
A phosphodiesterase inhibitor is a drug that blocks one or more of the five subtypes of the enzyme phosphodiesterase (PDE), thereby preventing the inactivation of the intracellular second messengers, cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) by the respective PDE subtype(s). The ubiquitous presence of this enzyme means that non-specific inhibitors have a wide range of actions, the actions in the heart, and lungs being some of the first to find a therapeutic use.
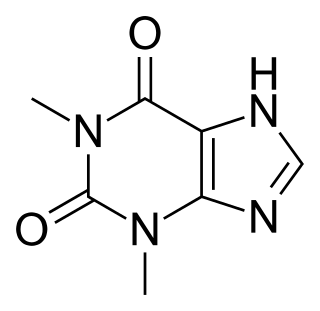
Theophylline, also known as 1,3-dimethylxanthine, is a drug that inhibits phosphodiesterase and blocks adenosine receptors. It is used to treat chronic obstructive pulmonary disease (COPD) and asthma. Its pharmacology is similar to other methylxanthine drugs. Trace amounts of theophylline are naturally present in tea, coffee, chocolate, yerba maté, guarana, and kola nut.

A phosphodiesterase (PDE) is an enzyme that breaks a phosphodiester bond. Usually, phosphodiesterase refers to cyclic nucleotide phosphodiesterases, which have great clinical significance and are described below. However, there are many other families of phosphodiesterases, including phospholipases C and D, autotaxin, sphingomyelin phosphodiesterase, DNases, RNases, and restriction endonucleases, as well as numerous less-well-characterized small-molecule phosphodiesterases.
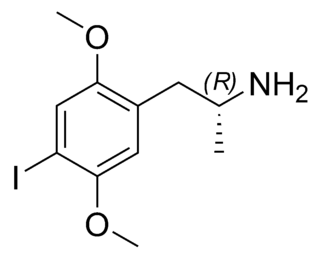
2,5-Dimethoxy-4-iodoamphetamine (DOI) is a psychedelic drug and a substituted amphetamine. Unlike many other substituted amphetamines, however, it is not primarily a stimulant. DOI has a stereocenter and R-(−)-DOI is the more active stereoisomer. In neuroscience research, [125I]-R-(−)-DOI is used as a radioligand and indicator of the presence of 5-HT2A serotonin receptors. DOI's effects have been compared to LSD, although there are differences that experienced users can distinguish. Besides the longer duration, the trip tends to be more energetic than an LSD trip, with more body load and a different subjective visual experience. The after effects include residual stimulation and difficulty sleeping, which, depending on the dose, may persist for days. While rare, it is sometimes sold as a substitute for LSD, or even sold falsely as LSD, which may be dangerous because DOI does not have the same established safety profile as LSD.

Cyclic guanosine monophosphate-specific phosphodiesterase type 5 is an enzyme from the phosphodiesterase class. It is found in various tissues, most prominently the corpus cavernosum of the clitoris and of the penis as well as the retina. It has also been recently discovered to play a vital role in the cardiovascular system.
The Controlled Drugs and Substances Act is Canada's federal drug control statute. Passed in 1996 under Prime Minister Jean Chrétien's government, it repeals the Narcotic Control Act and Parts III and IV of the Food and Drugs Act, and establishes eight Schedules of controlled substances and two Classes of precursors. It provides that "The Governor in Council may, by order, amend any of Schedules I to VIII by adding to them or deleting from them any item or portion of an item, where the Governor in Council deems the amendment to be necessary in the public interest."

Amrinone, also known as inamrinone, and sold as Inocor, is a pyridine phosphodiesterase 3 inhibitor. It is a drug that may improve the prognosis in patients with congestive heart failure. Amrinone has been shown to increase the contractions initiated in the heart by high-gain calcium induced calcium release (CICR). The positive inotropic effect of amrinone is mediated by the selective enhancement of high-gain CICR, which contributes to the contraction of myocytes by phosphorylation through cAMP dependent protein kinase A (PKA) and Ca2+ calmodulin kinase pathways.
Phosphodiesterase 1, PDE1, EC 3.1.4.1, systematic name oligonucleotide 5′-nucleotidohydrolase) is a phosphodiesterase enzyme also known as calcium- and calmodulin-dependent phosphodiesterase. It is one of the 11 families of phosphodiesterase (PDE1-PDE11). Phosphodiesterase 1 has three subtypes, PDE1A, PDE1B and PDE1C which divide further into various isoforms. The various isoforms exhibit different affinities for cAMP and cGMP.

