growth of mutated cells in the bone marrow. This is most often associated with a somatic mutation in the JAK2, CALR, or MPL genes. In PMF, the bony aspects of bone marrow are remodeled in a process called osteosclerosis; in addition, fibroblast secrete collagen and reticulin proteins that are collectively referred to as (fibrosis). These two pathological processes compromise the normal function of bone marrow resulting in decreased production of blood cells such as erythrocytes, granulocytes and megakaryocytes, the latter cells responsible for the production of platelets.
A Janus kinase inhibitor, also known as JAK inhibitor or jakinib, is a type of immune modulating medication, which inhibits the activity of one or more of the Janus kinase family of enzymes, thereby interfering with the JAK-STAT signaling pathway in lymphocytes.
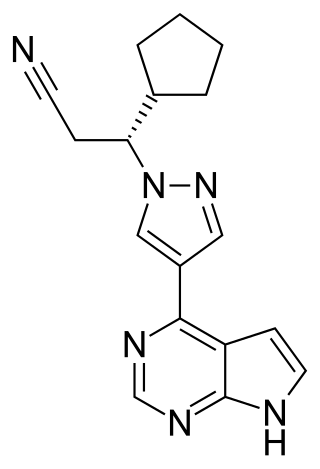
Ruxolitinib, sold under the brand name Jakafi among others, is a medication used for the treatment of intermediate or high-risk myelofibrosis, a type of myeloproliferative neoplasm that affects the bone marrow; polycythemia vera, when there has been an inadequate response to or intolerance of hydroxyurea; and steroid-refractory acute graft-versus-host disease. Ruxolitinib is a Janus kinase inhibitor. It was developed and marketed by Incyte Corp in the US under the brand name Jakafi, and by Novartis elsewhere in the world, under the brand name Jakavi.

Fedratinib, sold under the brand name Inrebic, is an anti-cancer medication used to treat myeloproliferative diseases including myelofibrosis. It is used in the form of fedratinib hydrochloride capsules that are taken by mouth. It is a semi-selective inhibitor of Janus kinase 2 (JAK-2). It was approved by the FDA on 16 August 2019.

Acalabrutinib, sold under the brand name Calquence, is a medication used to treat various types of non-Hodgkin lymphoma, including mantle cell lymphoma (MCL) and chronic lymphocytic leukemia/small lymphocytic Lymphoma (CLL/SLL). It may be used both in relapsed as well as in treatment-naive settings.

Alectinib (INN), sold under the brand name Alecensa, is an anticancer medication that is used to treat non-small-cell lung cancer (NSCLC). It blocks the activity of anaplastic lymphoma kinase (ALK). It is taken by mouth. It was developed by Chugai Pharmaceutical Co. Japan, which is part of the Hoffmann-La Roche group.
Inebilizumab, sold under the brand name Uplizna, is a medication for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adults. Inebilizumab is a humanized mAb that binds to and depletes CD19+ B cells including plasmablasts and plasma cells.
Burosumab, sold under the brand name Crysvita, is a human monoclonal antibody medication approved 2018 for the treatment of X-linked hypophosphatemia and tumor-induced osteomalacia.
Ravulizumab, sold under the brand name Ultomiris, is a humanized monoclonal antibody complement inhibitor medication designed for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uremic syndrome. It is designed to bind to and prevent the activation of Complement component 5 (C5).
Satralizumab, sold under the brand name Enspryng, is a humanized monoclonal antibody medication that is used for the treatment of neuromyelitis optica spectrum disorder (NMOSD), a rare autoimmune disease. The drug is being developed by Chugai Pharmaceutical, a subsidiary of Roche.
Sutimlimab, sold under the brand name Enjaymo, is a monoclonal antibody that is used to treat adults with cold agglutinin disease (CAD). It is given by intravenous infusion. Sutimlimab prevents complement-enhanced activation of autoimmune human B cells in vitro.

Pemigatinib, sold under the brand name Pemazyre, is an anti-cancer medication used for the treatment of bile duct cancer (cholangiocarcinoma). Pemigatinib works by blocking FGFR2 in tumor cells to prevent them from growing and spreading.

Ripretinib, sold under the brand name Qinlock, is a medication for the treatment of adults with advanced gastrointestinal stromal tumor (GIST), a type of tumor that originates in the gastrointestinal tract. It is taken by mouth. Ripretinib inhibits the activity of the kinases KIT and PDGFRA, which helps keep cancer cells from growing.
Tafasitamab, sold under the brand name Monjuvi, is a medication used in combination with lenalidomide for the treatment of adults with relapsed or refractory diffuse large B-cell lymphoma (DLBCL).
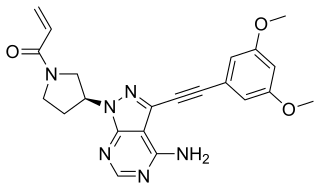
Futibatinib, sold under the brand name Lytgobi, is an anti-cancer medication used for the treatment of cholangiocarcinoma. It is a kinase inhibitor. It is taken by mouth.
Nadofaragene firadenovec, sold under the brand name Adstiladrin, is a gene therapy for the treatment of bladder cancer. It is a non-replicating adenoviral vector-based gene therapy.

Leniolisib (INN), sold under the brand name Joenja, is a medication used for the treatment of activated phosphoinositide 3-kinase delta syndrome (APDS). It is a kinase inhibitor that is taken by mouth.

Mavorixafor, sold under the brand name Xolremdi, is a medication used for the treatment of WHIM syndrome. It is a CXC chemokine receptor 4 antagonist. It is taken by mouth. It was developed by X4 Pharmaceuticals.
Lovotibeglogene autotemcel, sold under the brand name Lyfgenia, is a lentiviral gene therapy used for the treatment of sickle cell disease.









