Exocytosis is a form of active transport and bulk transport in which a cell transports molecules out of the cell. As an active transport mechanism, exocytosis requires the use of energy to transport material. Exocytosis and its counterpart, endocytosis, are used by all cells because most chemical substances important to them are large polar molecules that cannot pass through the hydrophobic portion of the cell membrane by passive means. Exocytosis is the process by which a large amount of molecules are released; thus it is a form of bulk transport. Exocytosis occurs via secretory portals at the cell plasma membrane called porosomes. Porosomes are permanent cup-shaped lipoprotein structures at the cell plasma membrane, where secretory vesicles transiently dock and fuse to release intra-vesicular contents from the cell.

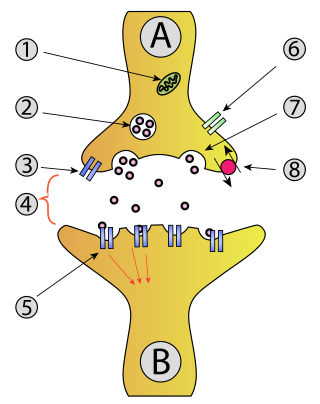
In a neuron, synaptic vesicles store various neurotransmitters that are released at the synapse. The release is regulated by a voltage-dependent calcium channel. Vesicles are essential for propagating nerve impulses between neurons and are constantly recreated by the cell. The area in the axon that holds groups of vesicles is an axon terminal or "terminal bouton". Up to 130 vesicles can be released per bouton over a ten-minute period of stimulation at 0.2 Hz. In the visual cortex of the human brain, synaptic vesicles have an average diameter of 39.5 nanometers (nm) with a standard deviation of 5.1 nm.
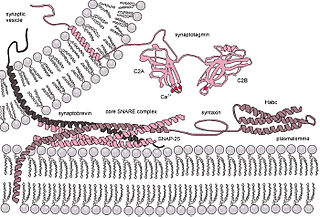
SNARE proteins – "SNAPREceptors" – are a large protein family consisting of at least 24 members in yeasts and more than 60 members in mammalian and plant cells. The primary role of SNARE proteins is to mediate the fusion of vesicles with the target membrane; this notably mediates exocytosis, but can also mediate the fusion of vesicles with membrane-bound compartments. The best studied SNAREs are those that mediate the release of synaptic vesicles containing neurotransmitters in neurons. These neuronal SNAREs are the targets of the neurotoxins responsible for botulism and tetanus produced by certain bacteria.

Synaptosomal-Associated Protein, 25kDa (SNAP-25) is a Target Soluble NSF (N-ethylmaleimide-sensitive factor) Attachment Protein Receptor (t-SNARE) protein encoded by the SNAP25 gene found on chromosome 20p12.2 in humans. SNAP-25 is a component of the trans-SNARE complex, which accounts for membrane fusion specificity and directly executes fusion by forming a tight complex that brings the synaptic vesicle and plasma membranes together.

Synaptotagmins (SYTs) constitute a family of membrane-trafficking proteins that are characterized by an N-terminal transmembrane region (TMR), a variable linker, and two C-terminal C2 domains - C2A and C2B. There are 17 isoforms in the mammalian synaptotagmin family. There are several C2-domain containing protein families that are related to synaptotagmins, including transmembrane (Ferlins, Extended-Synaptotagmin (E-Syt) membrane proteins, and MCTPs) and soluble (RIMS1 and RIMS2, UNC13D, synaptotagmin-related proteins and B/K) proteins. The family includes synaptotagmin 1, a Ca2+ sensor in the membrane of the pre-synaptic axon terminal, coded by gene SYT1.

Emodepside is an anthelmintic drug that is effective against a number of gastrointestinal nematodes, is licensed for use in cats and belongs to the class of drugs known as the octadepsipeptides, a relatively new class of anthelmintic, which are suspected to achieve their anti-parasitic effect by a novel mechanism of action due to their ability to kill nematodes resistant to other anthelmintics.
Complexin (also known as synaphin) refers to a one of a small set of eukaryotic cytoplasmic neuronal proteins which binds to the SNARE protein complex (SNAREpin) with a high affinity. These are called synaphin 1 and 2. In the presence of Ca2+, the transport vesicle protein synaptotagmin displaces complexin, allowing the SNARE protein complex to bind the transport vesicle to the presynaptic membrane.

Syntaxin-1A is a protein that in humans is encoded by the STX1A gene.

Synaptosomal-associated protein 23 is a protein that in humans is encoded by the SNAP23 gene. Two alternative transcript variants encoding different protein isoforms have been described for this gene.

