Metal hydrolysis and associated equilibrium constant values
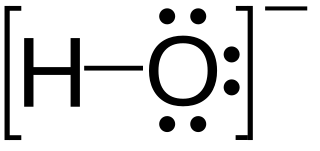
The hydrolysis reaction for a hydrated metal ion in aqueous solution can be written as:
- p Mz+ + q H2O ⇌ Mp(OH)q(pz–q) + q H+
and the corresponding formation constant as:
and associated equilibria can be written as:
- MOx(OH)z–2x(s) + z H+ ⇌ Mz+ + (z–x) H2O
- MOx(OH)z–2x(s) + x H2O ⇌ Mz+ + z OH−
- p MOx(OH)z–2x(s) + (pz–q) H+ ⇌ Mp(OH)q(pz–q) + (pz–px–q) H2O
Aluminium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [1] | Brown and Ekberg, 2016 [2] | Hummel and Thoenen, 2023 [3] |
|---|---|---|---|
| Al3+ + H2O ⇌ AlOH2+ + H+ | –4.97 | −4.98 ± 0.02 | −4.98 ± 0.02 |
| Al3+ + 2 H2O ⇌ Al(OH)2+ + 2 H+ | –9.3 | −10.63 ± 0.09 | −10.63 ± 0.09 |
| Al3+ + 3 H2O ⇌ Al(OH)3 + 3 H+ | –15.0 | −15.66 ± 0.23 | −15.99 ± 0.23 |
| Al3+ + 4 H2O ⇌ Al(OH)4– + 4 H+ | –23.0 | −22.91 ± 0.10 | −22.91 ± 0.10 |
| 2 Al3+ + 2 H2O ⇌ Al2(OH)24+ + 2 H+ | –7.7 | −7.62 ± 0.11 | −7.62 ± 0.11 |
| 3 Al3+ + 4 H2O ⇌ Al3(OH)45+ + 4 H+ | –13.94 | −14.06 ± 0.22 | −13.90 ± 0.12 |
| 13 Al3+ + 28 H2O ⇌ Al13O4(OH)247+ + 32 H+ | –98.73 | −100.03 ± 0.09 | −100.03 ± 0.09 |
| α-Al(OH)3(s) + 3 H+ ⇌ Al3+ + 3 H2O | 8.5 | 7.75 ± 0.08 | 7.75 ± 0.08 |
| γ-AlOOH(s) + 3 H+ ⇌ Al3+ + 2 H2O | 7.69 ± 0.15 | 9.4 ± 0.4 |
Americium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | NIST46 [4] | Brown and Ekberg, 2016 [5] | Grenthe et al, 2020 [6] |
|---|---|---|---|
| Am3+ + H2O ⇌ Am(OH)2+ + H+ | –6.5 ± 0.1 | –7.22 ± 0.03 | –7.2 ± 0.5 |
| Am3+ + 2 H2O ⇌ Am(OH)2+ + 2 H+ | –14.1 ± 0.3 | –14.9 ± 0.2 | –15.1 ± 0.7 |
| Am3+ + 3 H2O ⇌ Am(OH)3 + 3 H+ | –25.7 | –26.0 ± 0.2 | –26.2 ± 0.5 |
| Am3+ + 3 H2O ⇌ Am(OH)3(am) + 3 H+ | –16.9 ± 0.1 | –16.9 ± 0.8 | –16.9 ± 0.8 |
| Am3+ + 3 H2O ⇌ Am(OH)3(cr) + 3 H+ | –15.2 | –15.62 ± 0.04 | –15.6 ± 0.6 |
Americium(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [7] | Grenthe et al, 2020 [6] |
|---|---|---|
| AmO2+ + H2O ⇌ AmO2(OH) + H+ | –10.7 ± 0.2 | |
| AmO2+ + 2 H2O ⇌ AmO2(OH)2– + 2 H+ | –22.9 ± 0.7 | |
| AmO2+ + H2O ⇌ AmO2(OH)(am) + H+ | –5.4 ± 0.4 | –5.3 ± 0.5 |
Antimony(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [8] | Lothenbach et al., 1999; [9] Kitamura et al., 2010 [10] | Filella and May, 2003 [11] |
|---|---|---|---|
| Sb(OH)3 + H+ ⇌ Sb(OH)2+ + H2O | 1.41 | 1.30 | 1.371 |
| Sb(OH)3 + H2O ⇌ Sb(OH)4‒ + H+ | ‒11.82 | ‒11.93 | ‒11.70 |
| 0.5 Sb2O3(s) + 1.5 H2O ⇌ Sb(OH)3 | ‒4.24 | ||
| Sb2O3(rhombic,s) + 3 H2O ⇌ 2 Sb(OH)3 | ‒8.72 | ‒10.00 | |
| Sb2O3(cubic,s) + 3 H2O ⇌ 2 Sb(OH)3 | ‒11.40 |
Antimony(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [8] | Lothenbach et al., 1999; [9] Kitamura et al., 2010 [10] |
|---|---|---|
| Sb(OH)5 + H2O ⇌ Sb(OH)6‒ + H+ | ‒2.72 | ‒2.72 |
| 12 Sb(OH)5 + 4 H2O ⇌ Sb12(OH)644‒ + 4 H+ | 20.34 | 20.34 |
| 12 Sb(OH)5 + 5 H2O ⇌ Sb12(OH)655‒ + 5 H+ | 16.72 | 16.72 |
| 12 Sb(OH)5 + 6 H2O ⇌ Sb12(OH)666‒ + 6 H+ | 11.89 | 11.89 |
| 12 Sb(OH)5 + 7 H2O ⇌ Sb12(OH)677‒ + 7 H+ | 6.07 | 6.07 |
| 0.5 Sb2O5(s) + 2.5 H2O ⇌ Sb(OH)5 | ‒3.7 | |
| Sb2O5(am) + 5 H2O ⇌ 2 Sb(OH)5 | ‒7.400 |
Arsenic(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [12] | Nordstrom and Archer, 2003 [13] | Nordstrom et al., 2014 [14] |
|---|---|---|---|
| As(OH)4‒ + H+ ⇌ As(OH)3 + H2O | 9.29 | 9.17 | 9.24 ± 0.02 |
Arsenic(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer [12] | Khodakovsky et al. (1968) [15] | Nordstrom and Archer, 2003 [13] | Nordstrom et al., 2014 [14] |
|---|---|---|---|---|
| H2AsO4‒ + H+ ⇌ H3AsO4 | 2.24 | 2.21 | 2.26 ± 0.078 | 2.25 ± 0.04 |
| HAsO42‒ + H+ ⇌ H2AsO4‒ | 6.93 | 6.99 ± 0.1 | 6.98 ± 0.11 | |
| AsO43‒ + H+ ⇌ HAsO42‒ | 11.51 | 11.80 ± 0.1 | 11.58 ± 0.05 | |
| HAsO42‒ + 2 H+ ⇌H3AsO4 | 9.20 | |||
| AsO43‒ + 3 H+ ⇌ H3AsO4 | 20.70 |
Barium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [16] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [18] |
|---|---|---|---|
| Ba2+ + H2O ⇌ BaOH+ + H+ | –13.47 | –13.47 | –13.32 ± 0.07 |
Berkelium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [19] |
|---|---|
| Bk3+ + 3 H2O ⇌ Bk(OH)3(s) + 3 H+ | –13.5 ± 1.0 |
Beryllium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [20] |
|---|---|
| Be2+ + H2O ⇌ BeOH+ + H+ | –5.10 |
| Be2+ + 2 H2O ⇌ Be(OH)2 + 2 H+ | –23.65 |
| Be2+ + 3 H2O ⇌ Be(OH)3– + 3 H+ | –23.25 |
| Be2+ + 4 H2O ⇌ Be(OH)42– + 4 H+ | –37.42 |
| 2 Be2+ + H2O ⇌ Be2OH3+ + H+ | –3.97 |
| 3 Be2+ + 3 H2O ⇌ Be3(OH)33+ + 3 H+ | –8.92 |
| 6 Be2+ + 8 H2O ⇌ Be6(OH)84+ + 8 H+ | –27.2 |
| α-Be(OH)2(cr) + 2 H+ ⇌ Be2+ + 2 H2O | 6.69 |
Bismuth
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [21] | Lothenbach et al., 1999 [9] | NIST46 [4] | Kitamura et al., 2010 [10] | Brown and Ekberg, 2016 [22] |
|---|---|---|---|---|---|
| Bi3+ + H2O ⇌ BiOH2+ + H+ | –1.0 | –0.92 | –1.1 | –0.920 | –0.92 ± 0.15 |
| Bi3+ + 2 H2O ⇌ Bi(OH)2+ + 2 H+ | (–4) | –2.56 | –4.5 | –2.560 ± 1.000 | –2.59 ± 0.26 |
| Bi3+ + 3 H2O ⇌ Bi(OH)3 + 3 H+ | –8.86 | –5.31 | –9.0 | –8.940 ± 0.500 | –8.78 ± 0.20 |
| Bi3+ + 4 H2O ⇌ Bi(OH)4– + 4 H+ | –21.8 | –18.71 | –21.2 | –21.660 ± 0.870 | –22.06 ± 0.14 |
| 3 Bi3+ + 4 H2O ⇌ Bi3(OH)45+ + 4 H+ | –0.80 | –0.800 | |||
| 6 Bi3+ + 12 H2O ⇌ Bi6(OH)126+ + 12 H+ | 1.34 | 1.340 | 0.98 ± 0.13 | ||
| 9 Bi3+ + 20 H2O = Bi9(OH)207+ + 20 H+ | –1.36 | –1.360 | |||
| 9 Bi3+ + 21 H2O = Bi9(OH)216+ + 21 H+ | –3.25 | –3.250 | |||
| 9 Bi3+ + 22 H2O = Bi9(OH)225+ + 22 H+ | –4.86 | –4.860 | |||
| Bi(OH)3(am) + 3 H+ = Bi3+ + 3 H2O | 31.501 ± 0.927 | ||||
| α-Bi2O3(cr) + 6 H+ = 2 Bi3+ + 3 H2O | 0.76 | ||||
| BiO1.5(s, α) + 3 H+ = Bi3+ + 1.5 H2O | 3.46 | 31.501 ± 0.927 | 2.88 ± 0.64 |
Boron
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [23] | NIST46 [4] |
|---|---|---|
| B(OH)3 + H2O ⇌ Be(OH)4+ + H+ | –9.236 | –9.236 ± 0.002 |
| 2 B(OH)3 ⇌ B2(OH)5– + H+ | –9.36 | –9.306 |
| 3 B(OH)3 ⇌ B3O3(OH)4– + H+ + 2 H2O | –7.03 | –7.306 |
| 4 B(OH)3 ⇌ B4O5(OH)42– + 2 H+ + 3 H2O | –16.3 | –15.032 |
Cadmium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [24] | Powell et al., 2011 [25] | Brown and Ekberg, 2016 [26] |
