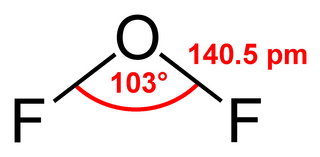
Oxygen difluoride is a chemical compound with the formula OF2. As predicted by VSEPR theory, the molecule adopts a bent molecular geometry. It is a strong oxidizer and has attracted attention in rocketry for this reason. With a boiling point of −144.75 °C, OF2 is the most volatile (isolable) triatomic compound. The compound is one of many known oxygen fluorides.
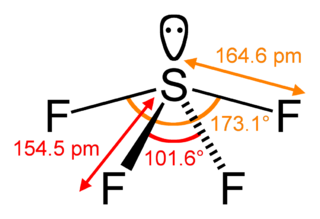
Sulfur tetrafluoride is the chemical compound with the formula SF4. It is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture. Despite these unwelcome characteristics, this compound is a useful reagent for the preparation of organofluorine compounds, some of which are important in the pharmaceutical and specialty chemical industries.
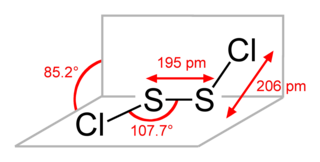
Disulfur dichloride is the inorganic compound of sulfur and chlorine with the formula S2Cl2. It is an amber oily liquid.
Oleg Nikolayevich Trubachyov was a Soviet and Russian linguist. A researcher of the etymology of Slavic languages and Slavic onomastics, specialist in historical linguistics and lexicographer. He was a Doctor of Sciences in Philological Sciences, an academician of the Russian Academy of Sciences and served as the editor-in-chief of the Etimologiya yearbook. His works are on the etymology of Slavic languages and on East Slavic onomastics.

Thiazyl fluoride, NSF, is a colourless, pungent gas at room temperature and condenses to a pale yellow liquid at 0.4 °C. Along with thiazyl trifluoride, NSF3, it is an important precursor to sulfur-nitrogen-fluorine compounds. It is notable for its extreme hygroscopicity.
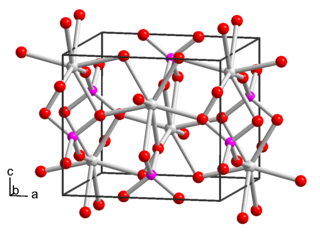
Yttrium phosphate, YPO4, is the phosphate salt of yttrium. It occurs in nature as minerals xenotime and weinschenkite.
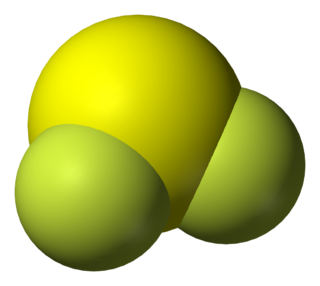
Sulfur difluoride is an inorganic compound with the chemical formula SF2. It can be generated by the reaction of sulfur dichloride and potassium fluoride or mercury(II) fluoride at low pressures:

Thiophosphoryl fluoride is an inorganic molecular gas with formula PSF3 containing phosphorus, sulfur and fluorine. It spontaneously ignites in air and burns with a cool flame. The discoverers were able to have flames around their hands without discomfort, and called it "probably one of the coldest flames known". The gas was discovered in 1888.

Bezbozhnik was an illustrated magazine, an organ of the Centre Soviet and Moscow Oblast Soviet of the League of the Militant Godless.
Nikolai Vasilyevich Rumyantsev was a Soviet author of several polemics on the history of Christianity, translator. He was also a member of the Soviet circle "Atheist" and the League of Militant Atheists. In the 1920s Rumyantsev was one of the leading proponents of the so-called mythological school in the Soviet academia. He authored such publications, as "The Pre-Christian Christ", "The Pagan Christs – the Ancient Precursors of Christianity", "The Apocalypse – The Revelation of John" and "Orthodox Holidays, Their Origin and Class Essence" (1936). "The Apocalypse – The Revelation of John" was published under the influence of Abram Ranovich. In that publication Rumyantsev, while not explicitly retracting his earlier opinions, offered a study of early Christianity which stressed not its derivation from supposed ancient mythology, but its intrinsic relation to revolutionary, messianic elements in contemporary Judaism.
The Türkmenbaşy Gulf or Türkmenbaşy Aýlagy or Türkmenbaşy Bay is a bay of the Caspian Sea in the coast of Turkmenistan.
Atheistic Dictionary is a one-volume reference work devoted to various aspects of religion and atheism. It contains more than 2,500 terms.
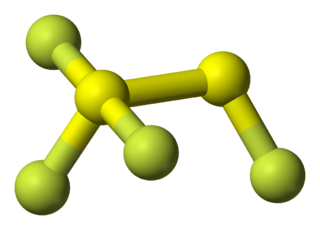
1,1,1,2-tetrafluorodisulfane, also known as 1,2-difluorodisulfane 1,1-difluoride or just difluorodisulfanedifluoride (FSSF3) is an unstable molecular compound of fluorine and sulfur. The molecule has a pair of sulfur atoms, with one fluorine atom on one sulfur, and three fluorine atoms on the other. It has the uncommon property that all the bond lengths are different. The bond strength is not correlated with bond length but is inversely correlated with the force constant (Badger's rule). The molecule can be considered as sulfur tetrafluoride in which a sulfur atom is inserted into a S-F bond.

Derevenskiy Bezbozhnik was an illustrated magazine, an organ of the Centre Soviet and Moscow Oblast Soviet of the League of the Militant Godless.
Cobalt(II) hydride is an inorganic compound with a chemical formula CoH2. It has dark grey crystals. It oxidizes slowly in air and reacts with water.
Praseodymium(III) fluoride is an inorganic compound with the formula PrF3, being the most stable fluoride of praseodymium.
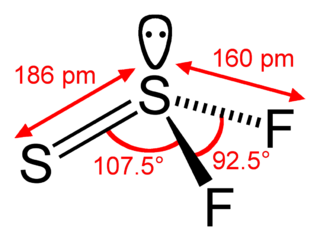
Thiothionyl fluoride is a chemical compound of fluorine and sulfur, with the chemical formula S=SF2. It is an isomer of disulfur difluoride (difluorodisulfane) F−S−S−F.

Ytterbium(III) acetate is an inorganic salt of ytterbium and acetic acid, with a chemical formula of Yb(CH3COO)3. It has colorless crystals that are soluble in water and can form hydrates.

Gadolinium acetate is the acetate salt of the lanthanide element gadolinium, with the chemical formula Gd(CH3COO)3. It is a colorless crystal that is soluble in water and can form a hydrate. Its tetrahydrate has ground state ferromagnetism.
Praseodymium bromide is an inorganic compound with the chemical formula Pr(BrO3)3. It is soluble in water and can form the dihydrate, tetrahydrate and nonahydrate. The nonahydrate melts in its own crystal water at 56.5 °C and completely loses its crystal water at 130 °C. It can be produced by the reaction of barium bromate and praseodymium sulfate.















