
An HIV vaccine is a potential vaccine that could be either a preventive vaccine or a therapeutic vaccine, which means it would either protect individuals from being infected with HIV or treat HIV-infected individuals.
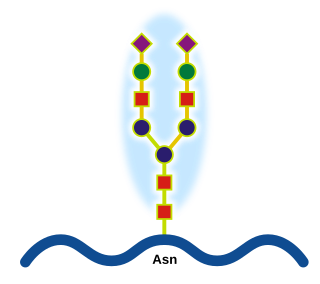
Glycoproteins are proteins which contain oligosaccharide chains covalently attached to amino acid side-chains. The carbohydrate is attached to the protein in a cotranslational or posttranslational modification. This process is known as glycosylation. Secreted extracellular proteins are often glycosylated.
Enfuvirtide (INN), sold under the brand name Fuzeon, is an HIV fusion inhibitor, the first of a class of antiretroviral drugs used in combination therapy for the treatment of AIDS/HIV.

Dengue virus (DENV) is the cause of dengue fever. It is a mosquito-borne, single positive-stranded RNA virus of the family Flaviviridae; genus Flavivirus. Four serotypes of the virus have been found, and a reported fifth has yet to be confirmed, all of which can cause the full spectrum of disease. Nevertheless, scientists' understanding of dengue virus may be simplistic as, rather than distinct antigenic groups, a continuum appears to exist. This same study identified 47 strains of dengue virus. Additionally, coinfection with and lack of rapid tests for Zika virus and chikungunya complicate matters in real-world infections.

A viral protein is both a component and a product of a virus. Viral proteins are grouped according to their functions, and groups of viral proteins include structural proteins, nonstructural proteins, regulatory proteins, and accessory proteins. Viruses are non-living and do not have the means to reproduce on their own, instead depending on their host cell's resources in order to reproduce. Thus, viruses do not code for many of their own viral proteins, and instead use the host cell's machinery to produce the viral proteins they require for replication.

In molecular biology, CD4 is a glycoprotein that serves as a co-receptor for the T-cell receptor (TCR). CD4 is found on the surface of immune cells such as T helper cells, monocytes, macrophages, and dendritic cells. It was discovered in the late 1970s and was originally known as leu-3 and T4 before being named CD4 in 1984. In humans, the CD4 protein is encoded by the CD4 gene.
The genome and proteins of HIV (human immunodeficiency virus) have been the subject of extensive research since the discovery of the virus in 1983. "In the search for the causative agent, it was initially believed that the virus was a form of the Human T-cell leukemia virus (HTLV), which was known at the time to affect the human immune system and cause certain leukemias. However, researchers at the Pasteur Institute in Paris isolated a previously unknown and genetically distinct retrovirus in patients with AIDS which was later named HIV." Each virion comprises a viral envelope and associated matrix enclosing a capsid, which itself encloses two copies of the single-stranded RNA genome and several enzymes. The discovery of the virus itself occurred two years following the report of the first major cases of AIDS-associated illnesses.
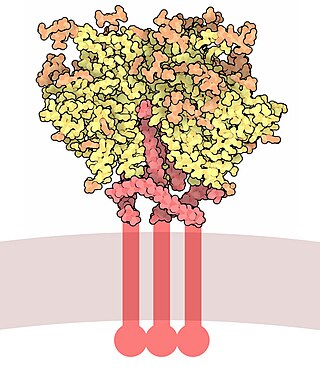
Envelope glycoprotein GP120 is a glycoprotein exposed on the surface of the HIV envelope. It was discovered by Professors Tun-Hou Lee and Myron "Max" Essex of the Harvard School of Public Health in 1988. The 120 in its name comes from its molecular weight of 120 kDa. Gp120 is essential for virus entry into cells as it plays a vital role in attachment to specific cell surface receptors. These receptors are DC-SIGN, Heparan Sulfate Proteoglycan and a specific interaction with the CD4 receptor, particularly on helper T-cells. Binding to CD4 induces the start of a cascade of conformational changes in gp120 and gp41 that lead to the fusion of the viral membrane with the host cell membrane. Binding to CD4 is mainly electrostatic although there are van der Waals interactions and hydrogen bonds.