|---|---|---|---|
| Cd2+ + H2O ⇌ CdOH+ + H+ | −10.08 | –9.80 ± 0.10 | −9.81 ± 0.10 |
| Cd2+ + 2 H2O ⇌ Cd(OH)2 + 2 H+ | –20.35 | –20.19 ± 0.13 | −20.6 ± 0.4 |
| Cd2+ + 3 H2O ⇌ Cd(OH)3– + 3 H+ | <–33.3 | –33.5 ± 0.5 | −33.5 ± 0.5 |
| Cd2+ + 4 H2O ⇌ Cd(OH)42– + 4 H+ | –47.35 | –47.28 ± 0.15 | −47.25 ± 0.15 |
| 2 Cd2+ + H2O ⇌ Cd2OH3+ + H+ | –9.390 | –8.73 ± 0.01 | −8.74 ± 0.10 |
| 4 Cd2+ + 4 H2O ⇌ Cd4(OH)44+ + H+ | –32.85 | ||
| Cd(OH)2(s) ⇌ Cd2+ + 2 OH– | –14.28 ± 0.12 | ||
| Cd(OH)2(s) + 2 H+ ⇌ Cd2+ + 2 H2O | 13.65 | 13.72 ± 0.12 | 13.71 ± 0.12 |
Calcium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [16] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [27] |
|---|---|---|---|
| Ca2+ + H2O ⇌ CaOH+ + H+ | –12.85 | –12.78 | –12.57 ± 0.03 |
| Ca(OH)2(cr) + 2 H+ ⇌ Ca2+ + 2 H2O | 22.80 | 22.8 | 22.75 ± 0.02 |
Californium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [19] |
|---|---|
| Cf3+ + 3 H2O ⇌ Bk(OH)3(s) + 3 H+ | –13.0 ± 1.0 |
Cerium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | NIST46 [4] | Brown and Ekberg, 2016 [29] |
|---|---|---|---|
| Ce3+ + H2O ⇌ CeOH2+ + H+ | –8.3 | –8.3 | –8.31 ± 0.03 |
| 2 Ce3+ + 2 H2O ⇌ Ce2(OH)24+ + 2 H+ | –16.0 ± 0.2 | ||
| 3 Ce3+ + 5 H2O ⇌ Ce3(OH)54+ + 5 H+ | –34.6 ± 0.3 | ||
| Ce(OH)3(s) + 3 H+ ⇌ Ce3+ + 3 H2O | 18.5 ± 0.5 | ||
| Ce(OH)3(s) ⇌ Ce3+ + 3 OH– | –22.1 ± 0.9 |
Chromium(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K (The divalent state is unstable in water, producing hydrogen whilst being oxidised to a higher valency state (Baes and Mesmer, 1976). The reliability of the data is in doubt.):
| Reaction | NIST46 [4] | Ball and Nordstrom, 1988 [30] |
|---|---|---|
| Cr2+ + H2O ⇌ CrOH+ + H+ | –5.5 | |
| Cr(OH)2(s) ⇌ Cr2+ + 2 OH– | –17 ± 0.02 |
Chromium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [31] | Rai et al., 1987 [32] | Ball and Nordstrom, 1988 [30] | Brown and Ekberg, 2016 [33] |
|---|---|---|---|---|
| Cr3+ + H2O ⇌ CrOH2+ + H+ | –4.0 | –3.57 ± 0.08 | –3.60 ± 0.07 | |
| Cr3+ + 2 H2O ⇌ Cr(OH)2+ + 2 H+ | –9.7 | –9.84 | –9.65 ± 0.20 | |
| Cr3+ + 3 H2O ⇌ Cr(OH)3 + 3 H+ | –18 | –16.19 | –16.25 ± 0.19 | |
| Cr3+ + 4 H2O ⇌ Cr(OH)4- + 4 H+ | –27.4 | –27.65 ± 0.12 | –27.56 ± 0.21 | |
| 2 Cr3+ + 2 H2O ⇌ Cr2(OH)24+ + 2 H+ | –5.06 | –5.0 | –5.29 ± 0.16 | |
| 3 Cr3+ + 4 H2O ⇌ Cr3(OH)45+ + 4 H+ | –8.15 | –10.75 ± 0.15 | –9.10 ± 0.14 | |
| Cr(OH)3(s) + 3 H+ ⇌ Cr3+ + 3 H2O | 12 | 9.35 | 9.41 ± 0.17 | |
| Cr2O3(s) + 6 H+ ⇌ 2 Cr3+ + 3 H2O | 8.52 | |||
| CrO1.5(s) + 3 H+ ⇌ Cr3+ + 1.5 H2O | 7.83 ± 0.10 |
Chromium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [34] | Ball and Nordstrom, 1998 [30] |
|---|---|---|
| CrO42– + H+ ⇌ HCrO4– | 6.51 | 6.55 ± 0.04 |
| HCrO4– + H+ ⇌ H2CrO4 | –0.20 | |
| CrO42– + 2 H+ ⇌ H2CrO4 | 6.31 | |
| 2 HCrO4– ⇌ Cr2O72– + H2O | 1.523 | |
| 2 CrO42– + 2 H+ ⇌ Cr2O72– + H2O | 14.7 ± 0.1 |
Cobalt(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [35] | Brown and Ekberg, 2016 [36] |
|---|---|---|
| Co2+ + H2O ⇌ CoOH+ + H+ | –9.65 | −9.61 ± 0.17 |
| Co2+ + 2 H2O ⇌ Co(OH)2 + 2 H+ | –18.8 | −19.77 ± 0.11 |
| Co2+ + 3 H2O ⇌ Co(OH)3– + 3 H+ | –31.5 | −32.01 ± 0.33 |
| Co2+ + 4 H2O ⇌ Co(OH)42– + 4 H+ | –46.3 | |
| 2 Co2+ + H2O ⇌ Co2(OH)3+ + H+ | –11.2 | |
| 4 Co2+ + 4 H2O ⇌ Co4(OH)44+ + 4H+ | –30.53 | |
| Co(OH)2(s) + 2 H+ ⇌ Co2+ + 2 H2O | 12.3 | 13.24 ± 0.12 |
| CoO(s) + 2 H+ ⇌ Co2+ + H2O | 13.71 ± 0.10 |
Cobalt(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [37] |
|---|---|
| Co3+ + H2O ⇌ CoOH2+ + H+ | −1.07 ± 0.11 |
Copper(I)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [38] |
|---|---|
| Cu+ + H2O ⇌ CuOH + H+ | –7.8 ± 0.4 |
| Cu+ + 2 H2O ⇌ Cu(OH)2– + 2 H+ | –18.6 ± 0.6 |
Copper(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [39] | NIST46 [4] | Plyasunova et al., 1997 [40] | Powell et al., 2007 [41] | Brown and Ekberg, 2016 [38] |
|---|---|---|---|---|---|
| Cu2+ + H2O ⇌ CuOH+ + H+ | < –8 | –7.7 | –7.97 ± 0.09 | –7.95 ± 0.16 | –7.64 ± 0.17 |
| Cu2+ + 2 H2O ⇌ Cu(OH)2 + 2 H+ | (< –17.3) | –17.3 | –16.23 ± 0.15 | –16.2 ± 0.2 | –16.24 ± 0.03 |
| Cu2+ + 3 H2O ⇌ Cu(OH)3– + 3 H+ | (< –27.8) | –27.8 | –26.63 ± 0.40 | –26.60 ± 0.09 | –26.65 ± 0.13 |
| Cu2+ + 4 H2O ⇌ Cu(OH)42– + 4 H+ | –39.6 | –39.6 | –39.73 ± 0.17 | –39.74 ± 0.18 | –39.70 ± 0.19 |
| 2 Cu2+ + H2O ⇌ Cu2(OH)3+ + H+ | –6.71 ± 0.30 | –6.40 ± 0.12 | –6.41 ± 0.17 | ||
| 2 Cu2+ + 2 H2O ⇌ Cu2(OH)22+ + 2 H+ | –10.36 | –10.3 | –10.55 ± 0.17 | –10.43 ± 0.07 | –10.55 ± 0.02 |
| 3 Cu2+ + 4 H2O ⇌ Cu3(OH)42+ + 4 H+ | –20.95 ± 0.30 | –21.1 ± 0.2 | –21.2 ± 0.4 | ||
| CuO(s) + 2 H+ ⇌ Cu2+ + H2O | 7.62 | 7.64 ± 0.06 | 7.64 ± 0.06 | 7.63 ± 0.05 | |
| Cu(OH)2(s) + 2 H+ ⇌ Cu2+ + 2 H2O | 8.67 ± 0.05 | 8.68 ± 0.10 |
Curium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [42] |
|---|---|
| Cm3+ + H2O ⇌ Cm(OH)2+ + H+ | −7.66 ± 0.07 |
| Cm3+ + 2 H2O ⇌ Cm(OH)2+ + 2 H+ | −15.9 ± 0.1 |
| Cm3+ + 3 H2O ⇌ Cm(OH)3(s) + 3 H+ | −13.9 ± 0.4 |
Dysprosium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [43] |
|---|---|---|
| Dy3+ + H2O ⇌ DyOH2+ + H+ | −8.0 | −7.53 ± 0.14 |
| Dy3+ + 2 H2O ⇌ Dy(OH)2+ + 2 H+ | (–16.2) | |
| Dy3+ + 3 H2O ⇌ Dy(OH)3 + 3 H+ | (–24.7) | |
| Dy3+ + 4 H2O ⇌ Dy(OH)4− + 4 H+ | –33.5 | |
| 2 Dy3+ + 2 H2O ⇌ Dy2(OH)24+ + 2 H+ | −13.76 ± 0.20 | |
| 3 Dy3+ + 5 H2O ⇌ Dy3(OH)54+ + 5 H+ | −30.6 ± 0.3 | |
| Dy(OH)3(s) + 3 H+ ⇌ Dy3+ + 3 H2O | 15.9 | 16.26 ± 0.30 |
| Dy(OH)3(c) + OH− ⇌ Dy(OH)4− | −3.6 | |
| Dy(OH)3(c) ⇌ Dy(OH)3 | −8.8 |
Erbium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [44] |
|---|---|---|
| Er3+ + H2O ⇌ ErOH2+ + H+ | −7.9 | −7.46 ± 0.09 |
| Er3+ + 2 H2O ⇌ Er(OH)2+ + 2 H+ | (−15.9) | |
| Er3+ + 3 H2O ⇌ Er(OH)3 + 3 H+ | (−24.2) | |
| Er3+ + 4 H2O ⇌ Er(OH)4− + 4 H+ | −32.6 | |
| 2 Er3+ + 2 H2O ⇌ Er2(OH)24+ + 2 H+ | −13.65 | −13.50 ± 0.20 |
| 3 Er3+ + 5 H2O ⇌ Er3(OH)54+ + 5 H+ | <−29.3 | −31.0 ± 0.3 |
| Er(OH)3(s) + 3 H+ ⇌ Er3+ + 3 H2O | 15.0 | 15.79 ± 0.30 |
| Er(OH)3(c) + OH− ⇌ Er(OH)4− | −3.6 | |
| Er(OH)3(c) ⇌ Er(OH)3 | ~ −9.2 |
Europium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | NIST46 [4] | Hummel et al., 2002 [45] | Brown and Ekberg, 2016 [29] |