Synaptotagmin-1 is a protein that in humans is encoded by the SYT1 gene.
Vesicle-associated membrane protein 2 (VAMP2) is a protein that in humans is encoded by the VAMP2 gene.

Syntaxin-binding protein 1 is a protein that in humans is encoded by the STXBP1 gene. This gene encodes a syntaxin-binding protein. The encoded protein appears to play a role in release of neurotransmitters via regulation of syntaxin, a transmembrane attachment protein receptor. Mutations in this gene have been associated with neurological disorders including epilepsy, intellectual disability, and movement disorders.

Syntaxin-6 is a protein that in humans is encoded by the STX6 gene.

Syntaxin-binding protein 5 is a protein that in humans is encoded by the STXBP5 gene. It is also known as tomosyn, after 友, "friend" in Japanese, for its role as a binding protein.

Axon terminals are distal terminations of the branches of an axon. An axon, also called a nerve fiber, is a long, slender projection of a nerve cell that conducts electrical impulses called action potentials away from the neuron's cell body to transmit those impulses to other neurons, muscle cells, or glands. Most presynaptic terminals in the central nervous system are formed along the axons, not at their ends.
The ribbon synapse is a type of neuronal synapse characterized by the presence of an electron-dense structure, the synaptic ribbon, that holds vesicles close to the active zone. It is characterized by a tight vesicle-calcium channel coupling that promotes rapid neurotransmitter release and sustained signal transmission. Ribbon synapses undergo a cycle of exocytosis and endocytosis in response to graded changes of membrane potential. It has been proposed that most ribbon synapses undergo a special type of exocytosis based on coordinated multivesicular release. This interpretation has recently been questioned at the inner hair cell ribbon synapse, where it has been instead proposed that exocytosis is described by uniquantal release shaped by a flickering vesicle fusion pore.
Vesicle fusion is the merging of a vesicle with other vesicles or a part of a cell membrane. In the latter case, it is the end stage of secretion from secretory vesicles, where their contents are expelled from the cell through exocytosis. Vesicles can also fuse with other target cell compartments, such as a lysosome. Exocytosis occurs when secretory vesicles transiently dock and fuse at the base of cup-shaped structures at the cell plasma membrane called porosome, the universal secretory machinery in cells. Vesicle fusion may depend on SNARE proteins in the presence of increased intracellular calcium (Ca2+) concentration.
Munc-18 proteins are the mammalian homologue of UNC-18 and are a member of the Sec1/Munc18-like (SM) protein family. Munc-18 proteins have been identified as essential components of the synaptic vesicle fusion protein complex and are crucial for the regulated exocytosis of neurons and neuroendocrine cells.
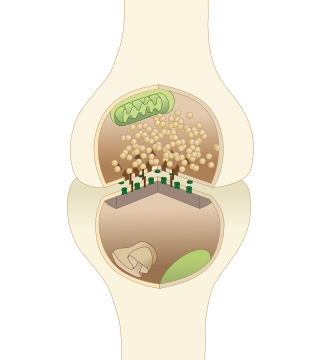
The active zone or synaptic active zone is a term first used by Couteaux and Pecot-Dechavassinein in 1970 to define the site of neurotransmitter release. Two neurons make near contact through structures called synapses allowing them to communicate with each other. As shown in the adjacent diagram, a synapse consists of the presynaptic bouton of one neuron which stores vesicles containing neurotransmitter, and a second, postsynaptic neuron which bears receptors for the neurotransmitter, together with a gap between the two called the synaptic cleft. When an action potential reaches the presynaptic bouton, the contents of the vesicles are released into the synaptic cleft and the released neurotransmitter travels across the cleft to the postsynaptic neuron and activates the receptors on the postsynaptic membrane.

Soluble N-ethylmaleimide-Sensitive Factor Attachment Proteins are a family of cytosolic adaptor proteins involved in vesicular fusion at membranes during intracellular transport and exocytosis. SNAPs interact with proteins of the SNARE complex and NSF to play a key role in recycling the components of the fusion complex. SNAPs are involved in the priming of the vesicle fusion complex during assembly, as well as in the disassembly following a vesicle fusion event. Following membrane fusion, the tethering SNARE proteins complex disassembles in response to steric changes originating from the ATPase NSF. The energy provided by NSF is transferred throughout the SNARE complex and SNAP, allowing the proteins to untangle, and recycled for future fusion events. Mammals have three SNAP genes: α-SNAP, β-SNAP, and γ-SNAP. α- and γ-SNAP are expressed throughout the body, while β-SNAP is specific to the brain. The yeast homolog of the human SNAP is Sec17, the structural diagram of which is included on this page.















