Related Research Articles
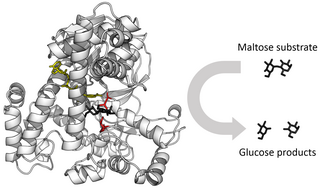
Enzymes are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called enzymology and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties.
The katal is the unit of catalytic activity in the International System of Units (SI) used for quantifying the catalytic activity of enzymes and other catalysts.

In biochemistry, Michaelis–Menten kinetics, named after Leonor Michaelis and Maud Menten, is the simplest case of enzyme kinetics, applied to enzyme-catalysed reactions of one substrate and one product. It takes the form of an equation describing the reaction rate to , the concentration of the substrate A. Its formula is given by the Michaelis–Menten equation:
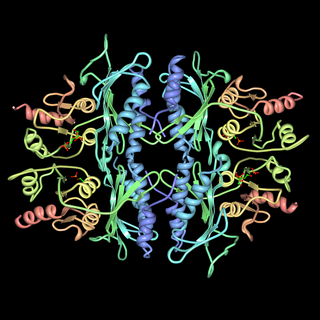
The enzyme fructose bisphosphatase (EC 3.1.3.11; systematic name D-fructose-1,6-bisphosphate 1-phosphohydrolase) catalyses the conversion of fructose-1,6-bisphosphate to fructose 6-phosphate in gluconeogenesis and the Calvin cycle, which are both anabolic pathways:
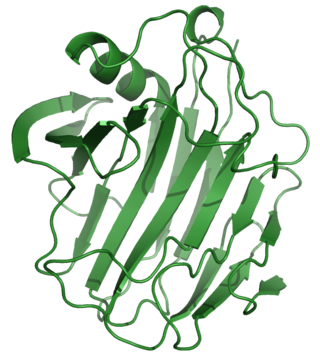
Cellulase is any of several enzymes produced chiefly by fungi, bacteria, and protozoans that catalyze cellulolysis, the decomposition of cellulose and of some related polysaccharides:
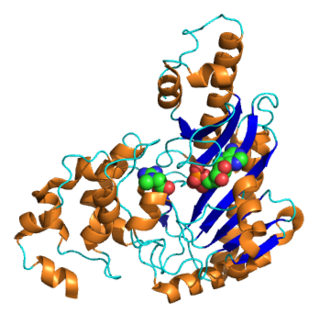
Creatine kinase (CK), also known as creatine phosphokinase (CPK) or phosphocreatine kinase, is an enzyme expressed by various tissues and cell types. CK catalyses the conversion of creatine and uses adenosine triphosphate (ATP) to create phosphocreatine (PCr) and adenosine diphosphate (ADP). This CK enzyme reaction is reversible and thus ATP can be generated from PCr and ADP.

In pharmacology, the international unit (IU) is a unit of measurement for the effect or biological activity of a substance, for the purpose of easier comparison across similar forms of substances. International units are used to quantify vitamins and biologics.
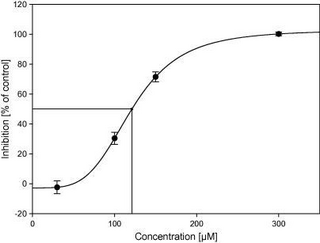
Half maximal inhibitory concentration (IC50) is a measure of the potency of a substance in inhibiting a specific biological or biochemical function. IC50 is a quantitative measure that indicates how much of a particular inhibitory substance (e.g. drug) is needed to inhibit, in vitro, a given biological process or biological component by 50%. The biological component could be an enzyme, cell, cell receptor or microorganism. IC50 values are typically expressed as molar concentration.

Enzyme assays are laboratory methods for measuring enzymatic activity. They are vital for the study of enzyme kinetics and enzyme inhibition.
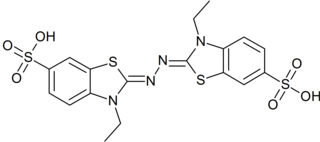
In biochemistry, ABTS is a chemical compound used to observe the reaction kinetics of specific enzymes. A common use for it is in the enzyme-linked immunosorbent assay (ELISA) to detect the binding of molecules to each other.