|---|---|---|---|---|
| Eu3+ + H2O ⇌ EuOH2+ + H+ | –7.8 | –7.64 ± 0.04 | –7.66 ± 0.05 | |
| Eu3+ + 2 H2O ⇌ Eu(OH)2+ + 2 H+ | –15.1 ± 0.2 | |||
| Eu3+ + 3 H2O ⇌ Eu(OH)3 + 3 H+ | –23.7 ± 0.1 | |||
| Eu3+ + 4 H2O ⇌ Eu(OH)4− + 4 H+ | –36.2 ± 0.5 | |||
| 2 Eu3+ + 2 H2O ⇌ Eu2(OH)24+ + 2 H+ | - | –14.1 ± 0.2 | ||
| 3 Eu3+ + 5 H2O ⇌ Eu3(OH)54+ + 5 H+ | - | –32.0 ± 0.3 | ||
| Eu(OH)3(s) + 3 H+ ⇌ Eu3+ + 3 H2O | 17.5 | 17.6 ± 0.8 (am) 14.9 ± 0.3 (cr) | 16.48 ± 0.30 | |
| Eu(OH)3(s) ⇌ Eu3+ + 3 OH– | –24.5 ± 0.7 (am) –26.5 (cr) |
Gadolinium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [46] | Brown and Ekberg, 2016 [47] |
|---|---|---|
| Gd3+ + H2O ⇌ GdOH2+ + H+ | –8.0 | –7.87 ± 0.05 |
| Gd3+ + 2 H2O ⇌ Gd(OH)2+ + 2 H+ | (–16.4) | |
| Gd3+ + 3 H2O ⇌ Gd(OH)3 + 3 H+ | (–25.2) | |
| Gd3+ + 4 H2O ⇌ Gd(OH)4– + 4 H+ | –34.4 | |
| 2 Gd3+ + 2 H2O ⇌ Gd2(OH)24+ + 2 H+ | –14.16 ± 0.20 | |
| 3 Gd3+ + 5 H2O ⇌ Gd3(OH)54+ + 5 H+ | –33.0 ± 0.3 | |
| Gd(OH)3(s) + 3 H+ ⇌ Gd3+ + 3 H2O | 15.6 | 17.20 ± 0.48 |
| Gd(OH)3(c) + OH– ⇌ Gd(OH)4– | –4.8 | |
| Gd(OH)3(c) ⇌ Gd(OH)3 | –9.6 |
Gallium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [48] | Smith et al., 2003 [49] | Brown and Ekberg, 2016 [50] |
|---|---|---|---|
| Ga3+ + H2O ⇌ GaOH2+ + H+ | –2.6 | –2.897 | –2.74 |
| Ga3+ + 2 H2O ⇌ Ga(OH)2+ + 2 H+ | –5.9 | –6.694 | –7.0 |
| Ga3+ + 3 H2O ⇌ Ga(OH)3 + 3 H+ | –10.3 | –11.96 | |
| Ga3+ + 4 H2O ⇌ Ga(OH)4– + 4 H+ | –16.6 | –16.588 | –15.52 |
| Ga(OH)3(s) ⇌ Ga3+ + 3 OH– | –37 | –37.0 | |
| GaO(OH)(s) + H2O ⇌ Ga3+ + 3 OH– | –39.06 | –39.1 | –40.51 |
Germanium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [51] | Wood and Samson, 2006 [52] | Filella and May, 2023 [53] |
|---|---|---|---|
| Ge(OH)4 ⇌ GeO(OH)3- + H+ | –9.31 | –9.32 ± 0.05 | –9.099 |
| Ge(OH)4 ⇌ GeO2(OH)22+ + 2 H+ | –21.9 | ||
| GeO2(OH)22– + H+ ⇌ GeO(OH)3– | 12.76 | ||
| 8 Ge(OH)4 ⇌ Ge8O16(OH)33- + 13 H2O + 3 H+ | –14.24 | ||
| 8 Ge(OH)4 + 3 OH– ⇌ Ge8(OH)353– | 28.33 | ||
| GeO2(s, hexa) + 2 H2O ⇌ Ge(OH)4 | –1.35 | –1.373 | |
| GeO2(s, tetra) + 2 H2O ⇌ Ge(OH)4 | -4.37 | –5.02 | –4.999 |
Gold(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [54] |
|---|---|
| Au(OH)3 +2 H+ ⇌ AuOH2+ + 2 H2O | 1.51 |
| Au(OH)3 + H+ ⇌ Au(OH)2+ + H2O | < 1.0 |
| Au(OH)3 + H2O ⇌ Au(OH)4– + H+ | –11.77 |
| Au(OH)3 + 2 H2O ⇌ Au(OH)52– + 2 H+ | –25.13 |
| Au(OH)52– + 3 H2O ⇌ Au(OH)63– + 3 H+ | < –41.1 |
| Au(OH)3(c) ⇌ Au(OH)3 | –5.51 |
Hafnium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [55] | Brown and Ekberg, 2016 [56] |
|---|---|---|
| Hf4+ + H2O ⇌ HfOH3+ + H+ | –0.25 | −0.26 ± 0.10 |
| Hf4+ + 2 H2O ⇌ Hf(OH)22+ + 2 H+ | (–2.4) | |
| Hf4+ + 3 H2O ⇌ Hf(OH)3+ + 3 H+ | (–6.0) | |
| Hf4+ + 4 H2O ⇌ Hf(OH)4 + 4 H+ | –10.7* | −3.75 ± 0.34* |
| Hf4+ + 5 H2O ⇌ Hf(OH)5– + 5 H+ | –17.2 | |
| 3 Hf4+ + 4 H2O ⇌ Hf3(OH)48+ + 4 H+ | 0.55 ± 0.30 | |
| 4 Hf4+ + 8 H2O ⇌ Hf4(OH)88+ + 8 H+ | 6.00 ± 0.30 | |
| HfO2(s) + 4 H+ ⇌ Hf4+ + 2 H2O | –1.2* | –5.56 ± 0.15* |
| HfO2(am) + 4 H+ ⇌ Hf4+ + 2 H2O | –3.11 ± 0.20 |
*Errors in compilations concerning equilibrium and/or data elaboration. Data not recommended. Strongly suggested to refer to the original papers.
Holmium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [57] |
|---|---|---|
| Ho3+ + H2O ⇌ HoOH2+ + H+ | −8.0 | −7.43 ± 0.05 |
| 2 Ho3+ + 2 H2O ⇌ Ho2(OH)24+ + 2 H+ | −13.5 ± 0.2 | |
| 3 Ho3+ + 5 H2O ⇌ Ho3(OH)54+ + 5 H+ | −30.9 ± 0.3 | |
| Ho(OH)3(s) + 3 H+ ⇌ Ho3+ + 3 H2O | 15.4 | 15.60 ± 0.30 |
Indium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [58] | NIST46 [4] | Brown and Ekberg, 2016 [59] |
|---|---|---|---|
| In3+ + H2O ⇌ InOH2+ + H+ | –4.00 | –3.927 | –3.96 |
| In3+ + 2 H2O ⇌ In(OH)2+ + 2 H+ | –7.82 | –7.794 | –9.16 |
| In3+ + 3 H2O ⇌ In(OH)3 + 3 H+ | –12.4 | –12.391 | |
| In3+ + 4 H2O ⇌ In(OH)4– + 4 H+ | –22.07 | –22.088 | –22.05 |
| In(OH)3(s) ⇌ In3+ + 3 OH– | –36.92 | –36.9 | –36.92 |
| 1/2 In2O3(s) + 3/2 H2O ⇌ In3+ + 3 OH– | –35.24 |
Iridium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [60] |
|---|---|
| Ir3+ + H2O ⇌ IrOH2+ + H+ | ‒3.77 ± 0.10 |
| Ir3+ + 2 H2O ⇌ Ir(OH)2+ + 2 H+ | ‒8.46 ± 0.20 |
| Ir(OH)3(s) + 3 H+ ⇌ Ir3+ + 3 H2O | 8.88 ± 0.20 |
Iron(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [61] | Nordstrom et al., 1990 [17] | Hummel et al., 2002 [45] | Lemire et al., 2013 [62] | Brown and Ekberg, 2016 [63] |
|---|---|---|---|---|---|
| Fe2+ + H2O ⇌ FeOH+ + H+ | –9.3 | –9.5 | –9.5 | –9.1 ± 0.4 | −9.43 ± 0.10 |
| Fe2+ + 2 H2O ⇌ Fe(OH)2 + 2 H+ | –20.5 | −20.52 ± 0.08 | |||
| Fe2+ + 3 H2O ⇌ Fe(OH)3- + 3 H+ | –29.4 | −32.68 ± 0.15 | |||
| Fe(OH)2(s) +2 H+ ⇌ Fe2+ + 2 H2O | 12.27 ± 0.88 |
Iron(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [61] | Lemire et al., 2013 [62] | Brown and Ekberg, 2016 [64] |
|---|---|---|---|
| Fe3+ + H2O ⇌ FeOH2+ + H+ | –2.19 | −2.15 ± 0.07 | –2.20 ± 0.02 |
| Fe3+ + 2 H2O ⇌ Fe(OH)2+ + 2 H+ | –5.67 | −4.8 ± 0.4 | –5.71 ± 0.10 |
| Fe3+ + 3 H2O ⇌ Fe(OH)3 + 3 H+ | <–12 | <–14 | –12.42 ± 0.20 |
| Fe3+ + 4 H2O ⇌ Fe(OH)4– + 4 H+ | –21.6 | −21.5 ± 0.5 | –21.60 ± 0.23 |
| 2 Fe3+ + 2 H2O ⇌ Fe2(OH)24+ + 2 H+ | –2.95 | –2.91 ± 0.07 | –2.91 ± 0.07 |
| 3 Fe3+ + 4 H2O ⇌ Fe3(OH)45+ + 4 H+ | –6.3 | −6.3 ± 0.1 | |
| Fe(OH)3(s) +3 H+ ⇌ Fe3+ + 3 H2O 2-line ferrihydrite | 2.5 | 3.5 | 3.50 ± 0.20 |
| Fe(OH)3(s) ⇌ Fe3+ + 3 OH− 6-line ferrihydrite | −38.97 ± 0.64 | ||
| α-FeOOH(s)+ 3 H+ ⇌ Fe3+ + 2 H2O goethite | 0.5 | 0.33 ± 0.10 | |
| α-FeOOH + H2O ⇌ Fe3+ + 3 OH− goethite | −41.83 ± 0.37 | ||
| 0.5 α-Fe2O3(s)+ 3 H+ ⇌ Fe3+ + 1.5 H2O hematite | 0.36 ± 0.40 | ||
| 0.5 α-Fe2O3 + 1.5 H2O ⇌ Fe3+ + 3 OH− hematite | −42.05 ± 0.26 | ||
| 0.5 γ-Fe2O3(s) + 3 H+ ⇌ Fe3+ + 1.5 H2O maghemite | 1.61 ± 0.61 | ||
| 0.5 γ-Fe2O3 + 1.5 H2O ⇌ Fe3+ + 3 OH− maghemite | −40.59 ± 0.29 | ||
| α-FeOOH(s)+ 3 H+ ⇌ Fe3+ + 2 H2O lepidocrocite | 1.85 ± 0.37 | ||
| γ-FeOOH + H2O ⇌ Fe3+ + 3 OH− lepidocrocite | −40.13 ± 0.37 | ||
| Fe(OH)3(s) + 3 H+ ⇌ Fe3+ + 3 H2O magnetite | −12.26 ± 0.26 |
Lanthanum
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [65] | Brown and Ekberg, 2016 [66] |
