
Lipids are a broad group of organic compounds which include fats, waxes, sterols, fat-soluble vitamins, monoglycerides, diglycerides, phospholipids, and others. The functions of lipids include storing energy, signaling, and acting as structural components of cell membranes. Lipids have applications in the cosmetic and food industries, and in nanotechnology.
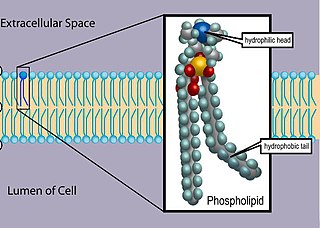
Phospholipids are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue. Marine phospholipids typically have omega-3 fatty acids EPA and DHA integrated as part of the phospholipid molecule. The phosphate group can be modified with simple organic molecules such as choline, ethanolamine or serine.
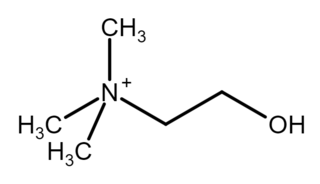
Choline is a cation with the chemical formula [(CH3)3NCH2CH2OH]+. Choline forms various salts, such as choline chloride and choline bitartrate. It is an essential nutrient for humans and many other animals, and is a structural part of phospholipids and cell membranes.

Among animals which produce eggs, the yolk is the nutrient-bearing portion of the egg whose primary function is to supply food for the development of the embryo. Some types of egg contain no yolk, for example because they are laid in situations where the food supply is sufficient or because the embryo develops in the parent's body, which supplies the food, usually through a placenta. Reproductive systems in which the mother's body supplies the embryo directly are said to be matrotrophic; those in which the embryo is supplied by yolk are said to be lecithotrophic. In many species, such as all birds, and most reptiles and insects, the yolk takes the form of a special storage organ constructed in the reproductive tract of the mother. In many other animals, especially very small species such as some fish and invertebrates, the yolk material is not in a special organ, but inside the egg cell.

Lecithin is a generic term to designate any group of yellow-brownish fatty substances occurring in animal and plant tissues which are amphiphilic – they attract both water and fatty substances, and are used for smoothing food textures, emulsifying, homogenizing liquid mixtures, and repelling sticking materials.
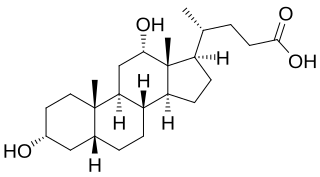
Deoxycholic acid is a bile acid. Deoxycholic acid is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. The two primary bile acids secreted by the liver are cholic acid and chenodeoxycholic acid. Bacteria metabolize chenodeoxycholic acid into the secondary bile acid lithocholic acid, and they metabolize cholic acid into deoxycholic acid. There are additional secondary bile acids, such as ursodeoxycholic acid. Deoxycholic acid is soluble in alcohol and acetic acid. When pure, it exists in a white to off-white crystalline powder form.

Phosphatidylinositol or inositol phospholipid is a biomolecule. It was initially called "inosite" when it was discovered by Léon Maquenne and Johann Joseph von Scherer in the late 19th century. It was discovered in bacteria but later also found in eukaryotes, and was found to be a signaling molecule.
Phosphatidic acids are anionic phospholipids important to cell signaling and direct activation of lipid-gated ion channels. Hydrolysis of phosphatidic acid gives rise to one molecule each of glycerol and phosphoric acid and two molecules of fatty acids. They constitute about 0.25% of phospholipids in the bilayer.
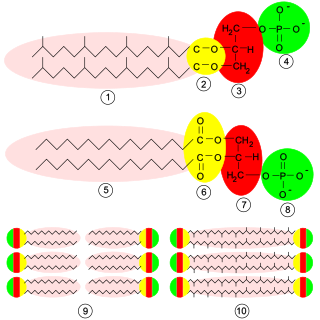
Glycerophospholipids or phosphoglycerides are glycerol-based phospholipids. They are the main component of biological membranes in eukaryotic cells. They are a type of lipid, of which its composition affects membrane structure and properties. Two major classes are known: those for bacteria and eukaryotes and a separate family for archaea.