HLA class II histocompatibility antigen, DR alpha chain is a protein that in humans is encoded by the HLA-DRA gene. HLA-DRA encodes the alpha subunit of HLA-DR. Unlike the alpha chains of other Human MHC class II molecules, the alpha subunit is practically invariable. However it can pair with, in any individual, the beta chain from 3 different DR beta loci, DRB1, and two of any DRB3, DRB4, or DRB5 alleles. Thus there is the potential that any given individual can form 4 different HLA-DR isoforms.
Entry inhibitors, also known as fusion inhibitors, are a class of antiviral drugs that prevent a virus from entering a cell, for example, by blocking a receptor. Entry inhibitors are used to treat conditions such as HIV and hepatitis D.
Env is a viral gene that encodes the protein forming the viral envelope. The expression of the env gene enables retroviruses to target and attach to specific cell types, and to infiltrate the target cell membrane.
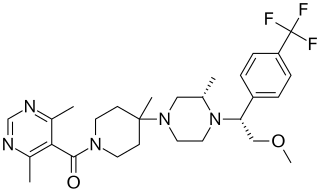
Vicriviroc, previously named SCH 417690 and SCH-D, is a pyrimidine CCR5 entry inhibitor of HIV-1. It was developed by the pharmaceutical company Schering-Plough. Merck decided to not pursue regulatory approval for use in treatment-experienced patients because the drug did not meet primary efficacy endpoints in late stage trials. Clinical trials continue in patients previously untreated for HIV.
A fusion mechanism is any mechanism by which cell fusion or virus–cell fusion takes place, as well as the machinery that facilitates these processes. Cell fusion is the formation of a hybrid cell from two separate cells. There are three major actions taken in both virus–cell fusion and cell–cell fusion: the dehydration of polar head groups, the promotion of a hemifusion stalk, and the opening and expansion of pores between fusing cells. Virus–cell fusions occur during infections of several viruses that are health concerns relevant today. Some of these include HIV, Ebola, and influenza. For example, HIV infects by fusing with the membranes of immune system cells. In order for HIV to fuse with a cell, it must be able to bind to the receptors CD4, CCR5, and CXCR4. Cell fusion also occurs in a multitude of mammalian cells including gametes and myoblasts.
T-cell surface glycoprotein CD3 gamma chain is a protein that in humans is encoded by the CD3G gene.

HLA class II histocompatibility antigen, DM beta chain is a protein that in humans is encoded by the HLA-DMB gene.

HLA class II histocompatibility antigen, DM alpha chain is a protein that in humans is encoded by the HLA-DMA gene.

HLA class II histocompatibility antigen, DO beta chain is a protein that in humans is encoded by the HLA-DOB gene.
2F5 is a broadly neutralizing human monoclonal antibody (mAb) that has been shown to bind to and neutralize HIV-1 in vitro, making it a potential candidate for use in vaccine synthesis. 2F5 recognizes an epitope in the membrane-proximal external region (MPER) of HIV-1 gp41. 2F5 then binds to this epitope and its constant region interacts with the viral lipid membrane, which neutralizes the virus.
CCR5 receptor antagonists are a class of small molecules that antagonize the CCR5 receptor. The C-C motif chemokine receptor CCR5 is involved in the process by which HIV, the virus that causes AIDS, enters cells. Hence antagonists of this receptor are entry inhibitors and have potential therapeutic applications in the treatment of HIV infections.

VIR-576 is an experimental drug that is under clinical trials for the treatment of HIV-1 infections. VIR-576 is synthetic peptide that binds to HIV-1's hydrophobic fusion peptide gp41, preventing the virus from inserting itself into a host cell's membrane to initiate an infection. This drug is a synthesized variant of a highly specific natural entry inhibitor designated as VIRIP.