|---|---|---|
| La3+ + H2O ⇌ LaOH2+ + H+ | –8.5 | –8.89 ± 0.10 |
| 2 La3+ + 2 H2O ⇌ La2(OH)24+ + 2 H+ | ≤ –17.5 | –17.57 ± 0.20 |
| 3 La3+ + 5 H2O ⇌ La3(OH)54+ + 5 H+ | ≤ –38.3 | –37.8 ± 0.3 |
| 5 La3+ + 9 H2O ⇌ La5(OH)96+ + 9 H+ | –71.2 | |
| La(OH)3(s) + 3 H+ ⇌ La3+ + 3 H2O | 20.3 | 19.72 ± 0.34 |
Lead(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [67] | NIST46 [4] | Powell et al, 2009 [68] | Brown and Ekberg, 2016 [69] | Cataldo et al., 2018 [70] |
|---|---|---|---|---|---|
| Pb2+ + H2O ⇌ PbOH+ + H+ | –7.71 | –7.6 | –7.46 ± 0.06 | –7.49 ± 0.13 | –6.47± 0.03 |
| Pb2+ + 2 H2O ⇌ Pb(OH)2 + 2 H+ | –17.12 | –17.1 | –16.94 ± 0.09 | –16.99 ± 0.06 | –16.12 ± 0.01 |
| Pb2+ + 3 H2O ⇌ Pb(OH)3- + 3 H+ | –28.06 | –28.1 | –28.03± 0.06 | –27.94 ± 0.21 | –28.4 ± 0.1 |
| Pb2+ + 4 H2O ⇌ Pb(OH)42- + 4 H+ | –40.8 | ||||
| 2 Pb2+ + H2O ⇌ Pb2(OH)3+ + H+ | –6.36 | –6.4 | –7.28± 0.09 | –6.73 ± 0.31 | |
| 3 Pb2+ + 4 H2O ⇌ Pb3(OH)42+ + 4 H+ | –23.88 | –23.9 | –23.01 ± 0.07 | –23.43 ± 0.10 | |
| 3 Pb2+ + 5 H2O ⇌ Pb3(OH)5+ + 5 H+ | –31.11 ± 0.10 | ||||
| 4 Pb2+ + 4 H2O ⇌ Pb4(OH)44+ + 4 H+ | –20.88 | –20.9 | –20.57± 0.06 | –20.71 ± 0.18 | |
| 6 Pb2+ + 8 H2O ⇌ Pb6(OH)84+ + 8 H+ | –43.61 | –43.6 | –42.89± 0.07 | –43.27 ± 0.47 | |
| PbO(s) + 2 H+ ⇌ Pb2+ + H2O | 12.62 (red) 12.90 (yellow) | ||||
| PbO(s) +H2O ⇌ Pb2+ + 2 OH– | –15.28 (red) | -15.3 | –15.3 (red) –15.1 (yellow) | –15.37 ± 0.04 (red) –15.1 ± 0.08 (yellow) | |
| Pb2O(OH)2(s) +H2O ⇌ 2 Pb2+ + 4 OH– | –14.9 | ||||
| PbO(s) +H2O ⇌ Pb(OH)2 | –4.4 (red) –4.2 (yellow) | ||||
| Pb2O(OH)2(s) +H2O ⇌ 2 Pb(OH)2 | –4.0 | ||||
| PbO(s) + 2 H2O ⇌ Pb(OH)3– + H+ | –1.4 (red) –1.2 (yellow) | ||||
| Pb2O(OH)2(s) + 2 H2O ⇌ 2 Pb(OH)3– + 2 H+ | –1.0 |
Lead(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Feitknecht and Schindler, 1963 [71] |
|---|---|
| β-PbO2 + 2 H2O ⇌ Pb4+ + 4 OH– | –64 |
| β-PbO2 + 2 H2O + 2 OH– ⇌ Pb(OH)62– | –4.5 |
Lithium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [72] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [73] |
|---|---|---|---|
| Li+ + H2O ⇌ LiOH + H+ | –13.64 | –13.64 | –13.84 ± 0.14 |
Magnesium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [74] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [75] |
|---|---|---|---|
| Mg2+ + H2O ⇌ MgOH+ + H+ | –11.44 | –11.44 | –11.70 ± 0.04 |
| 4 Mg2+ + 4 H2O ⇌ Mg4(OH)44+ + 4 H+ | –39.71 | ||
| Mg(OH)2(cr) + 2 H+ ⇌ Mg2+ + 2 H2O | 16.84 | 16.84 | 17.11 ± 0.04 |
Manganese(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Perrin et al., 1969 [76] | Baes and Mesmer, 1976 [77] | Nordstrom et al., 1990 [17] | Hummel et al., 2002 [45] | Brown and Ekberg, 2016 [78] |
|---|---|---|---|---|---|
| Mn2+ + H2O ⇌ MnOH+ + H+ | –10.59 | –10.59 | –10.59 | –10.59 | −10.58 ± 0.04 |
| Mn2+ + 2 H2O ⇌ Mn(OH)2 + 2 H+ | –22.2 | −22.18 ± 0.20 | |||
| Mn2+ + 3 H2O ⇌ Mn(OH)3– + 3 H+ | –34.8 | −34.34 ± 0.45 | |||
| Mn2+ + 4 H2O ⇌ Mn(OH)42– + 4 H+ | –48.3 | −48.28 ± 0.40 | |||
| 2 Mn2+ + H2O ⇌ Mn2OH3+ + H+ | –10.56 | ||||
| 2 Mn2+ + 3 H2O ⇌ Mn2(OH)3+ + 6 H+ | –23.90 | ||||
| Mn(OH)2(s) + 2 H+ ⇌ Mn2+ + 2 H2O | 15.2 | 15.2 | 15.2 | 15.19 ± 0.10 | |
| MnO(s) + 2 H+ ⇌ Mn2+ + H2O | 17.94 ± 0.12 |
Manganese(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [79] |
|---|---|
| Mn3+ + H2O ⇌ MnOH2+ + H+ | –11.70 ± 0.04 |
Mercury(I)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [80] | Brown and Ekberg, 2016 [81] |
|---|---|---|
| Hg22+ + H2O ⇌ Hg2OH+ + H+ | −5.0a | −4.45 ± 0.10 |
(a) 0.5 M HClO4
Mercury(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [82] | Powell et all, 2005 [83] | Brown and Ekberg, 2016 [79] |
|---|---|---|---|
| Hg2+ + H2O ⇌ HgOH+ + H+ | −3.40 | –3.40 ± 0.08 | –3.40 ± 0.08 |
| Hg2+ + 2 H2O ⇌ Hg(OH)2 + 2 H+ | -6.17 | –5.98 ± 0.06 | −5.96 ± 0.07 |
| Hg2+ + 3 H2O ⇌ Hg(OH)3– + 3 H+ | –21.1 | –21.1 ± 0.3 | |
| HgO(s) + 2 H+ ⇌ Hg2+ + H2O | 2.56 | 2.37 ± 0.08 | 2.37 ± 0.08 |
Molybdenum(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution, T = 298.15 K and I = 3 M NaClO4 (a) or 0.1 M Na+ medium, Data at I = 0 are not available (b):
| Reaction | Baes and Mesmer, 1976 [84] | Jolivet, 2000 [85] | NIST46 [4] | Crea et al., 2017 [86] |
|---|---|---|---|---|
| MoO42– + H+ ⇌ HMoO4– | 3.89a | 4.24 | 4.47 ± 0.02 | |
| MoO42– + 2 H+ ⇌ H2MoO4 | 7.50a | 8.12 ± 0.03 | ||
| HMoO4– + H+ ⇌ H2MoO4 | 4.0 | |||
| Mo7O246– + H+ ⇌ HMo7O245– | 4.4 | |||
| HMo7O245– + H+ ⇌ H2Mo7O244– | 3.5 | |||
| H2Mo7O244– + H+ ⇌ H3Mo7O243– | 2.5 | |||
| 7 MoO42-+ 8 H+ ⇌ Mo7O246– + 4 H2O | 57.74a | 52.99b | 51.93 ± 0.04 | |
| 7 MoO42– + 9 H+ ⇌ Mo7O23(OH)5– + 4 H2O | 62.14a | 58.90 ± 0.02 | ||
| 7 MoO42– + 10 H+ ⇌ Mo7O22(OH)24– + 4 H2O | 65.68a | 64.63 ± 0.05 | ||
| 7 MoO42– + 11 H+ ⇌ Mo7O21(OH)33– + 4 H2O | 68.21a | 68.68 ± 0.06 | ||
| 19 MoO42- + 34 H+ ⇌ Mo19O594– + 17 H2O | 196.3a | 196a | ||
| MoO3(s) + H2O ⇌ MoO42– + 2 H+ | –12.06a |
Neodymium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | NIST46 [4] | Neck et al., 2009 [87] | Brown and Ekberg, 2016 [29] |
|---|---|---|---|---|
| Nd3+ + H2O ⇌ NdOH2+ + H+ | –8.0 | –8.0 | –7.4 ± 0.4 | –8.13 ± 0.05 |
| Nd3+ + 2 H2O ⇌ Nd(OH)2+ + 2 H+ | (–16.9) | –15.7 ± 0.7 | ||
| Nd3+ + 3 H2O ⇌ Nd(OH)3(aq) + 3 H+ | (–26.5) | –26.2 ± 0.5 | ||
| Nd3+ + 4 H2O ⇌ Nd(OH)4− + 4 H+ | (–37.1) | –37.4 | –40.7 ± 0.7 | |
| 2 Nd3+ + 2 H2O ⇌ Nd2(OH)24+ + 2 H+ | –13.86 | –13.9 | –15.56 ± 0.20 | |
| 3 Nd3+ + 5 H2O ⇌ Nd3(OH)54+ + 5 H+ | < –28.5 | –34.2 ± 0.3 | ||
| Nd(OH)3(s) + 3 H+ ⇌ Nd3+ + 3 H2O | 18.6 | 17.2 ± 0.4 | 17.89 ± 0.09 | |
| Nd(OH)3(s) ⇌ Nd3+ + 3 OH– | –23.2 ± 0.9 | –21.5 (act) –23.1(inact) |
Neptunium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [88] | Grenthe et al, 2020 [6] |
|---|---|---|
| Np3+ + H2O ⇌ NpOH2+ + H+ | -7.3 ± 0.5 | –6.8 ± 0.3 |
Neptunium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [89] | NIST46 [4] | Brown and Ekberg, 2016 [90] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| Np4+ + H2O ⇌ NpOH3+ + H+ | –1.49 | –1.5 | –1.31 ± 0.05 | 0.5 ± 0.2 |
| Np4+ + 2 H2O ⇌ Np(OH)22+ + 2 H+ | –3.7 ± 0.3 | 0.3 ± 0.3 | ||
