
Sulfur (also spelled sulphur in British English) is a chemical element; it has symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with the chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature.
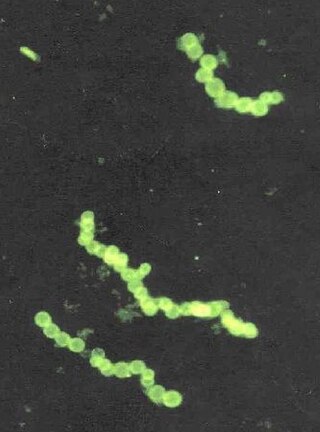
Thiomargarita namibiensis is a harmless, gram-negative, facultative anaerobic, coccoid bacterium found in the ocean sediments of the continental shelf of Namibia. The genus name Thiomargarita means "sulfur pearl." This refers to the appearance of the cells as they contain microscopic sulfur granules that scatter incident light, lending the cell a pearly luster. This causes the cells to form chains, resembling strings of pearls. The species name namibiensis means "of Namibia", an ode to their country of discovery and existence. Together, Thiomargarita namibiensis means “Sulfur pearl of Namibia".
Anaerobic respiration is respiration using electron acceptors other than molecular oxygen (O2). Although oxygen is not the final electron acceptor, the process still uses a respiratory electron transport chain.

The Chromatiaceae are one of the two families of purple sulfur bacteria, together with the Ectothiorhodospiraceae. They belong to the order Chromatiales of the class Gammaproteobacteria, which is composed by unicellular Gram-negative organisms. Most of the species are photolithoautotrophs and conduct an anoxygenic photosynthesis, but there are also representatives capable of growing under dark and/or microaerobic conditions as either chemolithoautotrophs or chemoorganoheterotrophs.

Sulfate-reducing microorganisms (SRM) or sulfate-reducing prokaryotes (SRP) are a group composed of sulfate-reducing bacteria (SRB) and sulfate-reducing archaea (SRA), both of which can perform anaerobic respiration utilizing sulfate (SO2−
4) as terminal electron acceptor, reducing it to hydrogen sulfide (H2S). Therefore, these sulfidogenic microorganisms "breathe" sulfate rather than molecular oxygen (O2), which is the terminal electron acceptor reduced to water (H2O) in aerobic respiration.
In chemistry, disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation state. The reverse of disproportionation, such as when a compound in an intermediate oxidation state is formed from precursors of lower and higher oxidation states, is called comproportionation, also known as symproportionation.
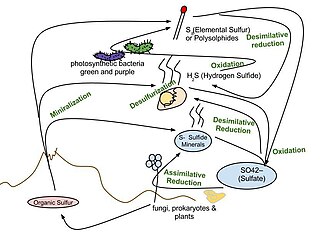
The important sulfur cycle is a biogeochemical cycle in which the sulfur moves between rocks, waterways and living systems. It is important in geology as it affects many minerals and in life because sulfur is an essential element (CHNOPS), being a constituent of many proteins and cofactors, and sulfur compounds can be used as oxidants or reductants in microbial respiration. The global sulfur cycle involves the transformations of sulfur species through different oxidation states, which play an important role in both geological and biological processes. Steps of the sulfur cycle are:

Sulfur-reducing bacteria are microorganisms able to reduce elemental sulfur (S0) to hydrogen sulfide (H2S). These microbes use inorganic sulfur compounds as electron acceptors to sustain several activities such as respiration, conserving energy and growth, in absence of oxygen. The final product of these processes, sulfide, has a considerable influence on the chemistry of the environment and, in addition, is used as electron donor for a large variety of microbial metabolisms. Several types of bacteria and many non-methanogenic archaea can reduce sulfur. Microbial sulfur reduction was already shown in early studies, which highlighted the first proof of S0 reduction in a vibrioid bacterium from mud, with sulfur as electron acceptor and H
2 as electron donor. The first pure cultured species of sulfur-reducing bacteria, Desulfuromonas acetoxidans, was discovered in 1976 and described by Pfennig Norbert and Biebel Hanno as an anaerobic sulfur-reducing and acetate-oxidizing bacterium, not able to reduce sulfate. Only few taxa are true sulfur-reducing bacteria, using sulfur reduction as the only or main catabolic reaction. Normally, they couple this reaction with the oxidation of acetate, succinate or other organic compounds. In general, sulfate-reducing bacteria are able to use both sulfate and elemental sulfur as electron acceptors. Thanks to its abundancy and thermodynamic stability, sulfate is the most studied electron acceptor for anaerobic respiration that involves sulfur compounds. Elemental sulfur, however, is very abundant and important, especially in deep-sea hydrothermal vents, hot springs and other extreme environments, making its isolation more difficult. Some bacteria – such as Proteus, Campylobacter, Pseudomonas and Salmonella – have the ability to reduce sulfur, but can also use oxygen and other terminal electron acceptors.

Beggiatoa is a genus of Gammaproteobacteria belonging to the order Thiotrichales, in the Pseudomonadota phylum. These bacteria form colorless filaments composed of cells that can be up to 200 μm in diameter, and are one of the largest prokaryotes on Earth. Beggiatoa are chemolithotrophic sulfur-oxidizers, using reduced sulfur species as an energy source. They live in sulfur-rich environments such as soil, both marine and freshwater, in the deep sea hydrothermal vents, and in polluted marine environments. In association with other sulfur bacteria, e.g. Thiothrix, they can form biofilms that are visible to the naked eye as mats of long white filaments; the white color is due to sulfur globules stored inside the cells.
Microbial metabolism is the means by which a microbe obtains the energy and nutrients it needs to live and reproduce. Microbes use many different types of metabolic strategies and species can often be differentiated from each other based on metabolic characteristics. The specific metabolic properties of a microbe are the major factors in determining that microbe's ecological niche, and often allow for that microbe to be useful in industrial processes or responsible for biogeochemical cycles.
Anaerobic oxidation of methane (AOM) is a methane-consuming microbial process occurring in anoxic marine and freshwater sediments. AOM is known to occur among mesophiles, but also in psychrophiles, thermophiles, halophiles, acidophiles, and alkophiles. During AOM, methane is oxidized with different terminal electron acceptors such as sulfate, nitrate, nitrite and metals, either alone or in syntrophy with a partner organism.
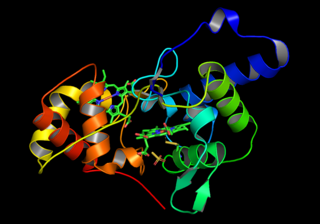
Thiosulfate dehydrogenase is an enzyme that catalyzes the chemical reaction:
Sulfur is metabolized by all organisms, from bacteria and archaea to plants and animals. Sulfur can have an oxidation state from -2 to +6 and is reduced or oxidized by a diverse range of organisms. The element is present in proteins, sulfate esters of polysaccharides, steroids, phenols, and sulfur-containing coenzymes.
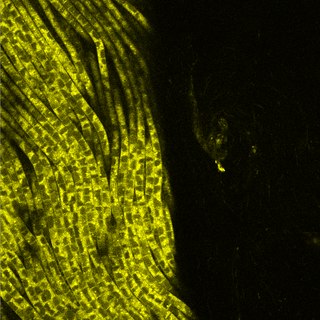
Thioploca is a genus of filamentous sulphur-oxidizing bacteria, in the order Thiotrichales. They inhabit both marine and freshwater environments, forming vast communities off the Pacific coast of South America and in other areas with a high organic matter sedimentation and bottom waters rich in nitrate and poor in oxygen. Their cells contain large vacuoles that occupy more than 80% of the cellular volume, used to store nitrate to oxidize sulphur for anaerobic respiration in the absence of oxygen, an important characteristic of the genus. With cell diameters ranging from 15-40 μm, they are some of the largest bacteria known. They provide an important link between the nitrogen and sulphur cycles, because they use both sulfur and nitrogen compounds. They secrete a sheath of mucus which they use as a tunnel to travel between sulphide-containing sediment and nitrate-containing sea water.
Sulfurimonas is a bacterial genus within the class of Campylobacterota, known for reducing nitrate, oxidizing both sulfur and hydrogen, and containing Group IV hydrogenases. This genus consists of four species: Sulfurimonas autorophica, Sulfurimonas denitrificans, Sulfurimonas gotlandica, and Sulfurimonas paralvinellae. The genus' name is derived from "sulfur" in Latin and "monas" from Greek, together meaning a “sulfur-oxidizing rod”. The size of the bacteria varies between about 1.5-2.5 μm in length and 0.5-1.0 μm in width. Members of the genus Sulfurimonas are found in a variety of different environments which include deep sea-vents, marine sediments, and terrestrial habitats. Their ability to survive in extreme conditions is attributed to multiple copies of one enzyme. Phylogenetic analysis suggests that members of the genus Sulfurimonas have limited dispersal ability and its speciation was affected by geographical isolation rather than hydrothermal composition. Deep ocean currents affect the dispersal of Sulfurimonas spp., influencing its speciation. As shown in the MLSA report of deep-sea hydrothermal vents Campylobacterota, Sulfurimonas has a higher dispersal capability compared with deep sea hydrothermal vent thermophiles, indicating allopatric speciation.
Desulfobulbus propionicus is a Gram-negative, anaerobic chemoorganotroph. Three separate strains have been identified: 1pr3T, 2pr4, and 3pr10. It is also the first pure culture example of successful disproportionation of elemental sulfur to sulfate and sulfide. Desulfobulbus propionicus has the potential to produce free energy and chemical products.

