Sarcolipin is a micropeptide protein that in humans is encoded by the SLN gene. [3] [4]
Sarcolipin is a micropeptide protein that in humans is encoded by the SLN gene. [3] [4]
Sarcoplasmic reticulum Ca2+-ATPases are transmembrane proteins that catalyze the ATP-dependent transport of Ca2+ from the cytosol into the lumen of the sarcoplasmic reticulum in muscle cells. The SLN gene encodes a small transmembrane proteolipid that regulates several sarcoplasmic reticulum Ca2+-ATPases by reducing the accumulation of Ca2+ in the sarcoplasmic reticulum without affecting the rate of ATP hydrolysis. [4]
Ablation of sarcolipin increases atrial Ca2+ transient amplitudes and enhanced atrial contractility. Furthermore, atria from sarcolipin-null mice have blunted response to isoproterenol stimulation, implicating sarcolipin as a mediator of beta-adrenergic responses in atria. [5]
Sarcolipin is an important mediator of muscle based non shivering thermogenesis (NST). It causes the sarcoplasmic reticulum Ca2+-ATPases to stop pumping Ca2+ ions but continue futilely hydrolysing ATP, thus releasing the energy as heat. [6] [7] Sarcolipin mediated heat production is very important for many organisms to maintain a warm body. In mammals thermogenesis by skeletal muscles is complemented by thermogenesis in the brown adipose tissue and beige adipose tissue. [8] Sarcolipin mediated heat production in contractile muscles helps endothermic fish like the opah heat its body. Some fishes like the billfishes have a specialised brain heater tissue that is derived from muscles that cannot contract but specialise in producing heat using sarcolipin.
SLN (gene) has been shown to interact with PLN [9] [10] and ATP2A1. [9] [10]
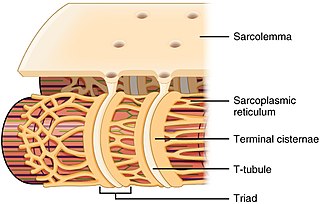
The sarcoplasmic reticulum (SR) is a membrane-bound structure found within muscle cells that is similar to the smooth endoplasmic reticulum in other cells. The main function of the SR is to store calcium ions (Ca2+). Calcium ion levels are kept relatively constant, with the concentration of calcium ions within a cell being 10,000 times smaller than the concentration of calcium ions outside the cell. This means that small increases in calcium ions within the cell are easily detected and can bring about important cellular changes (the calcium is said to be a second messenger). Calcium is used to make calcium carbonate (found in chalk) and calcium phosphate, two compounds that the body uses to make teeth and bones. This means that too much calcium within the cells can lead to hardening (calcification) of certain intracellular structures, including the mitochondria, leading to cell death. Therefore, it is vital that calcium ion levels are controlled tightly, and can be released into the cell when necessary and then removed from the cell.
SERCA, or sarco/endoplasmic reticulum Ca2+-ATPase, or SR Ca2+-ATPase, is a calcium ATPase-type P-ATPase. Its major function is to transport calcium from the cytosol into the sarcoplasmic reticulum.

Muscle contraction is the activation of tension-generating sites within muscle cells. In physiology, muscle contraction does not necessarily mean muscle shortening because muscle tension can be produced without changes in muscle length, such as when holding something heavy in the same position. The termination of muscle contraction is followed by muscle relaxation, which is a return of the muscle fibers to their low tension-generating state.

Phospholamban, also known as PLN or PLB, is a micropeptide protein that in humans is encoded by the PLN gene. Phospholamban is a 52-amino acid integral membrane protein that regulates the calcium (Ca2+) pump in cardiac muscle cells.

