
Insulin is a peptide hormone produced by beta cells of the pancreatic islets encoded in humans by the insulin (INS) gene. It is the main anabolic hormone of the body. It regulates the metabolism of carbohydrates, fats, and protein by promoting the absorption of glucose from the blood into cells of the liver, fat, and skeletal muscles. In these tissues the absorbed glucose is converted into either glycogen, via glycogenesis, or fats (triglycerides), via lipogenesis; in the liver, glucose is converted into both. Glucose production and secretion by the liver are strongly inhibited by high concentrations of insulin in the blood. Circulating insulin also affects the synthesis of proteins in a wide variety of tissues. It is thus an anabolic hormone, promoting the conversion of small molecules in the blood into large molecules in the cells. Low insulin in the blood has the opposite effect, promoting widespread catabolism, especially of reserve body fat.
Insulin resistance (IR) is a pathological condition in which cells in insulin-sensitive tissues in the body fail to respond normally to the hormone insulin or downregulate insulin receptors in response to hyperinsulinemia.

5' AMP-activated protein kinase or AMPK or 5' adenosine monophosphate-activated protein kinase is an enzyme that plays a role in cellular energy homeostasis, largely to activate glucose and fatty acid uptake and oxidation when cellular energy is low. It belongs to a highly conserved eukaryotic protein family and its orthologues are SNF1 in yeast, and SnRK1 in plants. It consists of three proteins (subunits) that together make a functional enzyme, conserved from yeast to humans. It is expressed in a number of tissues, including the liver, brain, and skeletal muscle. In response to binding AMP and ADP, the net effect of AMPK activation is stimulation of hepatic fatty acid oxidation, ketogenesis, stimulation of skeletal muscle fatty acid oxidation and glucose uptake, inhibition of cholesterol synthesis, lipogenesis, and triglyceride synthesis, inhibition of adipocyte lipogenesis, inhibition of adipocyte lipolysis, and modulation of insulin secretion by pancreatic β-cells.

In biology, a connexon, also known as a connexin hemichannel, is an assembly of six proteins called connexins that form the pore for a gap junction between the cytoplasm of two adjacent cells. This channel allows for bidirectional flow of ions and signaling molecules. The connexon is the hemichannel supplied by a cell on one side of the junction; two connexons from opposing cells normally come together to form the complete intercellular gap junction channel. In some cells, the hemichannel itself is active as a conduit between the cytoplasm and the extracellular space, allowing the transference of ions and small molecules lower than 1-2 KDa. Little is known about this function of connexons besides the new evidence suggesting their key role in intracellular signaling. In still other cells connexons have been shown to occur in mitochondrial membranes and appear to play a role in heart ischaemia.
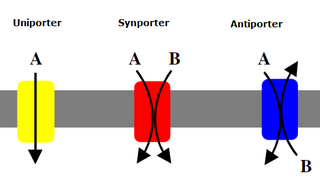
Uniporters, also known as solute carriers or facilitated transporters, are a type of membrane transport protein that passively transports solutes across a cell membrane. It uses facilitated diffusion for the movement of solutes down their concentration gradient from an area of high concentration to an area of low concentration. Unlike active transport, it does not require energy in the form of ATP to function. Uniporters are specialized to carry one specific ion or molecule and can be categorized as either channels or carriers. Facilitated diffusion may occur through three mechanisms: uniport, symport, or antiport. The difference between each mechanism depends on the direction of transport, in which uniport is the only transport not coupled to the transport of another solute.

Hyperinsulinemia is a condition in which there are excess levels of insulin circulating in the blood relative to the level of glucose. While it is often mistaken for diabetes or hyperglycaemia, hyperinsulinemia can result from a variety of metabolic diseases and conditions, as well as non-nutritive sugars in the diet. While hyperinsulinemia is often seen in people with early stage type 2 diabetes mellitus, it is not the cause of the condition and is only one symptom of the disease. Type 1 diabetes only occurs when pancreatic beta-cell function is impaired. Hyperinsulinemia can be seen in a variety of conditions including diabetes mellitus type 2, in neonates and in drug-induced hyperinsulinemia. It can also occur in congenital hyperinsulinism, including nesidioblastosis.
Glucose transporters are a wide group of membrane proteins that facilitate the transport of glucose across the plasma membrane, a process known as facilitated diffusion. Because glucose is a vital source of energy for all life, these transporters are present in all phyla. The GLUT or SLC2A family are a protein family that is found in most mammalian cells. 14 GLUTS are encoded by the human genome. GLUT is a type of uniporter transporter protein.
Method of glucose uptake differs throughout tissues depending on two factors; the metabolic needs of the tissue and availability of glucose. The two ways in which glucose uptake can take place are facilitated diffusion and secondary active transport. Active transport is the movement of ions or molecules going against the concentration gradient.

Blood sugar regulation is the process by which the levels of blood sugar, the common name for glucose dissolved in blood plasma, are maintained by the body within a narrow range.
The Randle cycle, also known as the glucose fatty-acid cycle, is a metabolic process involving the cross inhibition of glucose and fatty acids for substrates. It is theorized to play a role in explaining type 2 diabetes and insulin resistance.

Rac1, also known as Ras-related C3 botulinum toxin substrate 1, is a protein found in human cells. It is encoded by the RAC1 gene. This gene can produce a variety of alternatively spliced versions of the Rac1 protein, which appear to carry out different functions.

AS160, which was originally known as TBC1 domain family member 4 (TBC1D4), is a Rab GTPase-activating protein that in humans is encoded by the TBC1D4 gene.
LanC-like protein 2 is a protein that in humans is encoded by the LANCL2 gene. It is a protein broadly expressed in the plasma a nuclear membranes of immune, epithelial and muscle cells and a potential therapeutic target for chronic inflammatory, metabolic and immune-mediated diseases such as Crohn's disease and diabetes.

Chemerin, also known as retinoic acid receptor responder protein 2 (RARRES2), tazarotene-induced gene 2 protein (TIG2), or RAR-responsive protein TIG2 is a protein that in humans is encoded by the RARRES2 gene.
The insulin transduction pathway is a biochemical pathway by which insulin increases the uptake of glucose into fat and muscle cells and reduces the synthesis of glucose in the liver and hence is involved in maintaining glucose homeostasis. This pathway is also influenced by fed versus fasting states, stress levels, and a variety of other hormones.
Krüppel-like factor 15 is a protein that in humans is encoded by the KLF15 gene in the Krüppel-like factor family. Its former designation KKLF stands for kidney-enriched Krüppel-like factor.
Lipotoxicity is a metabolic syndrome that results from the accumulation of lipid intermediates in non-adipose tissue, leading to cellular dysfunction and death. The tissues normally affected include the kidneys, liver, heart and skeletal muscle. Lipotoxicity is believed to have a role in heart failure, obesity, and diabetes, and is estimated to affect approximately 25% of the adult American population.
In molecular biology, TUG-UBL1 refers to a protein that regulates a glucose transporter called GLUT4. TUG-UBL1 is an acronym for Tether containing UBX domain for GLUT4-Ubiquitin Like 1, this is encoded for by the gene, ASPSCR1.

Ras-related protein Rab-4B is a protein that in humans is encoded by the RAB4B gene.
Mark Anthony Febbraio is a physiologist, academic and researcher. He is a professor and head of the Cellular and Molecular Metabolism Laboratory at the Monash Institute of Pharmaceutical Sciences (MIPS) of Monash University and a Senior Principal Research Fellow of the National Health and Medical Research Council (NHMRC).