Théodore (Nicolas) Gobley (French:[ɡɔblɛ]; 11 May 1811, in Paris – 1 September 1876, in Bagnères-de-Luchon, was the first to isolate and ultimately determine the chemical structure of lecithin, the first identified and characterized member of the phospholipids class. He was also a pioneer researcher in the study and analysis of the chemical components of brain tissues.

Dipalmitoylphosphatidylcholine (DPPC) is a phospholipid (and a lecithin) consisting of two C16 palmitic acid groups attached to a phosphatidylcholine head-group.
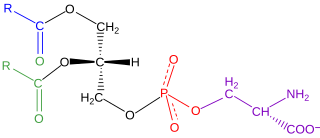
Phosphatidylserine is a phospholipid and is a component of the cell membrane. It plays a key role in cell cycle signaling, specifically in relation to apoptosis. It is a key pathway for viruses to enter cells via apoptotic mimicry. Its exposure on the outer surface of a membrane marks the cell for destruction via apoptosis.
Phospholipase D (EC 3.1.4.4, lipophosphodiesterase II, lecithinase D, choline phosphatase, PLD; systematic name phosphatidylcholine phosphatidohydrolase) is an anesthetic sensitive and mechanosensitive enzyme of the phospholipase superfamily that catalyses the following reaction

Phosphatidylethanolamine (PE) is a class of phospholipids found in biological membranes. They are synthesized by the addition of cytidine diphosphate-ethanolamine to diglycerides, releasing cytidine monophosphate. S-Adenosyl methionine can subsequently methylate the amine of phosphatidylethanolamines to yield phosphatidylcholines.
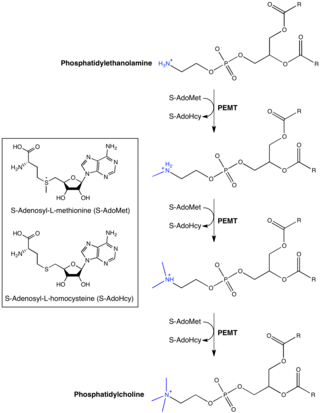
Phosphatidylethanolamine N-methyltransferase is a transferase enzyme which converts phosphatidylethanolamine (PE) to phosphatidylcholine (PC) in the liver. In humans it is encoded by the PEMT gene within the Smith–Magenis syndrome region on chromosome 17.
Sec14 is a cytosolic protein found in yeast which plays a role in the regulation of several cellular functions, specifically those related to intracellular transport. Encoded by the Sec14 gene, Sec14p may transport phosphatidylinositol and phosphatidylcholine produced in the endoplasmic reticulum and the Golgi body to other cellular membranes. Additionally, Sec14p potentially plays a role in the localization of lipid raft proteins. Sec14p is an essential gene in yeast, and is homologous in function to phosphatidylinositol transfer protein in mammals. A conditional mutant with non-functional Sec14p presents with Berkeley bodies and deficiencies in protein secretion.

Phosphatidylcholine transfer protein (PCTP), also known as StAR-related lipid transfer domain protein 2 (STARD2), is a specific intracellular phospholipid binding protein that can transfer phosphatidylcholine between different membranes in the cytosol.
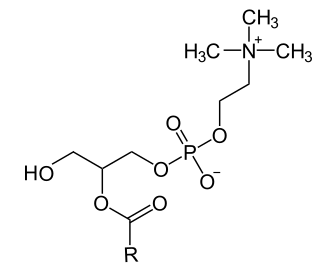
1-Lysophosphatidylcholines are a class of phospholipids that are intermediates in the metabolism of lipids. They result from the hydrolysis of an acyl group from the sn-1 position of phosphatidylcholine. They are also called 2-acyl-sn-glycero-3-phosphocholines. The synthesis of phosphatidylcholines with specific fatty acids occurs through the synthesis of 1-lysoPC. The formation of various other lipids generates 1-lysoPC as a by-product.

Egg lecithin is a type of lecithin, a group of compounds primarily containing phospholipids, that is derived from eggs.

The CDP-choline pathway, first identified by Eugene P. Kennedy in 1956, is the predominant mechanism by which mammalian cells synthesize phosphatidylcholine (PC) for incorporation into membranes or lipid-derived signalling molecules. The CDP-choline pathway represents one half of what is known as the Kennedy pathway. The other half is the CDP-ethanolamine pathway which is responsible for the biosynthesis of the phospholipid phosphatidylethanolamine (PE).























