
Tarragon, also known as estragon, is a species of perennial herb in the family Asteraceae. It is widespread in the wild across much of Eurasia and North America and is cultivated for culinary and medicinal purposes.

Anise, also called aniseed or rarely anix, is a flowering plant in the family Apiaceae native to the eastern Mediterranean region and Southwest Asia.

Caraway, also known as meridian fennel and Persian cumin, is a biennial plant in the family Apiaceae, native to western Asia, Europe, and North Africa.

Fennel is a flowering plant species in the carrot family. It is a hardy, perennial herb with yellow flowers and feathery leaves. It is indigenous to the shores of the Mediterranean but has become widely naturalized in many parts of the world, especially on dry soils near the sea coast and on riverbanks.

Illicium verum is a medium-sized evergreen tree native to northeast Vietnam and South China. It is a spice that closely resembles anise in flavor and is obtained from the star-shaped pericarps of the fruit of I. verum which are harvested just before ripening. Star anise oil is a highly fragrant oil used in cooking, perfumery, soaps, toothpastes, mouthwashes, and skin creams. Until 2012, when they switched to using genetically modified E. coli, Roche Pharmaceuticals used up to 90% of the world's annual star anise crop to produce oseltamivir (Tamiflu) via shikimic acid.
Ouzo is a dry anise-flavored aperitif that is widely consumed in Greece. It is made from rectified spirits that have undergone a process of distillation and flavoring. Its taste is similar to other anise liquors like pastis, sambuca, mastika, rakı and arak.
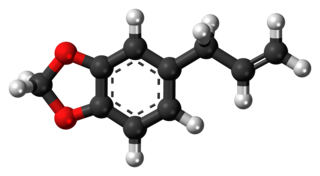
Safrole is an organic compound with the formula CH2O2C6H3CH2CH=CH2. It is a colorless oily liquid, although impure samples can appear yellow. A member of the phenylpropanoid family of natural products, it is found in sassafras plants, among others. Small amounts are found in a wide variety of plants, where it functions as a natural antifeedant. Ocotea pretiosa, which grows in Brazil, and Sassafras albidum, which grows in eastern North America, are the main natural sources of safrole. It has a characteristic "sweet-shop" aroma.
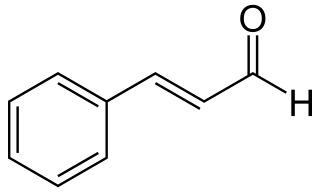
Cinnamaldehyde is an organic compound with the formula or C₆H₅CH=CHCHO. Occurring naturally as predominantly the trans (E) isomer, it gives cinnamon its flavor and odor. It is a phenylpropanoid that is naturally synthesized by the shikimate pathway. This pale yellow, viscous liquid occurs in the bark of cinnamon trees and other species of the genus Cinnamomum. The essential oil of cinnamon bark is about 90% cinnamaldehyde. Cinnamaldehyde decomposes to styrene because of oxidation as a result of bad storage or transport conditions. Styrene especially forms in high humidity and high temperatures. This is the reason why cinnamon contains small amounts of styrene.

Thymol, C10H14O, is a natural monoterpenoid phenol derivative of p-Cymene, isomeric with carvacrol, found in oil of thyme, and extracted from Thymus vulgaris, ajwain, and various other plants as a white crystalline substance of a pleasant aromatic odor and strong antiseptic properties. Thymol also provides the distinctive, strong flavor of the culinary herb thyme, also produced from T. vulgaris. Thymol is only slightly soluble in water at neutral pH, but it is extremely soluble in alcohols and other organic solvents. It is also soluble in strongly alkaline aqueous solutions due to deprotonation of the phenol. Its dissociation constant (pKa) is 10.59±0.10. Thymol absorbs maximum UV radiation at 274 nm.

Linalool refers to two enantiomers of a naturally occurring terpene alcohol found in many flowers and spice plants. Linalool has multiple commercial applications, the majority of which are based on its pleasant scent. A colorless oil, linalool is classified as an acyclic monoterpenoid. In plants, it is a metabolite, a volatile oil component, an antimicrobial agent, and an aroma compound. Linalool has uses in manufacturing of soaps, fragrances, food additives as flavors, household products, and insecticides. Esters of linalool are referred to as linalyl, e.g. linalyl pyrophosphate, an isomer of geranyl pyrophosphate.