| Np4+ + 4 H2O ⇌ Np(OH)4 + 4 H+ | –10.0 ± 0.9 | –8 ± 1 | ||
| Np4+ + 4 OH- ⇌ NpO2(am, hyd) + 2 H2O | 52 | 54.9 ± 0.4 | 57.5 ± 0.3 | 56.7 ± 0.5 |
Neptunium(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [89] | Brown and Ekberg, 2016 [91] | Grenthe et al, 2020 [6] |
|---|---|---|---|
| NpO2+ + + H2O ⇌ NpO2(OH) + H+ | –8.85 | –10.7 ± 0.5 | –11.3 ± 0.7 |
| NpO2+ + 2 H2O ⇌ NpO2(OH)2- + 2 H+ | –22.8 ± 0.7 | –23.6 ± 0.5 | |
| NpO2+ + H2O ⇌ NpO2(OH)(am, fresh) + H+ | ≤ –4.7 | –5.21 ± 0.05 | –5.3 ± 0.2 |
| NpO2+ + H2O ⇌ NpO2(OH)(am, aged) + H+ | –4.53 ± 0.06 | –4.7 ± 0.5 |
Neptunium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [92] | NIST46 [4] | Brown and Ekberg, 2016 [93] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| NpO22+ + H2O ⇌ NpO2(OH)+ + H+ | –5.15 | –5.12 | –5.1 ± 0.2 | –5.1 ± 0.4 |
| NpO22+ + 3 H2O ⇌ NpO2(OH)3- + 3 H+ | –21 ± 1 | |||
| NpO22+ + 4 H2O ⇌ NpO2(OH)42- + 4 H+ | –32 ± 1 | |||
| 2 NpO22+ + 2 H2O ⇌ (NpO2)2(OH)22+ + 2 H+ | –6.39 | –6.39 | –6.2 ± 0.2 | –6.2 ± 0.2 |
| 3 NpO22+ + 5 H2O ⇌ (NpO2)3(OH)5+ + 5 H+ | –17.49 | –17.49 | –17.0 ± 0.2 | –17.1 ± 0.2 |
| NpO22+ + 2 H2O ⇌ NpO3.H2O(cr) + 2 H+ | ≥-6.6 | –5.4 ± 0.4 | –5.4 ± 0.4 |
Nickel(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Feitknecht and Schindler, 1963 [71] | Baes and Messmer, 1976 [94] | NIST46 [4] | Gamsjäger et al., 2005 [95] | Thoenen et al., 2014 [96] | Brown and Ekberg, 2016 [97] |
|---|---|---|---|---|---|---|
| Ni2+ + H2O ⇌ NiOH+ + H+ | –9.86 | –9.9 | –9.54 ± 0.14 | –9.54 ± 0.14 | –9.90 ± 0.03 | |
| Ni2+ + 2 H2O ⇌ Ni(OH)2 + 2 H+ | –19 | –19 | < –18 | –21.15 ± 0.0 | ||
| Ni2+ + 3 H2O ⇌ Ni(OH)3– + 3 H+ | –30 | –30 | –29.2 ± 1.7 | –29.2 ± 1.7 | ||
| Ni2+ + 4 H2O ⇌ Ni(OH)42– + 4 H+ | < –44 | |||||
| 2 Ni2+ + H2O ⇌ Ni2(OH)3+ + H+ | –10.7 | –10.6 ± 1.0 | –10.6 ± 1.0 | –10.6 ± 1.0 | ||
| 4 Ni2+ + 4 H2O ⇌ Ni4(OH)44+ + 4 H+ | –27.74 | –27.7 | –27.52 ± 0.15 | –27.52 ± 0.15 | –27.9 ± 0.6 | |
| β-Ni(OH)2(s) + 2 H+ ⇌ Ni2+ + 2 H2O | 10.8 | 11.02 ± 0.20 | 10.96 ± 0.20 11.75 ± 0.13 (microcr) | |||
| Ni(OH)2(s) ⇌ Ni2+ + 2 OH– | –17.2 (inactive) | –17.2 | –16.97± 0.20 (β) –17.2 ± 1.3 (cr) | |||
| Ni(OH)2(s) + OH– ⇌ Ni(OH)3– | –4.2 (inactive) | |||||
| NiO(cr) + 2 H+ ⇌ Ni2+ + H2O | 12.38 ± 0.06 | 12.48 ± 0.15 |
Niobium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [72] | Filella and May, 2020 [98] |
|---|---|---|
| Nb(OH)5 + H+ ⇌ Nb(OH)4+ + H2O | ~ –0.6 | 1.603 |
| Nb(OH)5 + H2O ⇌ Nb(OH)6– + H+ | ~ –4.8 | –4.951 |
| Nb6O198– + H+ ⇌ HNb6O197– | 14.95 | |
| HNb6O197– + H+ ⇌ H2Nb6O196– | 13.23 | |
| H2Nb6O196– + H+ ⇌ H3Nb6O195– | 11.73 | |
| 1/2 Nb2O5(act) + 5/2 H2O ⇌ Nb(OH)5 | ~ –7.4 | |
| Nb(OH)5(am,s) ⇌ Nb(OH)5 | –7.510 | |
| Nb2O5(s) + 5 H2O ⇌ 2 Nb(OH)5 | –18.31 |
Osmium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution, I = 0.1 M and T = 298.15 K:
| Reaction | Galbács et al., 1983 [99] |
|---|---|
| OsO2(OH)42– + H+ ⇌ HOsO2(OH)4– | 10.4 |
| HOsO2(OH)4– + H+ ⇌ H2OsO2(OH)4 | 8.5 |
Osmium(VIII)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Galbács et al., 1983 [99] |
|---|---|
| OsO2(OH)3(O-)aq + H+ ⇌ OsO2(OH)4aq | 12.2a |
| OsO2(OH)2(O-)2aq + H+ ⇌ OsO2(OH)3(O-)aq | 14.4b |
(a) At I = 0.1 M (b) At I = 2.5 M
Palladium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Perrin et al., 1969 [100] | Hummel et al., 2002 [45] | Kitamura and Yul, 2010 [101] | Brown and Ekberg, 2016 [102] |
|---|---|---|---|---|
| Pd2+ + H2O ⇌ PdOH+ + H+ | −0.96 | −0.65 ± 0.64 | −1.16 ± 0.30 | |
| Pd2+ + 2 H2O ⇌ Pd(OH)2 + 2 H+ | −2.6 | −4 ± 1 | −3.11 ± 0.63 | −3.07 ± 0.16 |
| Pd2+ + 3 H2O ⇌ Pd(OH)3− + 3 H+ | −15.5 ± 1 | −14.20 ± 0.63 | ||
| Pd(OH)2(am) + 2 H+ ⇌ Pd2+ + 2 H2O | −3.3 ± 1 | −3.4 ± 0.2 |
Plutonium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [103] | NIST46 [4] | Brown and Ekberg, 2016 [104] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| Pu3+ + H2O ⇌ PuOH2+ + H+ | –7.0 | –6.9 ± 0.2 | –6.9 ± 0.3 | |
| Pu3+ + 3 H2O ⇌ Pu(OH)3(cr) + 3 H+ | –19.65 | –15.8 ± 0.8 | –15 ± 1 |
Plutonium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [105] | NIST46 [4] | Brown and Ekberg, 2016 [106] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| Pu4+ + H2O ⇌ PuOH 3+ + H+ | –0.5 | –0.5 | –0.7 ± 0.1 | 0.6 ± 0.2 |
| Pu4+ + 2 H2O ⇌ Pu(OH)22+ + 2 H+ | (–2.3) | 0.6 ± 0.3 | ||
| Pu4+ + 3 H2O ⇌ Pu(OH)3+ + 3 H+ | (–5.3) | –2.3 ± 0.4 | ||
| Pu4+ + 4 H2O ⇌ Pu(OH)4 + 4 H+ | –9.5 | –12.5 ± 0.7 | –8.5 ± 0.5 | |
| Pu4+ + 4 OH- ⇌ PuO2(am, hyd) + 2 H2O | 49.5 | 47.9 ± 0.4 (0w) 53.8 ± 0.5 (1w) | 58.3 ± 0.5 |
Plutonium(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [107] | NIST46 [4] | Brown and Ekberg, 2016 [108] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| PuO2+ + H2O ⇌ PuO2(OH) + H+ | –1.49 | –1.5 | –1.31 ± 0.05 | 0.5 ± 0.2 |
| PuO2+ + H2O ⇌ PuO2(OH)(am) + H+ | –3.7 ± 0.3 | 0.3 ± 0.3 |
Plutonium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [109] | NIST46 [4] | Brown and Ekberg, 2016 [110] | Grenthe et al, 2020 [6] |
|---|---|---|---|---|
| PuO22+ + H2O ⇌ PuO2(OH)+ + H+ | –5.6 | –5.6 | –5.36 ± 0.09 | –5.5 ± 0.5 |
| PuO22+ + 2 H2O ⇌ PuO2(OH)2 + 2 H+ | –12.9 ± 0.2 | –13 ± 1 | ||
| PuO22+ + 3 H2O ⇌ PuO2(OH)3- + 3 H+ | –24 ± 1 | |||
| 2 PuO22+ + 2 H2O ⇌ (PuO2)2(OH)22+ + 2 H+ | –8.36 | –8.36 | –7.8 ± 0.5 | –7 ± 1 |
| 3 PuO22+ + 5 H2O ⇌ (PuO2)3(OH)5+ + 5 H+ | –21.65 | –21.65 | ||
| PuO22+ + 2 OH- ⇌ PuO2(OH)2(am, hyd) | 22.8 ± 0.6 |
Potassium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [72] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [111] |
|---|---|---|---|
| K+ + H2O ⇌ KOH + H+ | –14.46 | –14.46 | –14.5 ± 0.4 |
Praseodymium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | NIST46 [4] | Brown and Ekberg, 2016 [29] |
|---|---|---|---|
| Pr3+ + H2O ⇌ PrOH2+ + H+ | –8.1 | –8.30 ± 0.03 | |
| 2 Pr3+ + 2 H2O ⇌ Pr2(OH)24+ + 2 H+ | –16.31 ± 0.20 | ||
| 3 Pr3+ + 5 H2O ⇌ Pr3(OH)54+ + 5 H+ | –35.0 ± 0.3 | ||
| Pr(OH)3(s) + 3 H+ ⇌ Pr3+ + 3 H2O | 19.5 | 18.57 ± 0.20 | |
| Pr(OH)3(s) ⇌ Pr3+ + 3 OH– | –22.3 ± 1.0 |
