
Meconium aspiration syndrome (MAS) also known as neonatal aspiration of meconium is a medical condition affecting newborn infants. It describes the spectrum of disorders and pathophysiology of newborns born in meconium-stained amniotic fluid (MSAF) and have meconium within their lungs. Therefore, MAS has a wide range of severity depending on what conditions and complications develop after parturition. Furthermore, the pathophysiology of MAS is multifactorial and extremely complex which is why it is the leading cause of morbidity and mortality in term infants.

Intrauterine growth restriction (IUGR), or fetal growth restriction, refers to poor growth of a fetus while in the womb during pregnancy. IUGR is defined by clinical features of malnutrition and evidence of reduced growth regardless of an infant's birth weight percentile. The causes of IUGR are broad and may involve maternal, fetal, or placental complications.

Patent ductus arteriosus (PDA) is a medical condition in which the ductus arteriosus fails to close after birth: this allows a portion of oxygenated blood from the left heart to flow back to the lungs through the aorta, which has a higher blood pressure, to the pulmonary artery, which has a lower blood pressure. Symptoms are uncommon at birth and shortly thereafter, but later in the first year of life there is often the onset of an increased work of breathing and failure to gain weight at a normal rate. With time, an uncorrected PDA usually leads to pulmonary hypertension followed by right-sided heart failure.
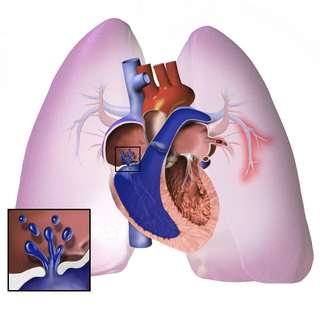
Pulmonary hypertension is a condition of increased blood pressure in the arteries of the lungs. Symptoms include shortness of breath, fainting, tiredness, chest pain, swelling of the legs, and a fast heartbeat. The condition may make it difficult to exercise. Onset is typically gradual.
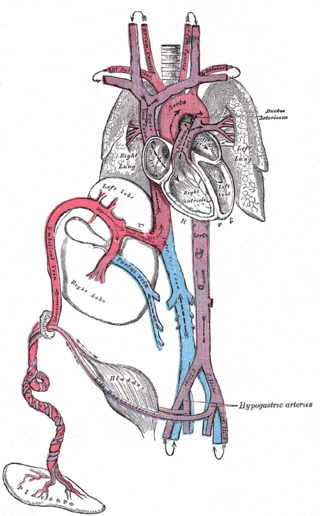
The ductus arteriosus, also called the ductus Botalli, named after the Italian physiologist Leonardo Botallo, is a blood vessel in the developing fetus connecting the trunk of the pulmonary artery to the proximal descending aorta. It allows most of the blood from the right ventricle to bypass the fetus's fluid-filled non-functioning lungs. Upon closure at birth, it becomes the ligamentum arteriosum.
Blue baby syndrome can refer to conditions that cause cyanosis, or blueness of the skin, in babies as a result of low oxygen levels in the blood. This term has traditionally been applied to cyanosis as a result of:
- Cyanotic heart disease, which is a category of congenital heart defect that results in low levels of oxygen in the blood. This can be caused by either reduced blood flow to the lungs or mixing of oxygenated and deoxygenated blood.
- Methemoglobinemia, which is a disease defined by high levels of methemoglobin in the blood. Increased levels of methemoglobin prevent oxygen from being released into the tissues and result in hypoxemia.

Infantile respiratory distress syndrome (IRDS), also called respiratory distress syndrome of newborn, or increasingly surfactant deficiency disorder (SDD), and previously called hyaline membrane disease (HMD), is a syndrome in premature infants caused by developmental insufficiency of pulmonary surfactant production and structural immaturity in the lungs. It can also be a consequence of neonatal infection and can result from a genetic problem with the production of surfactant-associated proteins.
At the end of pregnancy, the fetus must take the journey of childbirth to leave the reproductive mother. Upon its entry to the air-breathing world, the newborn must begin to adjust to life outside the uterus. This is true for all viviparous animals; this article discusses humans as the most-researched example.
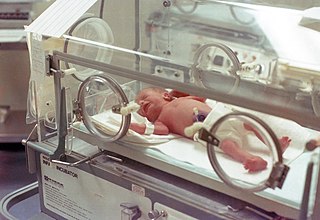
A neonatal intensive care unit (NICU), also known as an intensive care nursery (ICN), is an intensive care unit (ICU) specializing in the care of ill or premature newborn infants. The NICU is divided into several areas, including a critical care area for babies who require close monitoring and intervention, an intermediate care area for infants who are stable but still require specialized care, and a step down unit where babies who are ready to leave the hospital can receive additional care before being discharged.