Carvone is a member of a family of chemicals called terpenoids. Carvone is found naturally in many essential oils, but is most abundant in the oils from seeds of caraway, spearmint, and dill.

Citral is an acyclic monoterpene aldehyde. Being a monoterpene, it is made of two isoprene units. Citral is a collective term which covers two geometric isomers that have their own separate names; the E-isomer is named geranial or citral A. The Z-isomer is named neral or citral B. These stereoisomers occur as a mixture, often not in equal proportions; e.g. in essential oil of Australian ginger, the neral to geranial ratio is 0.61.
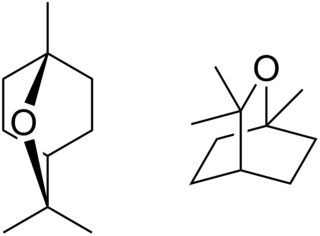
Eucalyptol is a monoterpenoid colorless liquid, and a bicyclic ether. It has a fresh camphor-like odor and a spicy, cooling taste. It is insoluble in water, but miscible with organic solvents. Eucalyptol makes up about 70–90% of eucalyptus oil. Eucalyptol forms crystalline adducts with hydrohalic acids, o-cresol, resorcinol, and phosphoric acid. Formation of these adducts is useful for purification.

Myrcene, or β-myrcene, is a monoterpene. A colorless oil, it occurs widely in essential oils. It is produced mainly semi-synthetically from Myrcia, from which it gets its name. It is an intermediate in the production of several fragrances. α-Myrcene is the name for the isomer 2-methyl-6-methylene-1,7-octadiene, which has not been found in nature.
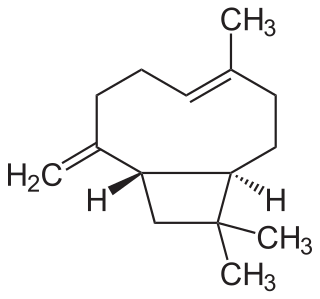
Caryophyllene, more formally (−)-β-caryophyllene (BCP), is a natural bicyclic sesquiterpene that is a constituent of many essential oils, especially clove oil, the oil from the stems and flowers of Syzygium aromaticum (cloves), the essential oil of Cannabis sativa, copaiba, rosemary, and hops. It is usually found as a mixture with isocaryophyllene and α-humulene, a ring-opened isomer. Caryophyllene is notable for having a cyclobutane ring, as well as a trans-double bond in a 9-membered ring, both rarities in nature.

Estragole is a phenylpropene, a natural organic compound. Its chemical structure consists of a benzene ring substituted with a methoxy group and an allyl group. It is an isomer of anethole, differing with respect to the location of the double bond. It is a colorless liquid, although impure samples can appear yellow. It is a component of various trees and plants, including turpentine, anise, fennel, bay, tarragon, and basil. It is used in the preparation of fragrances.

The ouzo effect, also known as the louche effect and spontaneous emulsification, is the phenomenon of formation of a milky oil-in-water emulsion when water is added to ouzo and other anise-flavored liqueurs and spirits, such as pastis, rakı, arak, sambuca and absinthe. Such emulsions occur with only minimal mixing and are highly stable.
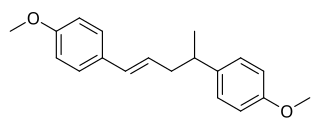
Dianethole is a naturally occurring organic compound that is found in anise and fennel. It is a dimeric polymer of anethole. It has estrogenic activity, and along with anethole and photoanethole, may be responsible for the estrogenic effects of anise and fennel. These compounds bear resemblance to the estrogens stilbene and diethylstilbestrol, which may explain their estrogenic activity. In fact, it is said that diethylstilbestrol and related drugs were originally modeled after dianethole and photoanethole.

Photoanethole is a naturally occurring organic compound that is found in anise and fennel. It has estrogenic activity, and along with anethole and dianethole, may be responsible for the estrogenic effects of anise and fennel. These compounds bear resemblance to the estrogens stilbene and diethylstilbestrol, which may explain their estrogenic activity. In fact, it is said that diethylstilbestrol and related drugs were originally modeled after photoanethole and dianethole.




