Radium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Nordstrom et al., 1990 [17] |
|---|---|
| Ra2+ + H2O ⇌ RaOH+ + H+ | –13.49 |
Rhodium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Perrin et al., 1969 [112] | Baes and Mesmer, 1976 [113] | Brown and Ekberg [114] |
|---|---|---|---|
| Rh3+ + H2O ⇌ RhOH2+ + H+ | ‒3.43 | ‒3.4 | ‒3.09 ± 0.1 |
| Rh(OH)3(c) + OH‒ ⇌ Rh(OH)4‒ | ‒3.9 |
Samarium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | NIST46 [4] | Brown and Ekberg [29] |
|---|---|---|---|
| Sm3+ + H2O ⇌ SmOH2+ + H+ | –7.9 | –7.9 | –7.84 ± 0.11 |
| 2 Sm3+ + 2 H2O ⇌ Sm2(OH)24+ + 2 H+ | –14.75 ± 0.20 | ||
| 3 Sm3+ + 5 H2O ⇌ Sm3(OH)54+ + 5 H+ | –33.9 ± 0.3 | ||
| Sm(OH)3(s) + 3H+ ⇌ Sm3+ + 3H2O | 16.5 | 17.19 ± 0.30 | |
| Sm(OH)3(s) ⇌ Sm3+ + 3 OH- | –23.9 ± 0.9 (am) –25.9 (cr) |
Scandium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [115] | Brown and Ekberg, 2016 [116] |
|---|---|---|
| Sc3+ + H2O ⇌ ScOH2+ + H+ | –4.3 | –4.16 ± 0.05 |
| Sc3+ + 2 H2O ⇌ Sc(OH)2+ + 2 H+ | –9.7 | –9.71 ± 0.30 |
| Sc3+ + 3 H2O ⇌ Sc(OH)3 + 3 H+ | –16.1 | –16.08 ± 0.30 |
| Sc3+ + 4 H2O ⇌ Sc(OH)4–+ 4 H+ | –26 | –26.7 ± 0.3 |
| 2 Sc3+ + 2 H2O ⇌ Sc2(OH)24+ + 2 H+ | –6.0 | –6.02 ± 0.10 |
| 3 Sc3+ + 5 H2O ⇌ Sc3(OH)54+ + 5 H+ | –16.34 | –16.33 ± 0.10 |
| Sc(OH)3(s) + 3 H+ ⇌ Sc3+ + 3 H2O | 9.17 ± 0.30 | |
| ScO1.5(s) + 3 H+ ⇌ Sc3+ + 1.5 H2O | 5.53 ± 0.30 | |
| ScO(OH)(c) + 3 H+ ⇌ Sc3+ + 2 H2O | 9.4 | |
| Sc(OH)3(c) + OH– ⇌ Sc(OH)4 | –3.5 ± 0.2 |
Selenium(–II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Olin et al., 2015 [117] | Thoenen et al., 2014 [96] |
|---|---|---|
| H2Se(g) ⇌ H2Se(aq) | –1.10 ± 0.01 | –1.10 ± 0.01 |
| H2Se ⇌ HSe– + H+ | –3.85 ± 0.05 | –3.85 ± 0.05 |
| HSe– ⇌ Se2– + H+ | –14.91 ± 0.20 |
Selenium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [118] | Olin et al., 2005 [117] | Thoenen et al., 2014 [96] |
|---|---|---|---|
| SeO32– + H+ ⇌ HSeO3– | 8.50 | 8.36 ± 0.23 | 8.36 ± 0.23 |
| HSeO3– + H+ ⇌ H2SeO3 | 2.75 | 2.64 ± 0.14 | 2.64 ± 0.14 |
Selenium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [119] | Olin et al., 2005 [117] | Thoenen et al., 2014 [96] |
|---|---|---|---|
| SeO42‒ + H+ ⇌ HSeO4‒ | 1.360 | 1.75 ± 0.10 | 1.75 ± 0.10 |
Silicon
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [120] | Thoenen et al., 2014 [96] |
|---|---|---|
| Si(OH)4 ⇌ SiO(OH)3– + H+ | –9.86 | –9.81 ± 0.02 |
| Si(OH)4 ⇌ SiO2(OH)22– + 2 H+ | –22.92 | –23.14 ± 0.09 |
| 4 Si(OH)4 ⇌ Si4O6(OH)64– + 2 H+ + 4 H2O | –13.44 | |
| 4 Si(OH)4 ⇌ Si4O8(OH)44– + 4 H+ + 4 H2O | –35.80 | –36.3 ± 0.2 |
| SiO2(quartz) + 2 H2O ⇌ Si(OH)4 | –4.0 | –3.739 ± 0.087 |
| SiO2(am) + 2 H2O ⇌ Si(OH)4 | –2.714 |
Silver
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [121] | Brown and Ekberg, 2016 [122] |
|---|---|---|
| Ag+ + H2O ⇌ AgOH + H+ | −12.0 | −11.75 ± 0.14 |
| Ag+ + 2 H2O ⇌ Ag(OH)2− + 2 H+ | −24.0 | −24.34 ± 0.14 |
| 0.5 Ag2O(am) + H+ ⇌ Ag+ + 0.5 H2O | 6.29 | 6.27 ± 0.05 |
Sodium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [72] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [123] |
|---|---|---|---|
| Na+ + H2O ⇌ NaOH + H+ | –14.18 | –14.18 | –14.4 ± 0.2 |
Strontium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [16] | Nordstrom et al., 1990 [17] | Brown and Ekberg, 2016 [124] |
|---|---|---|---|
| Sr2+ + H2O ⇌ SrOH+ + H+ | –13.29 | –13.29 | –13.15 ± 0.05 |
Tantalum
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [125] | Filella and May, 2019a [126] |
|---|---|---|
| Ta(OH)5 + H+ ⇌ Ta(OH)4+ + H2O | ~1 | 0.7007 |
| Ta(OH)5 + H2O ⇌ Ta(OH)6– + H+ | ~ –9.6 | |
| Ta6O198– + H+ ⇌ HTa6O197– | 16.35 | |
| HTa6O197– + H+ ⇌ H2Ta6O196– | 14.00 | |
| 1/2 Ta2O5(act) + 5/2 H2O ⇌ Ta(OH)5 | ~ –5.2 | |
| Ta(OH)5(s) ⇌ Ta(OH)5 | –5.295 | |
| Ta2O5(s) + 5 H2O ⇌ 2 Ta(OH)5 | –20.00 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Tellurium(-II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Filella and May, 2019a [127] |
|---|---|
| Te2‒ + H+ ⇌ HTe‒ | 11.81 |
| HTe‒ + H+ ⇌ H2Te | 2.476 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Tellurium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [128] | Filella and May, 2019a [127] |
|---|---|---|
| TeO32‒ + H+ ⇌ HTeO3‒ | 9.928 | |
| HTeO3‒ + H+ ⇌ H2TeO3 | 6.445 | |
| H2TeO3 ⇌ HTeO3‒ + H+ | ‒2.68 | |
| H2TeO3 ⇌ TeO32‒ + 2 H+ | ‒12.5 | |
| H2TeO3 + H+ ⇌ Te(OH)3+ | 3.13 | 2.415 |
| TeO2(s) + H2O ⇌ H2TeO3 | ‒4.709 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Tellurium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [128] | Filella and May, 2019a [127] |
|---|---|---|
| TeO2(OH)42‒ + H+ ⇌ TeO(OH)5‒ | 10.83 | |
| TeO(OH)5‒ + H+ ⇌ Te(OH)6 | 7.68 | 7.696 |
| TeO2(OH)42‒ + 2 H+ ⇌ Te(OH)6 | 18.68 | |
| TeO3(OH)33‒ + 3 H+ ⇌ Te(OH)6 | 34.3 | |
| 2 Te(OH)6 ⇌ Te2O(OH)11‒ + H+ | ‒6.929 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Terbium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [129] |
|---|---|---|
| Tb3+ + H2O ⇌ TbOH2+ + H+ | −7.9 | −7.60 ± 0.09 |
| 2 Tb3+ + 2 H2O ⇌ Tb2(OH)24+ + 2 H+ | −13.9 ± 0.2 | |
| 3 Tb3+ + 5 H2O ⇌ Tb3(OH)54+ + 5 H+ | −31.7 ± 0.3 | |
| Tb(OH)3(s) + 3 H+ ⇌ Tb3+ + 3 H2O | 16.5 | 16.33 ± 0.30 |
Thallium(I)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [130] | Brown and Ekberg, 2016 [131] |
|---|---|---|
| Tl+ + H2O ⇌ TlOH + H+ | –13.21 | |
| Tl+ + OH– ⇌ TlOH | 0.64 ± 0.05 | |
| Tl+ + 2 OH– ⇌ Tl(OH)2– | –0.7 ± 0.7 | |
| ½ Tl2O(s) + H+ ⇌ Tl+ + ½ H2O | 13.55 ± 0.20 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Thallium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [130] | Brown and Ekberg, 2016 [131] |
|---|---|---|
| Tl3+ + H2O ⇌ TlOH2+ + H+ | –0.62 | –0.22 ± 0.19 |
| Tl3+ + 2 H2O ⇌ Tl(OH)2+ + 2 H+ | –1.57 | |
| Tl3+ + 3 H2O ⇌ Tl(OH)3 + 3 H+ | –3.3 | |
| Tl3+ + 4 H2O ⇌ Tl(OH)4– + 4 H+ | –15.0 | |
| ½ Tl2O3(s) + 3 H+ ⇌ Tl3+ + ³⁄₂ H2O | –3.90 | –3.90 ± 0.10 |
(a) The number of significant figures are retained to minimise propagation of round-off errors; they should not be taken to indicate the relative uncertainty of the values, which is always at least one order of magnitude less than indicated.