Enzyme kinetics is the study of the rates of enzyme-catalysed chemical reactions. In enzyme kinetics, the reaction rate is measured and the effects of varying the conditions of the reaction are investigated. Studying an enzyme's kinetics in this way can reveal the catalytic mechanism of this enzyme, its role in metabolism, how its activity is controlled, and how a drug or a modifier might affect the rate.
Uncompetitive inhibition is a type of inhibition in which the apparent values of the Michaelis–Menten parameters and are decreased in the same proportion.
In medicine, the urea-to-creatinine ratio (UCR), known in the United States as BUN-to-creatinine ratio, is the ratio of the blood levels of urea (BUN) (mmol/L) and creatinine (Cr) (μmol/L). BUN only reflects the nitrogen content of urea and urea measurement reflects the whole of the molecule, urea is just over twice BUN. In the United States, both quantities are given in mg/dL The ratio may be used to determine the cause of acute kidney injury or dehydration.
°Lintner or degrees Lintner is a unit used to measure the ability of a malt to reduce starch to sugar, that is, its diastatic power. Degrees Lintner is an intensive unit, not an extensive one; it is independent of the quantity of malt used. While the measurement is applicable to any amylase, in general it refers to the combined α-amylase and β-amylase used in brewing. The term is also generalized to diastatic malt extracts and separately prepared brewing enzymes. The abbreviation °L is official, but in brewing applications it may conflict with °L used for degrees Lovibond.
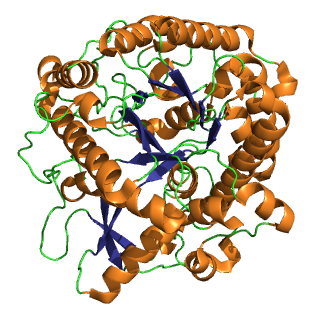
Myrosinase is a family of enzymes involved in plant defense against herbivores, specifically the mustard oil bomb. The three-dimensional structure has been elucidated and is available in the PDB.
Polyphenol oxidase, an enzyme involved in fruit browning, is a tetramer that contains four atoms of copper per molecule.
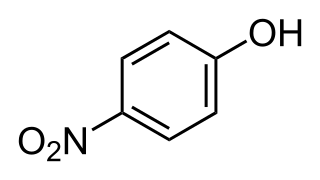
4-Nitrophenol is a phenolic compound that has a nitro group at the opposite position of the hydroxyl group on the benzene ring.
Plasma renin activity (PRA), also known as the renin (active) assay or random plasma renin, is a measure of the activity of the plasma enzyme renin, which plays a major role in the body's regulation of blood pressure, thirst, and urine output. Measure of direct renin concentration (DRC) is technically more demanding, and hence PRA is used instead. DRC assays are still in evolution, and generally a conversion factor of PRA (ng/mL/h) to DRC (mU/L) is 8.2. A recently developed and already commonly used automated DRC assay uses the conversion factor of 12. PRA is sometimes measured, specially in case of certain diseases which present with hypertension or hypotension. PRA is also raised in certain tumors. A PRA measurement may be compared to a plasma aldosterone concentration as an aldosterone-to-renin ratio (ARR).
Κ-casein, or kappa casein, is a mammalian milk protein involved in several important physiological processes. Chymosin splits K-casein into an insoluble peptide and water-soluble glycomacropeptide (GMP). GMP is responsible for an increased efficiency of digestion, prevention of neonate hypersensitivity to ingested proteins, and inhibition of gastric pathogens. The human gene for κ-casein is CSN3.

Extracellular enzymes or exoenzymes are synthesized inside the cell and then secreted outside the cell, where their function is to break down complex macromolecules into smaller units to be taken up by the cell for growth and assimilation. These enzymes degrade complex organic matter such as cellulose and hemicellulose into simple sugars that enzyme-producing organisms use as a source of carbon, energy, and nutrients. Grouped as hydrolases, lyases, oxidoreductases and transferases, these extracellular enzymes control soil enzyme activity through efficient degradation of biopolymers.
References
- ↑ Nomenclature Committee of the International Union of Biochemistry (NC-IUB) (1979). "Units of Enzyme Activity". Eur. J. Biochem. 97 (2): 319–20. doi: 10.1111/j.1432-1033.1979.tb13116.x .
- ↑ "Terminology of bioanalytical methods (IUPAC Recommendations 2018)". Chemistry International. 40 (3): 34. 2018-07-01. doi: 10.1515/ci-2018-0319 . hdl: 10138/237212 . ISSN 1365-2192.
- ↑ Principles of Biochemistry, page 94, 4th Edition, Lehninger
- ↑ Wharton, Christopher W.; Eisenthal, Robert (2013), Molecular Enzymology, Tertiary Level Biology, Springer Science and Business Media, p. 82, ISBN 9781461585329.
- ↑ Bommarius, Andreas S.; Riebel-Bommarius, Bettina R. (2007), Biocatalysis: Fundamentals and Applications, John Wiley and Sons, p. 30, ISBN 9783527606054.