
Electroconvulsive therapy (ECT) is a psychiatric treatment where a generalized seizure is electrically induced to manage refractory mental disorders. Typically, 70 to 120 volts are applied externally to the patient's head, resulting in approximately 800 milliamperes of direct current passing between the electrodes, for a duration of 100 milliseconds to 6 seconds, either from temple to temple or from front to back of one side of the head. However, only about 1% of the electrical current crosses the bony skull into the brain because skull impedance is about 100 times higher than skin impedance.
An orphan drug is a pharmaceutical agent that is developed to treat certain rare medical conditions. An orphan drug would not be profitable to produce without government assistance, due to the small population of patients affected by the conditions. The conditions that orphan drugs are used to treat are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy that depends on the legislation of the country.

Nootropics are numerous natural, semi-synthetic and synthetic molecules that claim to improve cognitive functions.

Piracetam is a drug marketed as a treatment for myoclonus. It is also used as a cognitive enhancer to improve memory, attention, and learning. Evidence to support its use is unclear, with some studies showing modest benefits in specific populations and others showing minimal or no benefit. Piracetam is sold as a medication in many European countries. Sale of piracetam is not illegal in the United States, although it is not regulated nor approved by the FDA so it is legally sold for research use only.
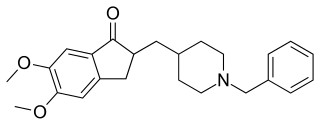
Donepezil, sold under the brand name Aricept among others, is a medication used to treat dementia of the Alzheimer's type. It appears to result in a small benefit in mental function and ability to function. Use, however, has not been shown to change the progression of the disease. Treatment should be stopped if no benefit is seen. It is taken by mouth or via a transdermal patch.

Fasoracetam is a research chemical of the racetam family. It is a putative nootropic that failed to show sufficient efficacy in clinical trials for vascular dementia. It is currently being studied for its potential use for attention deficit hyperactivity disorder.

Amifampridine is used as a drug, predominantly in the treatment of a number of rare muscle diseases. The free base form of the drug has been used to treat congenital myasthenic syndromes and Lambert–Eaton myasthenic syndrome (LEMS) through compassionate use programs since the 1990s and was recommended as a first line treatment for LEMS in 2006, using ad hoc forms of the drug, since there was no marketed form.

Afamelanotide, sold under the brand name Scenesse, is a synthetic peptide and analogue of α-melanocyte stimulating hormone. It has been used to prevent skin damage from the sun in people with erythropoietic protoporphyria in the European Union since January 2015 and the United States since October 2019. As a medication, it is administered in subcutaneous implant form. Each implant lasts two months.

Tasimelteon, sold under the brand name Hetlioz, is a medication approved by the U.S. Food and Drug Administration (FDA) in January 2014, for the treatment of non-24-hour sleep–wake disorder. In June 2014, the European Medicines Agency (EMA) accepted an EU filing application for tasimelteon and in July 2015, the drug was approved in the European Union for the treatment of non-24-hour sleep-wake rhythm disorder in totally blind adults, but not in the rarer case of non-24 in sighted people.

Pimavanserin, sold under the brand name Nuplazid, is an atypical antipsychotic which is approved for the treatment of Parkinson's disease psychosis and is also being studied for the treatment of Alzheimer’s disease psychosis, schizophrenia, agitation, and major depressive disorder. Unlike other antipsychotics, pimavanserin is not a dopamine receptor antagonist.

N-Phenylacetyl-l-prolylglycine ethyl ester is promoted as a nootropic and is a prodrug of cyclic glycine-proline. Other names include the brand name Noopept, developmental code GVS-111; proposed INN omberacetam.
Teprotumumab, sold under the brand name Tepezza, is a medication used to treat adults with thyroid eye disease, a rare condition where the muscles and fatty tissues behind the eye become inflamed, causing the eyes to bulge outwards.
Catalyst Pharmaceuticals Inc. is a biopharmaceutical company based in Coral Gables, Florida. The company is developing medicines for rare diseases, including the phosphate salt of amifampridine for the treatment of Lambert–Eaton myasthenic syndrome (LEMS). The drug is referred to under the trade name Firdapse, which was approved by the FDA for approved use in children 6 years and older with LEMS in addition to the prior approval for use in adults with LEMS on November 28, 2018. Firdapse commercially launched in January 2019.
Duvelisib, sold under the brand name Copiktra, is a medication used to treat chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), and follicular lymphoma after other treatments have failed. It is taken by mouth. It is a PI3 kinase inhibitor.

Entrectinib, sold under the brand name Rozlytrek, is an anti-cancer medication used to treat ROS1-positive non-small cell lung cancer and NTRK fusion-positive solid tumors. It is a selective tyrosine kinase inhibitor (TKI), of the tropomyosin receptor kinases (TRK) A, B and C, C-ros oncogene 1 (ROS1) and anaplastic lymphoma kinase (ALK).
Inebilizumab, sold under the brand name Uplizna, is a medication for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adults. Inebilizumab is a humanized mAb that binds to and depletes CD19+ B cells including plasmablasts and plasma cells.
Di-deuterated ethyl linoleate is an experimental, orally-bioavailable synthetic deuterated polyunsaturated fatty acid (PUFA), a part of reinforced lipids. It is an isotopologue of linoleic acid, an essential omega-6 PUFA. The deuterated compound, while identical to natural linoleic acid except for the presence of deuterium, is resistant to lipid peroxidation which makes studies of its cell-protective properties worthwhile.

Pemigatinib, sold under the brand name Pemazyre, is an anti-cancer medication used for the treatment of bile duct cancer (cholangiocarcinoma). Pemigatinib works by blocking FGFR2 in tumor cells to prevent them from growing and spreading.

Flortaucipir (18F), sold under the brand name Tauvid, is a radioactive diagnostic agent indicated for use with positron emission tomography (PET) imaging to image the brain.
Pegcetacoplan, sold under the brand name Empaveli, among others, is a medication used to treat paroxysmal nocturnal hemoglobinuria and geographic atrophy of the retina. Pegcetacoplan is a complement inhibitor.