Thorium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [132] | Rand et al., 2008 [133] | Thoenen et al, 014 [134] | Brown and Ekberg, 2016 [135] |
|---|---|---|---|---|
| Th4+ + H2O ⇌ ThOH3+ + H+ | –3.20 | –2.5 ± 0.5 | –2.5 ± 0.5 | –2.5 ± 0.5 |
| Th4+ + 2 H2O ⇌ Th(OH)22+ + 2 H+ | –6.93 | –6.2 ± 0.5 | –6.2 ± 0.5 | –6.2 ± 0.5 |
| Th4+ + 3 H2O ⇌ Th(OH)3+ + 3 H+ | < –11.7 | |||
| Th4+ + 4 H2O ⇌ Th(OH)4 + 4 H+ | –15.9 | –17.4 ± 0.7 | –17.4 ± 0.7 | –17.4 ± 0.7 |
| 2Th4+ + 2 H2O ⇌ Th2(OH)26+ + 2 H+ | –6.14 | –5.9 ± 0.5 | –5.9 ± 0.5 | –5.9 ± 0.5 |
| 2Th4+ + 3 H2O ⇌ Th2(OH)35+ + 3 H+ | –6.8 ± 0.2 | –6.8 ± 0.2 | –6.8 ± 0.2 | |
| 4Th4+ + 8 H2O ⇌ Th4(OH)88+ + 8 H+ | –21.1 | –20.4 ± 0.4 | –20.4 ± 0.4 | –20.4 ± 0.4 |
| 4Th4+ + 12 H2O ⇌ Th4(OH)124+ + 12 H+ | –26.6 ± 0.2 | –26.6 ± 0.2 | –26.6 ± 0.2 | |
| 6Th4+ + 15 H2O(l) ⇌ Th6(OH)159+ + 15 H+ | –36.76 | –36.8 ± 1.5 | –36.8 ± 1.5 | –36.8 ± 1.5 |
| 6Th4+ + 14 H2O(l) ⇌ Th6(OH)1410+ + 14 H+ | –36.8 ± 1.2 | –36.8 ± 1.2 | –36.8 ± 1.2 | |
| ThO2(c) + 4 H+ ⇌ Th4+ + 2 H2O | 6.3 | |||
| ThO2(am) + 4 H+ ⇌ Th4+ + 2 H2O | 8.8 ± 1.0 | |||
| ThO2(am,hyd,fresh) + 4 H+ ⇌ Th4+ + 2 H2O | 9.3 ± 0.9 | |||
| ThO2(am,hyd,aged) + 4 H+ ⇌ Th4+ + 2 H2O | 8.5 ± 0.9 | |||
| Th4+ + 4 OH- ⇌ ThO2(am,hyd,fresh) + 2 H2O | 46.7 ± 0.9 | |||
| Th4+ + 4 OH- ⇌ ThO2(am,hyd,aged) + 2 H2O | 47.5 ± 0.9 |
Thulium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [136] |
|---|---|---|
| Tm3+ + H2O ⇌ TmOH2+ + H+ | −7.7 | −7.34 ± 0.09 |
| 2 Tm3+ + 2 H2O ⇌ Tm2(OH)24+ + 2 H+ | −13.2 ± 0.2 | |
| 3 Tm3+ + 5 H2O ⇌ Tm3(OH)54+ + 5 H+ | −30.5 ± 0.3 | |
| Tm(OH)3(s) + 3 H+ ⇌ Tm3+ + 3 H2O | 15.0 | 15.56 ± 0.40 |
Tin(II)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Feitknecht, 1963 [71] | Baes and Mesmer, 1976 [137] | Hummel et al., 2002 [45] | NIST46 [4] | Cigala et al, 2012 [138] | Gamsjäger et al, 2012 [139] | Brown and Ekberg, 2016 [140] |
|---|---|---|---|---|---|---|---|
| Sn2+ + H2O ⇌ SnOH+ + H+ | –3.40 | –3.8 ± 0.2 | –3.4 | –3.52 ± 0.05 | –3.53 ± 0.40 | –3.53 ± 0.40 | |
| Sn2+ + 2 H2O ⇌ Sn(OH)2 + 2 H+ | –7.06 | –7.7 ± 0.2 | –7.1 | –6.26 ± 0.06 | –7.68 ± 0.40 | –7.68 ± 0.40 | |
| Sn2+ + 3 H2O ⇌ Sn(OH)3– + 3 H+ | –16.61 | –17.5 ± 0.2 | –16.6 | –16.97 ± 0.17 | –17.00 ± 0.60 | –17.56 ± 0.40 | |
| 2 Sn2+ + 2 H2O ⇌ Sn2(OH)22+ + 2 H+ | –4.77 | –4.8 | –4.79 ± 0.05 | ||||
| 3 Sn2+ + 4 H2O ⇌ Sn3(OH)42+ + 4 H+ | –6.88 | –5.6 ± 1.6 | –6.88 | –5.88 ± 0.05 | –5.60 ± 0.47 | −5.60 ± 0.47 | |
| Sn(OH)2(s) ⇌ Sn2+ + 2 OH– | –25.8 | –26.28 ± 0.08 | |||||
| SnO(s) + 2 H+ ⇌ Sn2+ + H2O | 1.76 | 2.5± 0.5 | 1.60 ± 0.15 | ||||
| SnO(s) + H2O ⇌ Sn2+ + 2 OH– | –26.2 | ||||||
| SnO(s) + H2O ⇌ Sn(OH)2 | –5.3 | ||||||
| SnO(s) + 2 H2O ⇌ Sn(OH)3– + H+ | –0.9 |
Tin(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Hummel et al., 2002 [45] | Gamsjäger et al, 2012 [139] | Brown and Ekberg, 2016 [140] |
|---|---|---|---|
| Sn4+ + 4 H2O ⇌ Sn(OH)4 + 4 H+ | 7.53 ± 0.12 | ||
| Sn4+ + 5 H2O ⇌ Sn(OH)5– + 5 H+ | –1.07 ± 0.42 | ||
| Sn4+ + 6 H2O ⇌ Sn(OH)62– + 6 H+ | –1.07 ± 0.42 | ||
| Sn(OH)4 + H2O ⇌ Sn(OH)5– + H+ | –8.0 ± 0.3 | –8.60 ± 0.40 | |
| Sn(OH)4 + 2 H2O ⇌ Sn(OH)62– + 2 H+ | –18.4 ± 0.3 | –18.67 ± 0.30 | |
| SnO2(cr) + 2 H2O ⇌ Sn(OH)4 | –8.0 ± 0.2 | –8.06 ± 0.11 | |
| SnO2(am) + 2 H2O ⇌ Sn(OH)4 | –7.3 ± 0.3 | –7.22 ± 0.08 | |
| SnO2(s) + 4 H+ ⇌ Sn4+ + 2 H2O | –15.59 ± 0.04 |
Tungsten
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | NIST46 [4] |
|---|---|
| WO42– + H+ ⇌ HWO4– | 3.6 |
| WO42– + 2 H+ ⇌ H2WO4 | 5.8 |
| 6 WO42– + 7 H+ ⇌ HW6O215– + 3 H2O | 63.83 |
Titanium(III)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Perrin et al., 1969 [141] | Baes and Mesmer, 1976 [142] | Brown and Ekberg, 2016 [143] |
|---|---|---|---|
| Ti3+ + H2O ⇌ TiOH2+ + H+ | –1.29 | –2.2 | –1.65 ± 0.11 |
| 2 Ti3+ + 2 H2O ⇌ Ti2(OH)24+ + 2 H+ | –3.6 | –2.64 ± 0.10 |
Titanium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [142] | Brown and Ekberg, 2016 [143] |
|---|---|---|
| Ti(OH)22+ + H2O ⇌ Ti(OH)3+ + H+ | ⩽–2.3 | |
| Ti(OH)22+ + 2 H2O ⇌ Ti(OH)4 + 2 H+ | –4.8 | |
| TiO2+ + H2O ⇌ TiOOH+ + H+ | –2.48 ± 0.10 | |
| TiO2+ + 2 H2O ⇌ TiO(OH)2 + 2 H+ | –5.49 ± 0.14 | |
| TiO2+ + 3 H2O ⇌ TiO(OH)3– + 3 H+ | –17.4 ± 0.5 | |
| TiO(OH)2 + H2O ⇌ TiO(OH)3– + H+ | –11.9 ±0.5 | |
| TiO2(c) +2 H2O ⇌ Ti(OH)4 | ~ –4.8 | |
| TiO2(s) + H+ ⇌ TiOOH+ | –6.06 ± 0.30 | |
| TiO2(s) + H2O ⇌ TiO(OH)2 | –9.02 ± 0.02 | |
| TiO2 x H2O ⇌ Ti(OH)22+[OH–] | ||
| TiO2(s) + 4 H+ ⇌ Ti4+ + 2 H2O | –3.56 ± 0.10 |
Uranium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [144] | Thoenen et al., 2014 [145] | Brown and Ekberg, 2016 [146] | Grenthe et al., 2020 [6] |
|---|---|---|---|---|
| U4+ + H2O ⇌ UOH3+ + H+ | –0.65 | – 0.54 ± 0.06 | –0.58 ± 0.08 | – 0.54 ± 0.06 |
| U4+ + 2 H2O ⇌ U(OH)22+ + 2 H+ | (–2.6) | –1.1 ± 1.0 | –1.4 ± 0.2 | –1.9 ± 0.2 |
| U4+ + 3 H2O ⇌ U(OH)3+ + 3 H+ | (–5.8) | –4.7 ± 1.0 | –5.1 ± 0.3 | –5.2 ± 0.4 |
| U4+ + 4 H2O ⇌ U(OH)4 + 4 H+ | (–10.3) | –10.0 ± 1.4 | –10.4 ± 0.5 | –10.0 ± 1.4 |
| U4+ + 5 H2O ⇌ U(OH)5- + 5 H+ | –16.0 | |||
| UO2(am, hyd) + 4 H+ ⇌ U4+ + 2 H2O | 1.5 ± 1.0 | |||
| UO2(am,hyd) + 2 H2O ⇌ U4+ + 4 OH– | –54.500 ± 1.000 | –54.500 ± 1.000 | ||
| UO2(c) + 4 H+ ⇌ U4+ + 2 H2O | –1.8 | |||
| UO2(c) + 2 H2O ⇌ U4+ + 4 OH– | –60.860 ± 1.000 |
Uranium(VI)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [147] | Grenthe et al., 1992 [148] | NIST46 [4] | Brown and Ekberg, 2016 [149] | Grenthe et al., 2020 [6] |
|---|---|---|---|---|---|
| UO22+ + H2O ⇌ UO2(OH)+ + H+ | –5.8 | –5.2 ± 0.3 | –5.9 ± 0.1 | –5.13 ± 0.04 | –5.25 ± 0.24 |
| UO22+ + 2 H2O ⇌ UO2(OH)2 + 2 H+ | ≤-10.3 | –12.15 ± 0.20 | –12.15 ± 0.07 | ||
| UO22+ + 3 H2O ⇌ UO2(OH)3– + 3 H+ | –19.2 ± 0.4 | –20.25 ± 0.42 | –20.25 ± 0.42 | ||
