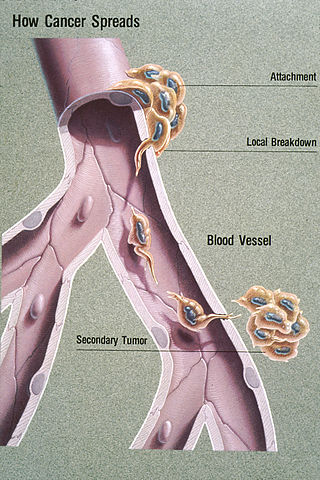
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then, are metastases (mets). It is generally distinguished from cancer invasion, which is the direct extension and penetration by cancer cells into neighboring tissues.

A biopsy is a medical test commonly performed by a surgeon, an interventional radiologist, or an interventional cardiologist. The process involves the extraction of sample cells or tissues for examination to determine the presence or extent of a disease. The tissue is then fixed, dehydrated, embedded, sectioned, stained and mounted before it is generally examined under a microscope by a pathologist; it may also be analyzed chemically. When an entire lump or suspicious area is removed, the procedure is called an excisional biopsy. An incisional biopsy or core biopsy samples a portion of the abnormal tissue without attempting to remove the entire lesion or tumor. When a sample of tissue or fluid is removed with a needle in such a way that cells are removed without preserving the histological architecture of the tissue cells, the procedure is called a needle aspiration biopsy. Biopsies are most commonly performed for insight into possible cancerous or inflammatory conditions.
Keratin 20, often abbreviated CK20, is a protein that in humans is encoded by the KRT20 gene.

Keratin, type I cytoskeletal 19 (Keratin-19)) also known as cytokeratin-19 (CK-19) is a 40 kDa protein that in humans is encoded by the KRT19 gene. Keratin-19 is a type I keratin.

Hybridoma technology is a method for producing large numbers of identical antibodies, also called monoclonal antibodies. This process starts by injecting a mouse with an antigen that provokes an immune response. A type of white blood cell, the B cell, produces antibodies that bind to the injected antigen. These antibody producing B-cells are then harvested from the mouse and, in turn, fused with immortal myeloma cancer cells, to produce a hybrid cell line called a hybridoma, which has both the antibody-producing ability of the B-cell and the longevity and reproductivity of the myeloma.
The epithelial–mesenchymal transition (EMT) is a process by which epithelial cells lose their cell polarity and cell–cell adhesion, and gain migratory and invasive properties to become mesenchymal stem cells; these are multipotent stromal cells that can differentiate into a variety of cell types. EMT is essential for numerous developmental processes including mesoderm formation and neural tube formation. EMT has also been shown to occur in wound healing, in organ fibrosis and in the initiation of metastasis in cancer progression.
Adecatumumab (MT201) is a recombinant human IgG1 monoclonal antibody which is used to target tumor cells. It binds to the epithelial cell adhesion molecule, with the intent to trigger antibody-dependent cellular cytotoxicity. It was developed by Micromet Inc, which was acquired by Amgen.

Keratin, type II cytoskeletal 8 also known as cytokeratin-8 (CK-8) or keratin-8 (K8) is a keratin protein that is encoded in humans by the KRT8 gene. It is often paired with keratin 18.
Triple-negative breast cancer (TNBC) is any breast cancer that either lacks or shows low levels of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2) overexpression and/or gene amplification. Triple-negative is sometimes used as a surrogate term for basal-like.

Bone metastasis, or osseous metastatic disease, is a category of cancer metastases that result from primary tumor invasions into bones. Bone-originating primary tumors such as osteosarcoma, chondrosarcoma, and Ewing sarcoma are rare; the most common bone tumor is a metastasis. Bone metastases can be classified as osteolytic, osteoblastic, or both. Unlike hematologic malignancies which originate in the blood and form non-solid tumors, bone metastases generally arise from epithelial tumors and form a solid mass inside the bone. Primary breast cancer patients are particularly vulnerable to develop bone metastases. Bone metastases, especially in a state of advanced disease, can cause severe pain, characterized by a dull, constant ache with periodic spikes of incident pain.

Metastatic breast cancer, also referred to as metastases, advanced breast cancer, secondary tumors, secondaries or stage IV breast cancer, is a stage of breast cancer where the breast cancer cells have spread to distant sites beyond the axillary lymph nodes. There is no cure for metastatic breast cancer; there is no stage after IV.
Porocarcinoma (PCA) is a rare form of skin cancer that develops in eccrine sweat glands, i.e. the body's widely distributed major type of sweat glands, as opposed to the apocrine sweat glands which are located primarily in the armpits and perineal area. This cancer typically develops in individuals as a single cutaneous tumor in the intraepidermal spiral part of these sweat glands' ducts at or near to where they open on the skin's surface. PCA tumors are classified as one form of the cutaneous adnexal tumors; in a study of 2,205 cases, PCA was the most common (11.8%) form of these tumors.
M30 Apoptosense® ELISA is an enzyme-linked immunosorbent assay developed for the detection of soluble caspase-cleaved keratin 18.

A cancer biomarker refers to a substance or process that is indicative of the presence of cancer in the body. A biomarker may be a molecule secreted by a tumor or a specific response of the body to the presence of cancer. Genetic, epigenetic, proteomic, glycomic, and imaging biomarkers can be used for cancer diagnosis, prognosis, and epidemiology. Ideally, such biomarkers can be assayed in non-invasively collected biofluids like blood or serum.
Migration inducting gene 7 is a gene that corresponds to a cysteine-rich protein localized to the cell membrane and cytoplasm. It is the first-in-class of novel proteins translated from what are thought to be long Non-coding RNAs.
A liquid biopsy, also known as fluid biopsy or fluid phase biopsy, is the sampling and analysis of non-solid biological tissue, primarily blood. Like traditional biopsy, this type of technique is mainly used as a diagnostic and monitoring tool for diseases such as cancer, with the added benefit of being largely non-invasive. Liquid biopsies may also be used to validate the efficiency of a cancer treatment drug by taking multiple samples in the span of a few weeks. The technology may also prove beneficial for patients after treatment to monitor relapse.
Breast cancer metastatic mouse models are experimental approaches in which mice are genetically manipulated to develop a mammary tumor leading to distant focal lesions of mammary epithelium created by metastasis. Mammary cancers in mice can be caused by genetic mutations that have been identified in human cancer. This means models can be generated based upon molecular lesions consistent with the human disease.
In cellular biology, angiopellosis is the movement of cells out of the circulatory system, into the surrounding tissue. This process is specific to non-leukocytic cells; white blood cells (leukocytes) employ diapedesis for movement out of circulation. Angiopellosis was discovered by studying the way that stem cells reach damaged tissue when injected or infused into the circulatory system. It has been found that circulating tumor cells (CTCs) possess this ability to exit blood vessels through angiopellosis during the process of metastasis.

Richard James Cote is a pathologist, academic and author. He is the Edward Mallinckrodt Professor and Chair of the Department of Pathology and Immunology at Washington University in St. Louis School of Medicine, and the Pathologist-in-Chief at Barnes Jewish Hospital.
The cancer exodus hypothesis establishes that circulating tumor cell clusters maintain their multicellular structure throughout the metastatic process. It was previously thought that these clusters must dissociate into single cells during metastasis. According to the hypothesis, CTC clusters intravasate, travel through circulation as a cohesive unit, and extravasate at distant sites without disaggregating, significantly enhancing their metastatic potential. This concept is considered a key advancement in understanding of cancer biology and CTCs role in cancer metastasis.












