
Immunohistochemistry is a form of immunostaining. It involves the process of selectively identifying antigens (proteins) in cells and tissue, by exploiting the principle of antibodies binding specifically to antigens in biological tissues. Albert Hewett Coons, Ernest Berliner, Norman Jones and Hugh J Creech was the first to develop immunofluorescence in 1941. This led to the later development of immunohistochemistry.
This is a list of terms related to oncology. The original source for this list was the US National Cancer Institute's public domain Dictionary of Cancer Terms.
Affinity chromatography is a method of separating a biomolecule from a mixture, based on a highly specific macromolecular binding interaction between the biomolecule and another substance. The specific type of binding interaction depends on the biomolecule of interest; antigen and antibody, enzyme and substrate, receptor and ligand, or protein and nucleic acid binding interactions are frequently exploited for isolation of various biomolecules. Affinity chromatography is useful for its high selectivity and resolution of separation, compared to other chromatographic methods.

Carcinoembryonic antigen (CEA) describes a set of highly-related glycoproteins involved in cell adhesion. CEA is normally produced in gastrointestinal tissue during fetal development, but the production stops before birth. Consequently, CEA is usually present at very low levels in the blood of healthy adults. However, the serum levels are raised in some types of cancer, which means that it can be used as a tumor marker in clinical tests. Serum levels can also be elevated in heavy smokers.

A single-chain variable fragment (scFv) is not actually a fragment of an antibody, but instead is a fusion protein of the variable regions of the heavy (VH) and light chains (VL) of immunoglobulins, connected with a short linker peptide of ten to about 25 amino acids. The linker is usually rich in glycine for flexibility, as well as serine or threonine for solubility, and can either connect the N-terminus of the VH with the C-terminus of the VL, or vice versa. This protein retains the specificity of the original immunoglobulin, despite removal of the constant regions and the introduction of the linker. The image to the right shows how this modification usually leaves the specificity unaltered.

Cancer immunotherapy (immuno-oncotherapy) is the stimulation of the immune system to treat cancer, improving the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology (immuno-oncology) and a growing subspecialty of oncology.
An oncolytic virus is a virus that preferentially infects and kills cancer cells. As the infected cancer cells are destroyed by oncolysis, they release new infectious virus particles or virions to help destroy the remaining tumour. Oncolytic viruses are thought not only to cause direct destruction of the tumour cells, but also to stimulate host anti-tumour immune system responses. Oncolytic viruses also have the ability to affect the tumor micro-environment in multiple ways.

A single-domain antibody (sdAb), also known as a Nanobody, is an antibody fragment consisting of a single monomeric variable antibody domain. Like a whole antibody, it is able to bind selectively to a specific antigen. With a molecular weight of only 12–15 kDa, single-domain antibodies are much smaller than common antibodies which are composed of two heavy protein chains and two light chains, and even smaller than Fab fragments and single-chain variable fragments.

Integrin alpha M (ITGAM) is one protein subunit that forms heterodimeric integrin alpha-M beta-2 (αMβ2) molecule, also known as macrophage-1 antigen (Mac-1) or complement receptor 3 (CR3). ITGAM is also known as CR3A, and cluster of differentiation molecule 11B (CD11B). The second chain of αMβ2 is the common integrin β2 subunit known as CD18, and integrin αMβ2 thus belongs to the β2 subfamily integrins.

Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective, to induce a specific cell signal, to cause the immune system to attack specific cells, or to bring a drug to a specific cell type.

Abagovomab is a mouse anti-idiotype monoclonal antibody whose variable epitope mirrors a tumour antigen (CA-125) highly expressed in the epithelial ovarian cancer. Abagovomab does not bind directly to CA-125, but it works as a "surrogate" antigen, enabling the immune system to identify and attack tumour cells displaying the CA-125 protein. Through this, it is hoped that the body's immune system may be able to combat any remaining individual tumour cells and thus prevent recurrence of the disease.
A bispecific monoclonal antibody is an artificial protein that can simultaneously bind to two different types of antigen or two different epitopes on the same antigen. Naturally occurring antibodies typically only target one antigen. BsAbs can be manufactured in several structural formats. BsAbs can be designed to recruit and activate immune cells, to interfere with receptor signaling and inactivate signaling ligands, and to force association of protein complexes. BsAbs have been explored for cancer immunotherapy, drug delivery, and Alzheimer's disease.

Trophoblast glycoprotein, also known as TPBG, 5T4, Wnt-Activated Inhibitory Factor 1 or WAIF1, is a human protein encoded by a TPBG gene. TPBG is an antagonist of Wnt/β-catenin signalling pathway.
Tumor-associated glycoprotein 72 (TAG-72) is a glycoprotein found on the surface of many cancer cells, including ovary, breast, colon, lung, and pancreatic cancers. It is a mucin-like molecule with a molar mass of over 1000 kDa.

Lloyd John Old was one of the founders and standard-bearers of the field of cancer immunology. When Old began his career in 1958, tumor immunology was in its infancy. Today, cancer immunotherapies are emerging as a significant advance in cancer therapy.
Solitomab is an artificial bispecific monoclonal antibody that is being investigated as an anti-cancer drug. It is a fusion protein consisting of two single-chain variable fragments (scFvs) of different antibodies on a single peptide chain of about 55 kilodaltons. One of the scFvs binds to T cells via the CD3 receptor, and the other to EpCAM as a tumor antigen against gastrointestinal, lung, and other cancers.
Directed enzyme prodrug therapy (DEPT) uses enzymes artificially introduced into the body to convert prodrugs, which have no or poor biologically activity, to the active form in the desired location within the body. Many chemotherapy drugs for cancer lack tumour specificity and the doses required to reach therapeutic levels in the tumour are often toxic to other tissues. DEPT strategies are an experimental method of reducing the systemic toxicity of a drug, by achieving high levels of the active drug only at the desired site. This article describes the variations of DEPT technology.
Translational glycobiology or applied glycobiology is the branch of glycobiology and glycochemistry that focuses on developing new pharmaceuticals through glycomics and glycoengineering. Although research in this field presents many difficulties, translational glycobiology presents applications with therapeutic glycoconjugates, with treating various bone diseases, and developing therapeutic cancer vaccines and other targeted therapies. Some mechanisms of action include using the glycan for drug targeting, engineering protein glycosylation for better efficacy, and glycans as drugs themselves.
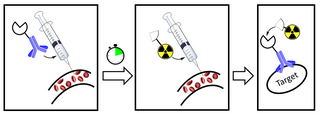
Pretargeting (imaging) is a tool for nuclear medicine and radiotherapy. Imaging studies require a high contrast of target to background. This can be provided by using a biomarker which has a high affinity and specificity for its target.
Immunoliposome therapy is a targeted drug delivery method that involves the use of liposomes coupled with monoclonal antibodies to deliver therapeutic agents to specific sites or tissues in the body. The antibody modified liposomes target tissue through cell-specific antibodies with the release of drugs contained within the assimilated liposomes. Immunoliposome aims to improve drug stability, personalize treatments, and increased drug efficacy. This form of therapy has been used to target specific cells, protecting the encapsulated drugs from degradation in order to enhance their stability, to facilitate sustained drug release and hence to advance current traditional cancer treatment.