| UO22+ + 4 H2O ⇌ UO2(OH)42– + 4 H+ | –33 ± 2 | –32.40 ± 0.68 | –32.40 ± 0.68 | ||
| 2 UO22+ + 2 H2O ⇌ (UO2)2(OH)22+ + 2 H+ | –5.62 | –5.62 ± 0.04 | –5.58 ± 0.04 | –5.68 ± 0.05 | –5.62 ± 0.08 |
| 3 UO22+ + 5 H2O ⇌ (UO2)3(OH)5+ + 5 H+ | –15.63 | –15.55 ± 0.12 | –15.6 | –15.75 ± 0.12 | –15.55 ± 0.12 |
| 3 UO22+ + 4 H2O ⇌ (UO2)3(OH)42+ + 4 H+ | (–11.75) | –11.9 ± 0.3 | –11.78 ± 0.05 | –11.9 ± 0.3 | |
| 3 UO22+ + 7 H2O ⇌ (UO2)3(OH)7– + 7 H+ | –31 ± 2.0 | –32.2 ± 0.8 | –32.2 ± 0.8 | ||
| 4 UO22+ + 7 H2O ⇌ (UO2)4(OH)7+ + 7 H+ | –21.9 ± 1.0 | –22.1 ± 0.2 | –21.9 ± 1.0 | ||
| 2 UO22+ + H2O ⇌ (UO2)2(OH)3+ + H+ | –2.7 ± 1.0 | –2.7 ± 1.0 | |||
| UO2(OH)2(s) + 2H+ ⇌ UO22+ + 2 H2O | 5.6 | 6.0 | 4.81 ± 0.20 | ||
| UO3·2H2O(cr) + 2H+ ⇌ UO22+ + 3 H2O | 5.350 ± 0.130 |
Vanadium(IV)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Brown and Ekberg, 2016 [79] |
|---|---|
| VO2+ + H2O ⇌ VO(OH)+ + H+ | –5.30 ± 0.13 |
| 2 VO2+ + 2 H2O ⇌ (VO)2(OH)22+ + 2 H+ | –6.71 ± 0.10 |
Vanadium(V)
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [150] | Brown and Ekberg, 2016 [151] |
|---|---|---|
| VO2+ + 2 H2O ⇌ VO(OH)3 + H+ | –3.3 | |
| VO2+ + 2 H2O ⇌ VO2(OH)2– + 2 H+ | –7.3 | –7.18 ± 0.12 |
| 10 VO2+ + 8 H2O ⇌ V10O26(OH)24– + 14 H+ | –10.7 | |
| VO2(OH)2– ⇌ VO3(OH)2– + H+ | –8.55 | |
| 2 VO2(OH)2– ⇌ V2O6(OH)23– + H+ + H2O | –6.53 | |
| VO3(OH)2– ⇌ VO43– + H+ | –14.26 | |
| 2 VO3(OH)2– ⇌ V2O74– + H2O | 0.56 | |
| 3 VO3(OH)2– + 3 H+⇌ V3O93– + 3 H2O | 31.81 | |
| V10O26(OH)24– ⇌ V10O27(OH)5– + 3 H+ | –3.6 | |
| V10O27(OH)5– ⇌ V10O286– + H+ | –6.15 | |
| VO2+ + H2O ⇌ VO2OH + H+ | –3.25 ± 0.1 | |
| VO2+ + 3 H2O ⇌ VO2(OH)32- + 3 H+ | –15.74 ± 0.19 | |
| VO2+ + 4 H2O ⇌ VO2(OH)43- + 4 H+ | –30.03 ± 0.24 | |
| 2 VO2+ + 4 H2O ⇌ (VO2)2(OH)42- + 4 H+ | –11.66 ± 0.53 | |
| 2 VO2+ + 5 H2O ⇌ (VO2)2(OH)53- + 5 H+ | –20.91 ± 0.22 | |
| 2 VO2+ + 6 H2O ⇌ (VO2)2(OH)64- + 6 H+ | –32.43 ± 0.30 | |
| 4 VO2+ + 8 H2O ⇌ (VO2)4(OH)84- + 8 H+ | –20.78 ± 0.33 | |
| 4 VO2+ + 9 H2O ⇌ (VO2)4(OH)95- + 9 H+ | –31.85 ± 0.26 | |
| 4 VO2+ + 10 H2O ⇌ (VO2)4(OH)106- + 10 H+ | –45.85 ± 0.26 | |
| 5 VO2+ + 10 H2O ⇌ (VO2)5(OH)105- + 10 H+ | –27.02 ± 0.34 | |
| 10 VO2+ + 14 H2O ⇌ (VO2)10(OH)144- + 14 H+ | –10.5 ± 0.3 | |
| 10 VO2+ + 15 H2O ⇌ (VO2)10(OH)155- + 15 H+ | –15.73 ± 0.33 | |
| 10 VO2+ + 16 H2O ⇌ (VO2)10(OH)166- + 16 H+ | –23.90 ± 0.35 | |
| ½ V2O5(c) + H+ ⇌ VO2+ + ½ H2O | –0.66 | |
| V2O5(s) + 2 H+ ⇌ 2 VO2+ + H2O | –0.64 ± 0.09 |
Ytterbium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [28] | Brown and Ekberg, 2016 [152] |
|---|---|---|
| Yb3+ + H2O ⇌ YbOH2+ + H+ | −7.7 | −7.31 ± 0.18 |
| Yb3+ + 2 H2O ⇌ Yb(OH)2+ + 2 H+ | (−15.8) | |
| Yb3+ + 3 H2O ⇌ Yb(OH)3 + 3 H+ | (−24.1) | |
| Yb3+ + 4 H2O ⇌ Yb(OH)4− + 4 H+ | −32.7 | |
| 2 Yb3+ + 2 H2O ⇌ Yb2(OH)24+ + 2 H+ | −13.76 ± 0.20 | |
| 3 Yb3+ + 5 H2O ⇌ Yb3(OH)54+ + 5 H+ | −30.6 ± 0.3 | |
| Yb(OH)3(s) + 3 H+ ⇌ Yb3+ + 3 H2O | 14.7 | 15.35 ± 0.20 |
Yttrium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [46] | Brown and Ekberg, 2016 [66] |
|---|---|---|
| Y3+ + H2O ⇌ YOH2+ + H+ | –7.7 | –7.77 ± 0.06 |
| Y3+ + 2 H2O ⇌ Y(OH)2+ + 2 H+ | (–16.4) [Estimation] | |
| Y3+ + 3 H2O ⇌ Y(OH)3 + 3 H+ | (–26.0) [Estimation] | |
| Y3+ + 4 H2O ⇌ Y(OH)4-+ 4 H+ | –36.5 | |
| 2 Y3+ + 2 H2O ⇌ Y2(OH)24+ + 2 H+ | –14.23 | –14.1 ± 0.2 |
| 3 Y3+ + 5 H2O ⇌ Y3(OH)54+ + 5 H+ | –31.6 | –32.7 ± 0.3 |
| Y(OH)3(s) + 3 H+ ⇌ Y3+ + 3 H2O | 17.5 | 17.32 ± 0.30 |
Zinc
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [153] | Powell and Brown, 2013 [154] | Brown and Ekberg, 2016 [155] |
|---|---|---|---|
| Zn2+ + H2O ⇌ ZnOH+ + H+ | −8.96 | −8.96 ± 0.05 | −8.94 ± 0.06 |
| Zn2+ + 2 H2O ⇌ Zn(OH)2 + 2 H+ | −16.9 | –17.82 ± 0.08 | −17.89 ± 0.15 |
| Zn2+ + 3 H2O ⇌ Zn(OH)3- + 3 H+ | −28.4 | –28.05 ± 0.05 | −27.98 ± 0.10 |
| Zn2+ + 4 H2O ⇌ Zn(OH)42- + 4 H+ | −41.2 | –40.41 ± 0.12 | −40.35 ± 0.22 |
| 2 Zn2+ + H2O ⇌ Zn2OH3+ + H+ | −9.0 | –7.9 ± 0.2 | −7.89 ± 0.31 |
| 2 Zn2+ + 6 H2O ⇌ Zn2(OH)62- + 6 H+ | −57.8 | ||
| ZnO(s) + 2 H+ ⇌ Zn2+ + H2O | 11.14 | 11.12 ± 0.05 | 11.11 ± 0.10 |
| ε-Zn(OH)2(s) + 2 H+ ⇌ Zn2+ + 2 H2O | 11.38 ± 0.20 | 11.38± 0.20 | |
| β1-Zn(OH)2(s) + 2 H+ ⇌ Zn2+ + 2 H2O | 11.72 ± 0.04 | ||
| β2-Zn(OH)2(s) + 2 H+ ⇌ Zn2+ + 2 H2O | 11.76 ± 0.04 | ||
| γ-Zn(OH)2(s) + 2 H+ ⇌ Zn2+ + 2 H2O | 11.70 ± 0.04 | ||
| δ-Zn(OH)2(s) + 2 H+ ⇌ Zn2+ + 2 H2O | 11.81 ± 0.04 |
Zirconium
Hydrolysis constants (log values) in critical compilations at infinite dilution and T = 298.15 K:
| Reaction | Baes and Mesmer, 1976 [55] | Thoenen et al., 2014 [96] | Brown and Ekberg, 2016 [156] |
|---|---|---|---|
| Zr4+ + H2O ⇌ ZrOH3+ + H+ | 0.32 | 0.32 ± 0.22 | 0.12 ± 0.12 |
| Zr4+ + 2 H2O ⇌ Zr(OH)22+ + 2 H+ | (−1.7)* | 0.98 ± 1.06* | −0.18 ± 0.17* |
| Zr4+ + 3 H2O ⇌ Zr(OH)3+ + 3 H+ | (−5.1) | ||
| Zr4+ + 4 H2O ⇌ Zr(OH)4 + 4 H+ | –9.7* | –2.19 ± 0.70* | −4.53 ± 0.37* |
| Zr4+ + 5 H2O ⇌ Zr(OH)5– + 5 H+ | –16.0 | ||
| Zr4+ + 6 H2O ⇌ Zr(OH)62– + 6 H+ | –29± 0.70 | –30.5 ± 0.3 | |
| 3 Zr4+ + 4 H2O ⇌ Zr3(OH)48+ + 4 H+ | –0.6 | 0.4 ± 0.3 | 0.90 ± 0.18 |
| 3 Zr4+ + 5 H2O ⇌ Zr3(OH)57+ + 5 H+ | 3.70 | ||
| 3 Zr4+ + 9 H2O ⇌ Zr3(OH)93+ + 9 H+ | 12.19 ± 0.20 | 12.19 ± 0.20 | |
| 4 Zr4+ + 8 H2O ⇌ Zr4(OH)88+ + 8 H+ | 6.0 | 6.52 ± 0.05 | 6.52 ± 0.05 |
| 4 Zr4+ + 15 H2O ⇌ Zr4(OH)15+ + 15 H+ | 12.58± 0.24 | ||
| 4 Zr4+ + 16 H2O ⇌ Zr4(OH)16 + 16 H+ | 8.39± 0.80 | ||
| ZrO2(s) + 4 H+ ⇌ Zr4+ + 2 H2O | –1.9* | –5.37 ± 0.42* | |
| ZrO2(s, baddeleyite) + 4 H+ ⇌ Zr4+ + 2 H2O | –7 ± 1.6 | ||
| ZrO2(am) + 4 H+ ⇌ Zr4+ + 2 H2O | –3.24± 0.10 | –2.97 ± 0.18 |
*Errors in compilations concerning equilibrium and/or data elaboration. Data not recommended. It is strongly suggested to refer to the original papers.