
Pernicious anemia is a disease where not enough red blood cells are produced due to a deficiency of vitamin B12. Those affected often have a gradual onset. The most common initial symptoms are feeling tired and weak. Other symptoms may include shortness of breath, feeling faint, a smooth red tongue, pale skin, chest pain, nausea and vomiting, loss of appetite, heartburn, numbness in the hands and feet, difficulty walking, memory loss, muscle weakness, poor reflexes, blurred vision, clumsiness, depression, and confusion. Without treatment, some of these problems may become permanent.
Methylmalonic acidemias, also called methylmalonic acidurias, are a group of inherited metabolic disorders, that prevent the body from properly breaking down proteins and fats. This leads to a buildup of a toxic level of methylmalonic acid in body liquids and tissues. Due to the disturbed branched-chain amino acids (BCAA) metabolism, they are among the classical organic acidemias.
Megaloblastic anemia is a type of macrocytic anemia. An anemia is a red blood cell defect that can lead to an undersupply of oxygen. Megaloblastic anemia results from inhibition of DNA synthesis during red blood cell production. When DNA synthesis is impaired, the cell cycle cannot progress from the G2 growth stage to the mitosis (M) stage. This leads to continuing cell growth without division, which presents as macrocytosis. Megaloblastic anemia has a rather slow onset, especially when compared to that of other anemias. The defect in red cell DNA synthesis is most often due to hypovitaminosis, specifically vitamin B12 deficiency or folate deficiency. Loss of micronutrients may also be a cause.

Methionine synthase (MS, MeSe, MTR) is primarily responsible for the regeneration of methionine from homocysteine in most individuals. In humans it is encoded by the MTR gene (5-methyltetrahydrofolate-homocysteine methyltransferase). Methionine synthase forms part of the S-adenosylmethionine (SAMe) biosynthesis and regeneration cycle, and is the enzyme responsible for linking the cycle to one-carbon metabolism via the folate cycle. There are two primary forms of this enzyme, the Vitamin B12 (cobalamin)-dependent (MetH) and independent (MetE) forms, although minimal core methionine synthases that do not fit cleanly into either category have also been described in some anaerobic bacteria. The two dominant forms of the enzymes appear to be evolutionary independent and rely on considerably different chemical mechanisms. Mammals and other higher eukaryotes express only the cobalamin-dependent form. In contrast, the distribution of the two forms in Archaeplastida (plants and algae) is more complex. Plants exclusively possess the cobalamin-independent form, while algae have either one of the two, depending on species. Many different microorganisms express both the cobalamin-dependent and cobalamin-independent forms.

Methylmalonyl-CoA mutase is a mitochondrial homodimer apoenzyme that focuses on the catalysis of methylmalonyl CoA to succinyl CoA. The enzyme is bound to adenosylcobalamin, a hormonal derivative of vitamin B12 in order to function. Methylmalonyl-CoA mutase deficiency is caused by genetic defect in the MUT gene responsible for encoding the enzyme. Deficiency in this enzyme accounts for 60% of the cases of methylmalonic acidemia.

Methylcobalamin (mecobalamin, MeCbl, or MeB12) is a cobalamin, a form of vitamin B12. It differs from cyanocobalamin in that the cyano group at the cobalt is replaced with a methyl group. Methylcobalamin features an octahedral cobalt(III) centre and can be obtained as bright red crystals. From the perspective of coordination chemistry, methylcobalamin is notable as a rare example of a compound that contains metal–alkyl bonds. Nickel–methyl intermediates have been proposed for the final step of methanogenesis.

Methylmalonyl-CoA mutase (EC 5.4.99.2, MCM), mitochondrial, also known as methylmalonyl-CoA isomerase, is a protein that in humans is encoded by the MUT gene. This vitamin B12-dependent enzyme catalyzes the isomerization of methylmalonyl-CoA to succinyl-CoA in humans. Mutations in MUT gene may lead to various types of methylmalonic aciduria.

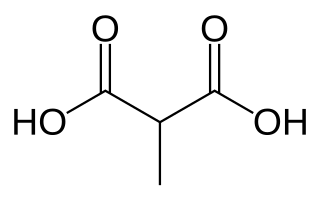
Methylmalonic acid (MMA) is a chemical compound from the group of dicarboxylic acids. It consists of the basic structure of malonic acid and also carries a methyl group. The salts of methylmalonic acid are called methylmalonates.

Hydroxocobalamin, also known as vitamin B12a and hydroxycobalamin, is a vitamin found in food and used as a dietary supplement. As a supplement it is used to treat vitamin B12 deficiency including pernicious anemia. Other uses include treatment for cyanide poisoning, Leber's optic atrophy, and toxic amblyopia. It is given by injection into a muscle or vein, by pill or sublingually.

