
In cellular biology, paracrine signaling is a form of cell signaling, a type of cellular communication in which a cell produces a signal to induce changes in nearby cells, altering the behaviour of those cells. Signaling molecules known as paracrine factors diffuse over a relatively short distance, as opposed to cell signaling by endocrine factors, hormones which travel considerably longer distances via the circulatory system; juxtacrine interactions; and autocrine signaling. Cells that produce paracrine factors secrete them into the immediate extracellular environment. Factors then travel to nearby cells in which the gradient of factor received determines the outcome. However, the exact distance that paracrine factors can travel is not certain.
Autocrine signaling is a form of cell signaling in which a cell secretes a hormone or chemical messenger that binds to autocrine receptors on that same cell, leading to changes in the cell. This can be contrasted with paracrine signaling, intracrine signaling, or classical endocrine signaling.
A biochemical cascade, also known as a signaling cascade or signaling pathway, is a series of chemical reactions that occur within a biological cell when initiated by a stimulus. This stimulus, known as a first messenger, acts on a receptor that is transduced to the cell interior through second messengers which amplify the signal and transfer it to effector molecules, causing the cell to respond to the initial stimulus. Most biochemical cascades are series of events, in which one event triggers the next, in a linear fashion. At each step of the signaling cascade, various controlling factors are involved to regulate cellular actions, in order to respond effectively to cues about their changing internal and external environments.

Catenins are a family of proteins found in complexes with cadherin cell adhesion molecules of animal cells. The first two catenins that were identified became known as α-catenin and β-catenin. α-Catenin can bind to β-catenin and can also bind filamentous actin (F-actin). β-Catenin binds directly to the cytoplasmic tail of classical cadherins. Additional catenins such as γ-catenin and δ-catenin have been identified. The name "catenin" was originally selected because it was suspected that catenins might link cadherins to the cytoskeleton.

Chondroblasts, or perichondrial cells, is the name given to mesenchymal progenitor cells in situ which, from endochondral ossification, will form chondrocytes in the growing cartilage matrix. Another name for them is subchondral cortico-spongious progenitors. They have euchromatic nuclei and stain by basic dyes.
Frzb is a Wnt-binding protein especially important in embryonic development. It is a competitor for the cell-surface G-protein receptor Frizzled.

Catenin beta-1, also known as β-catenin (beta-catenin), is a protein that in humans is encoded by the CTNNB1 gene.
In the field of developmental biology, regional differentiation is the process by which different areas are identified in the development of the early embryo. The process by which the cells become specified differs between organisms.

Frizzled is a family of atypical G protein-coupled receptors that serve as receptors in the Wnt signaling pathway and other signaling pathways. When activated, Frizzled leads to activation of Dishevelled in the cytosol.

Transcription factor 7-like 2 , also known as TCF7L2 or TCF4, is a protein acting as a transcription factor that, in humans, is encoded by the TCF7L2 gene. The TCF7L2 gene is located on chromosome 10q25.2–q25.3, contains 19 exons. As a member of the TCF family, TCF7L2 can form a bipartite transcription factor and influence several biological pathways, including the Wnt signalling pathway.

Axin-1 is a protein that in humans is encoded by the AXIN1 gene.

Lymphoid enhancer-binding factor 1 (LEF1) is a protein that in humans is encoded by the LEF1 gene. It is a member of T cell factor/lymphoid enhancer factor (TCF/LEF) family.

Proto-oncogene Wnt-1, or Proto-oncogene Int-1 homolog is a protein that in humans is encoded by the WNT1 gene.
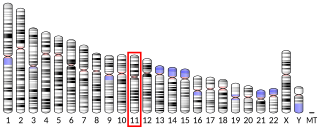
Low-density lipoprotein receptor-related protein 5 is a protein that in humans is encoded by the LRP5 gene. LRP5 is a key component of the LRP5/LRP6/Frizzled co-receptor group that is involved in canonical Wnt pathway. Mutations in LRP5 can lead to considerable changes in bone mass. A loss-of-function mutation causes osteoporosis pseudoglioma syndrome with a decrease in bone mass, while a gain-of-function mutation causes drastic increases in bone mass.

Secreted frizzled-related protein 1, also known as SFRP1, is a protein which in humans is encoded by the SFRP1 gene.

Protein Wnt-5a is a protein that in humans is encoded by the WNT5A gene.

Low-density lipoprotein receptor-related protein 6 is a protein that in humans is encoded by the LRP6 gene. LRP6 is a key component of the LRP5/LRP6/Frizzled co-receptor group that is involved in canonical Wnt pathway.

Protein Wnt-3a is a protein that in humans is encoded by the WNT3A gene.

Dishevelled (Dsh) is a family of proteins involved in canonical and non-canonical Wnt signalling pathways. Dsh is a cytoplasmic phosphoprotein that acts directly downstream of frizzled receptors. It takes its name from its initial discovery in flies, where a mutation in the dishevelled gene was observed to cause improper orientation of body and wing hairs. There are vertebrate homologs in zebrafish, Xenopus (Xdsh), mice and humans. Dsh relays complex Wnt signals in tissues and cells, in normal and abnormal contexts. It is thought to interact with the SPATS1 protein when regulating the Wnt Signalling pathway.

The TCF/LEF family is a group of genes that encode transcription factors which bind to DNA through a SOX-like high mobility group domain. They are involved in the Wnt signaling pathway, particularly during embryonic and stem-cell development, but also had been found to play a role in cancer and diabetes. TCF/LEF factors recruit the coactivator beta-catenin to enhancer elements of genes they target. They can also recruit members of the Groucho family of corepressors.





