Ca2+ ATPase is a form of P-ATPase that transfers calcium after a muscle has contracted. The two kinds of calcium ATPase are:
The plasma membrane Ca2+ ATPase (PMCA) is a transport protein in the plasma membrane of cells and functions to remove calcium (Ca2+) from the cell. PMCA function is vital for regulating the amount of Ca2+ within all eukaryotic cells. There is a very large transmembrane electrochemical gradient of Ca2+ driving the entry of the ion into cells, yet it is very important that they maintain low concentrations of Ca2+ for proper cell signalling. Thus, it is necessary for cells to employ ion pumps to remove the Ca2+. The PMCA and the sodium calcium exchanger (NCX) are together the main regulators of intracellular Ca2+ concentrations. Since it transports Ca2+ into the extracellular space, the PMCA is also an important regulator of the calcium concentration in the extracellular space.
Sarcalumenin is a protein that in humans is encoded by the SRL gene.
The Bowditch effect, also known as the Treppe phenomenon and the Treppe effect, is an autoregulation method by which myocardial tension increases with an increase in heart rate. It was first observed by Henry Pickering Bowditch in 1871.
The Anrep effect is an autoregulation method in which myocardial contractility increases with afterload. It was experimentally determined that increasing afterload caused a proportional linear increase in ventricular inotropy. This effect is found in denervated heart preparations, such as the Starling Preparation, and represents an intrinsic autoregulation mechanism.
ATP2A2 also known as sarcoplasmic/endoplasmic reticulum calcium ATPase 2 (SERCA2) is an ATPase associated with Darier's disease and Acrokeratosis verruciformis.
Lusitropy is the rate of myocardial relaxation. The increase in cytosolic calcium of cardiomyocytes via increased uptake leads to increased myocardial contractility, but the myocardial relaxation, or lusitropy, decreases. This should not be confused, however, with catecholamine-induced calcium uptake into the sarcoplasmic reticulum, which increases lusitropy.

Ryanodine receptor 2 (RYR2) is one of a class of ryanodine receptors and a protein found primarily in cardiac muscle. In humans, it is encoded by the RYR2 gene. In the process of cardiac calcium-induced calcium release, RYR2 is the major mediator for sarcoplasmic release of stored calcium ions.
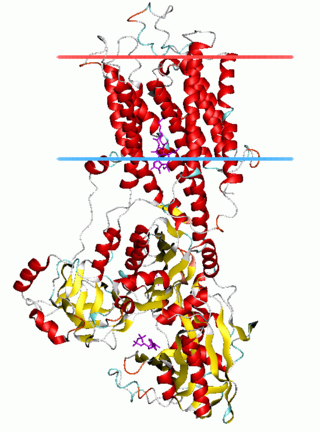
The P-type ATPases, also known as E1-E2 ATPases, are a large group of evolutionarily related ion and lipid pumps that are found in bacteria, archaea, and eukaryotes. P-type ATPases are α-helical bundle primary transporters named based upon their ability to catalyze auto- (or self-) phosphorylation (hence P) of a key conserved aspartate residue within the pump and their energy source, adenosine triphosphate (ATP). In addition, they all appear to interconvert between at least two different conformations, denoted by E1 and E2. P-type ATPases fall under the P-type ATPase (P-ATPase) Superfamily (TC# 3.A.3) which, as of early 2016, includes 20 different protein families.

Sarcoplasmic/endoplasmic reticulum calcium ATPase 1 (SERCA1) is an enzyme that in humans is encoded by the ATP2A1 gene.
Sarcoplasmic/endoplasmic reticulum calcium ATPase 3 is an enzyme that in humans is encoded by the ATP2A3 gene.

Sarcoplasmic reticulum histidine-rich calcium-binding protein is a protein that in humans is encoded by the HRC gene.
Calcium pumps are a family of ion transporters found in the cell membrane of all animal cells. They are responsible for the active transport of calcium out of the cell for the maintenance of the steep Ca2+ electrochemical gradient across the cell membrane. Calcium pumps play a crucial role in proper cell signalling by keeping the intracellular calcium concentration roughly 10,000 times lower than the extracellular concentration. Failure to do so is one cause of muscle cramps.

Brody myopathy, also called Brody disease, is a rare disorder that affects skeletal muscle function. BD was first characterized in 1969 by Dr. Irwin A. Brody at Duke University Medical Center. Individuals with BD have difficulty relaxing their muscles after exercise. This difficulty in relaxation leads to symptoms including cramps, stiffness, and discomfort in the muscles of the limbs and face. Symptoms are heightened by exercise and commonly progress in severity throughout adulthood.
CXL 1020 is an experimental drug that is being investigated as a treatment for acute decompensated heart failure. CXL 1020 functions as a nitroxyl donor; nitroxyl is the reduced, protonated version of nitric oxide. Nitroxyl is capable of enhancing left ventricular contractility without increasing heart rate by modifying normal Ca2+ cycling through the sarcoplasmic reticulum as well as increasing the sensitivity of cardiac myofilaments to Ca2+.

Istaroxime is an investigational drug under development for treatment of acute decompensated heart failure