Crizotinib, sold under the brand name Xalkori among others, is an anti-cancer medication used for the treatment of non-small cell lung carcinoma (NSCLC). Crizotinib inhibits the c-Met/Hepatocyte growth factor receptor (HGFR) tyrosine kinase, which is involved in the oncogenesis of a number of other histological forms of malignant neoplasms. It also acts as an ALK and ROS1 inhibitor.
Nivolumab, sold under the brand name Opdivo, is an anti-cancer medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urothelial carcinoma, colon cancer, esophageal squamous cell carcinoma, liver cancer, gastric cancer, and esophageal or gastroesophageal junction cancer. It is administered intravenously.

Ceritinib is a prescription-only drug used for the treatment of non-small cell lung cancer (NSCLC). It was developed by Novartis and received FDA approval for use in April 2014.

Atezolizumab, sold under the brand name Tecentriq among others, is a monoclonal antibody medication used to treat urothelial carcinoma, non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC), hepatocellular carcinoma and alveolar soft part sarcoma, but discontinued for use in triple-negative breast cancer (TNBC). It is a fully humanized, engineered monoclonal antibody of IgG1 isotype against the protein programmed cell death-ligand 1 (PD-L1).

Sacituzumab govitecan, sold under the brand name Trodelvy by Gilead Sciences, is a Trop-2-directed antibody and topoisomerase inhibitor drug conjugate used for the treatment of metastatic triple-negative breast cancer and metastatic urothelial cancer.

Osimertinib, sold under the brand name Tagrisso, is a medication used to treat non-small-cell lung carcinomas with specific mutations. It is a third-generation epidermal growth factor receptor tyrosine kinase inhibitor.

Alectinib (INN), sold under the brand name Alecensa, is an anticancer medication that is used to treat non-small-cell lung cancer (NSCLC). It blocks the activity of anaplastic lymphoma kinase (ALK). It is taken by mouth. It was developed by Chugai Pharmaceutical Co. Japan, which is part of the Hoffmann-La Roche group.

Entrectinib, sold under the brand name Rozlytrek, is an anti-cancer medication used to treat ROS1-positive non-small cell lung cancer and NTRK fusion-positive solid tumors. It is a selective tyrosine kinase inhibitor (TKI), of the tropomyosin receptor kinases (TRK) A, B and C, C-ros oncogene 1 (ROS1) and anaplastic lymphoma kinase (ALK).

Capmatinib, sold under the brand name Tabrecta, is an anticancer medication used for the treatment of metastatic non-small cell lung cancer whose tumors have a mutation that leads to the exon 14 skipping of the MET gene, which codes for the membrane receptor HGFR.

Lorlatinib, sold under the brand name Lorbrena in the United States, Canada, and Japan, and Lorviqua in the European Union, is an anti-cancer medication used for the treatment of non-small cell lung cancer. It is an orally administered inhibitor of anaplastic lymphoma kinase (ALK) and C-ros oncogene 1 (ROS1), two enzymes that play a role in the development of cancer. It was developed by Pfizer.
Cemiplimab, sold under the brand name Libtayo, is a monoclonal antibody medication for the treatment of squamous cell skin cancer. Cemiplimab belongs to a class of drugs that binds to the programmed death receptor-1 (PD-1), blocking the PD-1/PD-L1 pathway.

Tepotinib, sold under the brand name Tepmetko, is an anti-cancer medication used for the treatment of adults with non-small cell lung cancer (NSCLC).

Sotorasib, sold under the brand names Lumakras and Lumykras, is an anti-cancer medication used to treat non-small-cell lung cancer. It targets a specific mutation, G12C, in the protein K-Ras encoded by gene KRAS which is responsible for various forms of cancer. Sotorasib is an inhibitor of the RAS GTPase family.
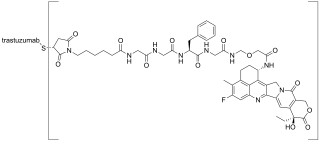
Trastuzumab deruxtecan, sold under the brand name Enhertu, is an antibody-drug conjugate consisting of the humanized monoclonal antibody trastuzumab (Herceptin) covalently linked to the topoisomerase I inhibitor deruxtecan. It is licensed for the treatment of breast cancer or gastric or gastroesophageal adenocarcinoma. Trastuzumab binds to and blocks signaling through epidermal growth factor receptor 2 (HER2/neu) on cancers that rely on it for growth. Additionally, once bound to HER2 receptors, the antibody is internalized by the cell, carrying the bound deruxtecan along with it, where it interferes with the cell's ability to make DNA structural changes and replicate its DNA during cell division, leading to DNA damage when the cell attempts to replicate itself, destroying the cell.

Pemigatinib, sold under the brand name Pemazyre, is an anti-cancer medication used for the treatment of bile duct cancer (cholangiocarcinoma). Pemigatinib works by blocking FGFR2 in tumor cells to prevent them from growing and spreading.

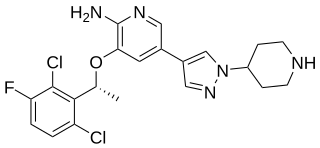
Selpercatinib, sold under the brand name Retevmo among others, is a medication for the treatment of cancers in people whose tumors have an alteration in a specific gene. It is taken by mouth.
Pertuzumab/trastuzumab/hyaluronidase, sold under the brand name Phesgo, is a fixed-dose combination medication to treat adults with HER2-positive breast cancer that has spread to other parts of the body, and for treatment of adults with early HER2-positive breast cancer. It contains pertuzumab, trastuzumab, and hyaluronidase–zzxf. It is injected under the skin via subcutaneous injection in the thigh. In the European Union, Phesgo contains the active ingredients pertuzumab and trastuzumab along with the enzyme vorhyaluronidase alfa.

Mobocertinib, sold under the brand name Exkivity, is used for the treatment of non-small cell lung cancer.

Adagrasib, sold under the brand name Krazati, is an anticancer medication used to treat non-small cell lung cancer. Adagrasib is an inhibitor of the RAS GTPase family. It is taken by mouth. It is being developed by Mirati Therapeutics.
Amivantamab, sold under the brand name Rybrevant, is a bispecific monoclonal antibody used to treat non-small cell lung cancer. Amivantamab is a bispecific epidermal growth factor (EGF) receptor-directed and mesenchymal–epithelial transition (MET) receptor-directed antibody. It is the first treatment for adults with non-small cell lung cancer whose tumors have specific types of genetic mutations: epidermal growth factor receptor (EGFR) exon 20 insertion mutations.

