Acidithiobacillus thiooxidans, formerly known as Thiobacillus thiooxidans until its reclassification into the newly designated genus Acidithiobacillus of the Acidithiobacillia subclass of Pseudomonadota, is a Gram-negative, rod-shaped bacterium that uses sulfur as its primary energy source. It is mesophilic, with a temperature optimum of 28 °C. This bacterium is commonly found in soil, sewer pipes, and cave biofilms called snottites. A. thiooxidans is used in the mining technique known as bioleaching, where metals are extracted from their ores through the action of microbes.
The sulfate-methane transition zone (SMTZ) is a zone in oceans, lakes, and rivers typically found below the sediment surface in which sulfate and methane coexist. The formation of a SMTZ is driven by the diffusion of sulfate down the sediment column and the diffusion of methane up the sediments. At the SMTZ, their diffusion profiles meet and sulfate and methane react with one another, which allows the SMTZ to harbor a unique microbial community whose main form of metabolism is anaerobic oxidation of methane (AOM). The presence of AOM marks the transition from dissimilatory sulfate reduction to methanogenesis as the main metabolism utilized by organisms.

The hydrothermal vent microbial community includes all unicellular organisms that live and reproduce in a chemically distinct area around hydrothermal vents. These include organisms in the microbial mat, free floating cells, or bacteria in an endosymbiotic relationship with animals. Chemolithoautotrophic bacteria derive nutrients and energy from the geological activity at Hydrothermal vents to fix carbon into organic forms. Viruses are also a part of the hydrothermal vent microbial community and their influence on the microbial ecology in these ecosystems is a burgeoning field of research.
Sulfur isotope biogeochemistry is the study of the distribution of sulfur isotopes in biological and geological materials. In addition to its common isotope, 32S, sulfur has three rare stable isotopes: 34S, 36S, and 33S. The distribution of these isotopes in the environment is controlled by many biochemical and physical processes, including biological metabolisms, mineral formation processes, and atmospheric chemistry. Measuring the abundance of sulfur stable isotopes in natural materials, like bacterial cultures, minerals, or seawater, can reveal information about these processes both in the modern environment and over Earth history.













