
gamma-Aminobutyric acid, or γ-aminobutyric acid, or GABA, is the chief inhibitory neurotransmitter in the developmentally mature mammalian central nervous system. Its principal role is reducing neuronal excitability throughout the nervous system. GABA is sold as a dietary supplement.

Phenelzine is a non-selective and irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class which is used as an antidepressant and anxiolytic. Along with tranylcypromine and isocarboxazid, phenelzine is one of the few non-selective and irreversible MAOIs still in widespread clinical use. It is available in 15 mg tablets and doses usually range from 45–90 mg per day.

The GABA receptors are a class of receptors that respond to the neurotransmitter gamma-aminobutyric acid (GABA), the chief inhibitory compound in the mature vertebrate central nervous system. There are two classes of GABA receptors: GABAA and GABAB. GABAA receptors are ligand-gated ion channels ; whereas GABAB receptors are G protein-coupled receptors, also called metabotropic receptors.
GABAB receptors (GABABR) are metabotropic transmembrane receptors for gamma-aminobutyric acid (GABA) that are linked via G-proteins to potassium channels. The changing potassium concentrations hyperpolarize the cell at the end of an action potential. The reversal potential of the GABAB-mediated IPSP is –100 mV, which is much more hyperpolarized than the GABAA IPSP. GABAB receptors are found in the central nervous system and the autonomic division of the peripheral nervous system.

Progabide is an analogue and prodrug of γ-aminobutyric acid (GABA) used in the treatment of epilepsy. Via conversion into GABA, progabide behaves as an agonist of the GABAA, GABAB, and GABAA-ρ receptors.
In biochemistry and pharmacology, GABA transaminase enzymes comprise a family of transaminases which degrade the neurotransmitter GABA. GABA transaminases include 4-aminobutyrate transaminase, which catalyzes the chemical reaction:

γ-Amino-β-hydroxybutyric acid (GABOB), also known as β-hydroxy-γ-aminobutyric acid (β-hydroxy-GABA), and sold under the brand name Gamibetal among others, is an anticonvulsant which is used for the treatment of epilepsy in Europe, Japan, and Mexico. It is a GABA analogue, or an analogue of the neurotransmitter γ-aminobutyric acid (GABA), and has been found to be an endogenous metabolite of GABA.

Gamma-aminobutyric acid (GABA) B receptor, 1 (GABAB1), is a G-protein coupled receptor subunit encoded by the GABBR1 gene.
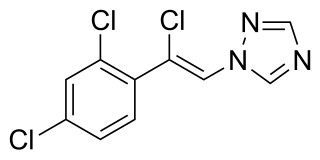
Loreclezole is a sedative and an anticonvulsant which acts as a GABAA receptor positive allosteric modulator. The binding site of loreclezole has been shown experimentally to be shared by valerenic acid, an extract of the root of the valerian plant. Structurally, loreclezole is a triazole derivative. In animal seizure models, loreclezole is protective against pentylenetetrazol seizures but is less active in the maximal electroshock test. In addition, at low, nontoxic doses, the drug has anti-absence activity in a genetic model of generalized absence epilepsy. Consequently, loreclezole has a profile of activity similar to that of benzodiazepines. A potential benzodiazepine-like interaction with GABA receptors is suggested by the observation that the anticonvulsant effects of loreclezole can be reversed by benzodiazepine receptor inverse agonists. The benzodiazepine antagonist flumazenil, however, fails to alter the anticonvulsant activity of loreclezole, indicating that loreclezole is not a benzodiazepine receptor agonist. Using native rat and cloned human GABA-A receptors, loreclezole strongly potentiated GABA-activated chloride current. However, activity of the drug did not require the presence of the γ-subunit and was not blocked by flumazenil, confirming that loreclezole does not interact with the benzodiazepine recognition site.
In enzymology, 4-aminobutyrate transaminase, also called GABA transaminase or 4-aminobutyrate aminotransferase, is an enzyme that catalyzes the chemical reaction:

GABA transporter 1 (GAT1) also known as sodium- and chloride-dependent GABA transporter 1 is a protein that in humans is encoded by the SLC6A1 gene.

TPA-023 (MK-0777) is an anxiolytic drug with a novel chemical structure, which is used in scientific research. It has similar effects to benzodiazepine drugs, but is structurally distinct and so is classed as a nonbenzodiazepine anxiolytic. It is a subtype-selective, mixed agonist-antagonist at GABAA receptors, which acts as a partial agonist at the α2 and α3 subtypes, but as a silent antagonist at α1 and α5 subtypes. It has primarily anxiolytic and anticonvulsant effects in animal tests, but with no sedative effects even at 50 times the effective anxiolytic dose.

Gabaculine is a naturally occurring neurotoxin first isolated from the bacteria Streptomyces toyacaensis, which acts as a potent and irreversible GABA transaminase inhibitor, and also a GABA reuptake inhibitor. Gabaculine is also known as 3-amino-2,3-dihydrobenzoic acid hydrochloride and 5-amino cyclohexa-1,3 dienyl carboxylic acid. Gabaculine increased GABA levels in the brain and had an effect on convulsivity in mice.
GABA transporters (Gamma-Aminobutyric acid transporters) belong to the family of neurotransmitters known as sodium symporters, also known as solute carrier 6 (SLC6). These are large family of neurotransmitter which are Na+ concentration dependent. They are found in various regions of the brain in different cell types, such as neurons and astrocytes.
In pharmacology, a GABA transaminase inhibitor is an enzyme inhibitor that acts upon GABA transaminase. Inhibition of GABA transaminase enzymes reduces the degradation of GABA, leading to increased neuronal GABA concentrations.

Pivagabine, also known as N-pivaloyl-γ-aminobutyric acid or N-pivaloyl-GABA, is an antidepressant and anxiolytic drug which was introduced in Italy in 1997 for the treatment of depressive and maladaptive syndromes. But it was discontinued in Italy. Originally believed to function as a prodrug to GABA, pivagabine is now believed to act somehow via modulation of corticotropin-releasing factor (CRF).

Aminooxyacetic acid, often abbreviated AOA or AOAA, is a compound that inhibits 4-aminobutyrate aminotransferase (GABA-T) activity in vitro and in vivo, leading to less gamma-aminobutyric acid (GABA) being broken down. Subsequently, the level of GABA is increased in tissues. At concentrations high enough to fully inhibit 4-aminobutyrate aminotransferase activity, aminooxyacetic acid is indicated as a useful tool to study regional GABA turnover in rats.
4-aminobutyrate---pyruvate transaminase is an enzyme with systematic name 4-aminobutanoate:pyruvate aminotransferase. This enzyme is a type of GABA transaminase, which degrades the neurotransmitter GABA. The enzyme catalyses the following chemical reaction

CI-966 (developmental code name) is a central nervous system depressant acting as a GABA reuptake inhibitor, specifically a highly potent and selective blocker of the GABA transporter 1 (GAT-1) (IC50 = 0.26 μM), and hence indirect and non-selective GABA receptor full agonist. It was investigated as a potential anticonvulsant, anxiolytic, and neuroprotective therapeutic but was discontinued during clinical development due to the incidence of severe adverse effects at higher doses and hence was never marketed.

In pharmacology, GABAA receptor positive allosteric modulators are positive allosteric modulator (PAM) molecules that increase the activity of the GABAA receptor protein in the vertebrate central nervous system.