The PDE2 enzyme is one of 21 different phosphodiesterases (PDE) found in mammals. These different PDEs can be subdivided to 11 families. The different PDEs of the same family are functionally related despite the fact that their amino acid sequences show considerable divergence. The PDEs have different substrate specificities. Some are cAMP selective hydrolases, others are cGMP selective hydrolases and the rest can hydrolyse both cAMP and cGMP.

A PDE3 inhibitor is a drug which inhibits the action of the phosphodiesterase enzyme PDE3. They are used for the therapy of acute heart failure and cardiogenic shock.

Ibudilast is an anti-inflammatory drug used mainly in Japan, which acts as a phosphodiesterase inhibitor, inhibiting the PDE4 subtype to the greatest extent, but also showing significant inhibition of other PDE subtypes.

cAMP-specific 3',5'-cyclic phosphodiesterase 4B is an enzyme that in humans is encoded by the PDE4B gene.

Cilomilast is a drug which was developed for the treatment of respiratory disorders such as asthma and chronic obstructive pulmonary disease (COPD). It is orally active and acts as a selective phosphodiesterase-4 inhibitor.
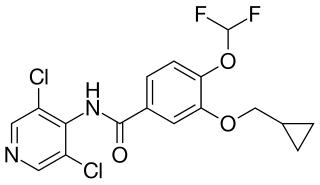
Roflumilast, sold under the brand name Daxas among others, is a medication used for the treatment of chronic obstructive pulmonary disease, plaque psoriasis, seborrheic dermatitis, and atopic dermatitis. It acts as a selective, long-acting inhibitor of the enzyme phosphodiesterase-4 (PDE-4). It has anti-inflammatory effects.

A phosphodiesterase-4 inhibitor, commonly referred to as a PDE4 inhibitor, is a drug used to block the degradative action of phosphodiesterase 4 (PDE4) on cyclic adenosine monophosphate (cAMP). It is a member of the larger family of PDE inhibitors. The PDE4 family of enzymes are the most prevalent PDE in immune cells. They are predominantly responsible for hydrolyzing cAMP within both immune cells and cells in the central nervous system.

Indantadol is a drug which was formerly being investigated as an anticonvulsant and neuroprotective and is now under development for the treatment of neuropathic pain and chronic cough in Europe by Vernalis and Chiesi. It acts as a competitive, reversible, and non-selective monoamine oxidase inhibitor, and as a low affinity, non-competitive NMDA receptor antagonist. A pilot study of indantadol for chronic cough was initiated in October 2009 and in April 2010 it failed to achieve significant efficacy in neuropathic pain in phase IIb clinical trials.
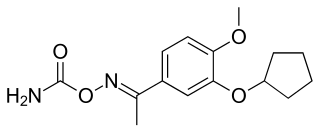
Filaminast was a drug candidate developed by Wyeth-Ayerst. It is a phosphodiesterase 4 inhibitor and an analog of rolipram, which served as a prototype molecule for several development efforts. It was discontinued after a Phase II trial showed that its therapeutic window was too narrow; it could not be dosed high enough without causing significant side effects, which was a problem with the rolipram class of molecules.
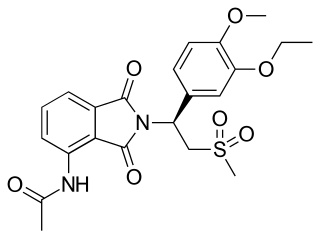
Apremilast, sold under the brand name Otezla among others, is a medication for the treatment of certain types of psoriasis and psoriatic arthritis. The drug acts as a selective inhibitor of the enzyme phosphodiesterase 4 (PDE4). It is taken by mouth.
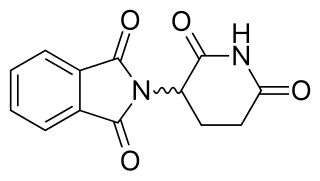
Cereblon E3 ligase modulators, also known as immunomodulatory imide drugs (IMiDs), are a class of immunomodulatory drugs containing an imide group. The IMiD class includes thalidomide and its analogues. These drugs may also be referred to as 'Cereblon modulators'. Cereblon (CRBN) is the protein targeted by this class of drugs.
Benjamin Weiss is an American neuropharmacologist, Emeritus Professor of Pharmacology and Physiology at Drexel University College of Medicine. He is best known for his work with cyclic nucleotide phosphodiesterases. He was the first to propose, based on his experimental work, that selective inhibition of phosphodiesterases which are expressed differentially in all tissues, could be used as a target for drug development. His work is the basis for many marketed and developmental human drugs that selectively inhibit cyclic nucleotide phosphodiesterases.
