Methylmalonyl-CoA is the thioester consisting of coenzyme A linked to methylmalonic acid. It is an important intermediate in the biosynthesis of succinyl-CoA, which plays an essential role in the tricarboxylic acid cycle.

Adenosylcobalamin (AdoCbl), also known as coenzyme B12, cobamamide, and dibencozide, is, along with methylcobalamin (MeCbl), one of the biologically active forms of vitamin B12.

Cobalamin riboswitch is a cis-regulatory element which is widely distributed in 5' untranslated regions of vitamin B12 (Cobalamin) related genes in bacteria.

Vitamin B12 deficiency, also known as cobalamin deficiency, is the medical condition in which the blood and tissue have a lower than normal level of vitamin B12. Symptoms can vary from none to severe. Mild deficiency may have few or absent symptoms. In moderate deficiency, feeling tired, headaches, soreness of the tongue, mouth ulcers, breathlessness, feeling faint, rapid heartbeat, low blood pressure, pallor, hair loss, decreased ability to think and severe joint pain and the beginning of neurological symptoms, including abnormal sensations such as pins and needles, numbness and tinnitus may occur. Severe deficiency may include symptoms of reduced heart function as well as more severe neurological symptoms, including changes in reflexes, poor muscle function, memory problems, blurred vision, irritability, ataxia, decreased smell and taste, decreased level of consciousness, depression, anxiety, guilt and psychosis. If left untreated, some of these changes can become permanent. Temporary infertility, reversible with treatment, may occur. A late finding type of anemia known as megaloblastic anemia is often but not always present. In exclusively breastfed infants of vegan mothers, undetected and untreated deficiency can lead to poor growth, poor development, and difficulties with movement.

Vitamin B12, also known as cobalamin, is a water-soluble vitamin involved in metabolism. It is one of eight B vitamins. It is required by animals, which use it as a cofactor in DNA synthesis, and in both fatty acid and amino acid metabolism. It is important in the normal functioning of the nervous system via its role in the synthesis of myelin, and in the circulatory system in the maturation of red blood cells in the bone marrow. Plants do not need cobalamin and carry out the reactions with enzymes that are not dependent on it.

Methylmalonic aciduria type A protein, mitochondrial also known as MMAA is a protein that in humans is encoded by the MMAA gene.

Methylmalonic aciduria and homocystinuria type C protein (MMACHC) is a protein that in humans is encoded by the MMACHC gene.

Imerslund–Gräsbeck syndrome is a rare autosomal recessive, familial form of vitamin B12 deficiency caused by malfunction of the "Cubam" receptor located in the terminal ileum. This receptor is composed of two proteins, amnionless (AMN), and cubilin. A defect in either of these protein components can cause this syndrome. This is a rare disease, with a prevalence about 1 in 200,000, and is usually seen in patients of European ancestry.

In molecular biology, the vitamin B12-binding domain is a protein domain which binds to cobalamin. It can bind two different forms of the cobalamin cofactor, with cobalt bonded either to a methyl group (methylcobalamin) or to 5'-deoxyadenosine (adenosylcobalamin). Cobalamin-binding domains are mainly found in two families of enzymes present in animals and prokaryotes, which perform distinct kinds of reactions at the cobalt-carbon bond. Enzymes that require methylcobalamin carry out methyl transfer reactions. Enzymes that require adenosylcobalamin catalyse reactions in which the first step is the cleavage of adenosylcobalamin to form cob(II)alamin and the 5'-deoxyadenosyl radical, and thus act as radical generators. In both types of enzymes the B12-binding domain uses a histidine to bind the cobalt atom of cobalamin cofactors. This histidine is embedded in a DXHXXG sequence, the most conserved primary sequence motif of the domain. Proteins containing the cobalamin-binding domain include:

Cobalamin biosynthesis is the process by which bacteria and archea make cobalamin, vitamin B12. Many steps are involved in converting aminolevulinic acid via uroporphyrinogen III and adenosylcobyric acid to the final forms in which it is used by enzymes in both the producing organisms and other species, including humans who acquire it through their diet.

Cobalt is essential to the metabolism of all animals. It is a key constituent of cobalamin, also known as vitamin B12, the primary biological reservoir of cobalt as an ultratrace element. Bacteria in the stomachs of ruminant animals convert cobalt salts into vitamin B12, a compound which can only be produced by bacteria or archaea. A minimal presence of cobalt in soils therefore markedly improves the health of grazing animals, and an uptake of 0.20 mg/kg a day is recommended because they have no other source of vitamin B12.



