Hydrops fetalis or hydrops foetalis is a condition in the fetus characterized by an accumulation of fluid, or edema, in at least two fetal compartments. By comparison, hydrops allantois or hydrops amnion is an accumulation of excessive fluid in the allantoic or amniotic space, respectively.
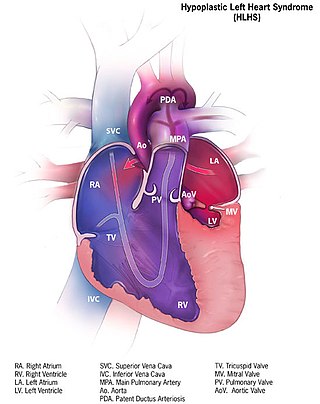
Hypoplastic left heart syndrome (HLHS) is a rare congenital heart defect in which the left side of the heart is severely underdeveloped and incapable of supporting the systemic circulation. It is estimated to account for 2-3% of all congenital heart disease. Early signs and symptoms include poor feeding, cyanosis, and diminished pulse in the extremities. The etiology is believed to be multifactorial resulting from a combination of genetic mutations and defects resulting in altered blood flow in the heart. Several structures can be affected including the left ventricle, aorta, aortic valve, or mitral valve all resulting in decreased systemic blood flow.

Congenital diaphragmatic hernia (CDH) is a birth defect of the diaphragm. The most common type of CDH is a Bochdalek hernia; other types include Morgagni hernia, diaphragm eventration and central tendon defects of the diaphragm. Malformation of the diaphragm allows the abdominal organs to push into the chest cavity, hindering proper lung formation.

Pulmonary atresia is a congenital malformation of the pulmonary valve in which the valve orifice fails to develop. The valve is completely closed thereby obstructing the outflow of blood from the heart to the lungs. The pulmonary valve is located on the right side of the heart between the right ventricle and pulmonary artery. In a normal functioning heart, the opening to the pulmonary valve has three flaps that open and close.

In humans, the circulatory system is different before and after birth. The fetal circulation is composed of the placenta, umbilical blood vessels encapsulated by the umbilical cord, heart and systemic blood vessels. A major difference between the fetal circulation and postnatal circulation is that the lungs are not used during the fetal stage resulting in the presence of shunts to move oxygenated blood and nutrients from the placenta to the fetal tissue. At birth, the start of breathing and the severance of the umbilical cord prompt various changes that quickly transform fetal circulation into postnatal circulation.
Respiratory diseases, or lung diseases, are pathological conditions affecting the organs and tissues that make gas exchange difficult in air-breathing animals. They include conditions of the respiratory tract including the trachea, bronchi, bronchioles, alveoli, pleurae, pleural cavity, the nerves and muscles of respiration. Respiratory diseases range from mild and self-limiting, such as the common cold, influenza, and pharyngitis to life-threatening diseases such as bacterial pneumonia, pulmonary embolism, tuberculosis, acute asthma, lung cancer, and severe acute respiratory syndromes, such as COVID-19. Respiratory diseases can be classified in many different ways, including by the organ or tissue involved, by the type and pattern of associated signs and symptoms, or by the cause of the disease.

Intrauterine hypoxia occurs when the fetus is deprived of an adequate supply of oxygen. It may be due to a variety of reasons such as prolapse or occlusion of the umbilical cord, placental infarction, maternal diabetes and maternal smoking. Intrauterine growth restriction may cause or be the result of hypoxia. Intrauterine hypoxia can cause cellular damage that occurs within the central nervous system. This results in an increased mortality rate, including an increased risk of sudden infant death syndrome (SIDS). Oxygen deprivation in the fetus and neonate have been implicated as either a primary or as a contributing risk factor in numerous neurological and neuropsychiatric disorders such as epilepsy, attention deficit hyperactivity disorder, eating disorders and cerebral palsy.

Pulmonary hypoplasia is incomplete development of the lungs, resulting in an abnormally low number or small size of bronchopulmonary segments or alveoli. A congenital malformation, it most often occurs secondary to other fetal abnormalities that interfere with normal development of the lungs. Primary (idiopathic) pulmonary hypoplasia is rare and usually not associated with other maternal or fetal abnormalities.
Alveolar capillary dysplasia (ACD) is a rare, congenital diffuse lung disease characterized by abnormal blood vessels in the lungs that cause highly elevated pulmonary blood pressure and an inability to effectively oxygenate and remove carbon dioxide from the blood. ACD typically presents in newborn babies within hours of birth as rapid and labored breathing, blue-colored lips or skin, quickly leading to respiratory failure and death. Atypical forms of ACD have been reported with initially milder symptoms and survival of many months before the onset of respiratory failure or lung transplantation.
Major aortopulmonary collateral arteries are arteries that develop to supply blood to the lungs when native pulmonary circulation is underdeveloped. Instead of coming from the pulmonary trunk, supply develops from the aorta and other systemic arteries.
A high-risk pregnancy is one where the mother or the fetus has an increased risk of adverse outcomes compared to uncomplicated pregnancies. No concrete guidelines currently exist for distinguishing “high-risk” pregnancies from “low-risk” pregnancies; however, there are certain studied conditions that have been shown to put the mother or fetus at a higher risk of poor outcomes. These conditions can be classified into three main categories: health problems in the mother that occur before she becomes pregnant, health problems in the mother that occur during pregnancy, and certain health conditions with the fetus.
















