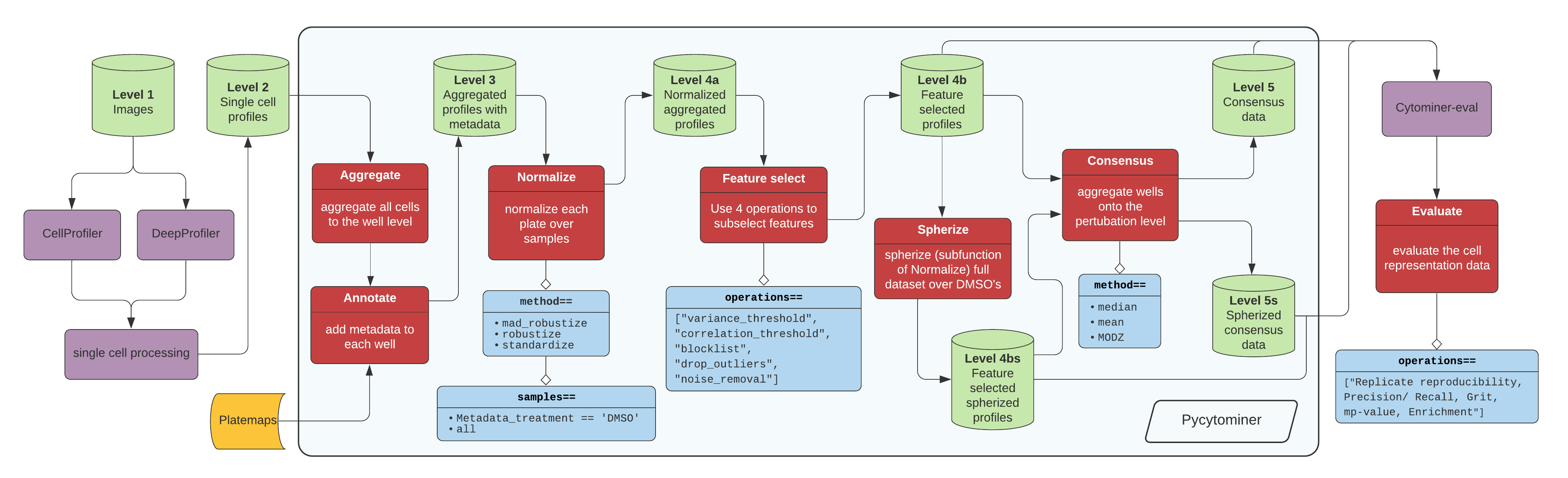
Pycytominer is a suite of common functions used to process high dimensional readouts from high-throughput cell experiments. The tool is most often used for processing data through the following pipeline:
Image data flow from the microscope to segmentation and feature extraction tools (e.g. CellProfiler or DeepProfiler). From here, additional single cell processing tools curate the single cell readouts into a form manageable for pycytominer input. For CellProfiler, we use cytominer-database or cytominer-transport. For DeepProfiler, we include single cell processing tools in pycytominer.cyto_utils.
From the single cell output, we perform five steps using a simple API (described below), before passing along our data to cytominer-eval for quality and perturbation strength evaluation.
The API is consistent for the five major processing functions:
- Aggregate
- Annotate
- Normalize
- Feature select
- Consensus
Each processing function has unique arguments, see our documentation for more details.
Pycytominer is still in beta, and can only be installed from GitHub:
pip install git+git://github.com/cytomining/pycytominerSince the project is actively being developed, with new features added regularly, we recommend installation using a hash:
# Example:
pip install git+git://github.com/cytomining/pycytominer@2aa8638d7e505ab510f1d5282098dd59bb2cb470If running your images on a cluster, unless you have a MySQL or similar large database set up then you will likely end up with lots of different folders from the different cluster runs (often one per well or one per site), each one containing an Image.csv, Nuclei.csv, etc.
In order to look at full plates, therefore, we first need to collate all of these CSVs into a single file (currently SQLite) per plate.
We currently do this with a library called cytominer-database.
If you want to perform this data collation inside pycytominer using the cyto_utils function collate (and/or you want to be able to run the tests and have them all pass!), you will need cytominer-database==0.3.4; this will change your installation commands slightly:
# Example for general case commit:
pip install "pycytominer[collate] @ git+git://github.com/cytomining/pycytominer"
# Example for specific commit:
pip install "pycytominer[collate] @ git+git://github.com/cytomining/pycytominer@2aa8638d7e505ab510f1d5282098dd59bb2cb470"If using pycytominer in a conda environment, in order to run collate.py, you will also want to make sure to add cytominer-database=0.3.4 to your list of dependencies.
The CellLocation class offers a convenient way to augment a LoadData file with X,Y locations of cells in each image.
The locations information is obtained from a single cell SQLite file.
To use this functionality, you will need to modify your installation command, similar to above:
# Example for general case commit:
pip install "pycytominer[cell_locations] @ git+git://github.com/cytomining/pycytominer"Example using this functionality:
metadata_input="s3://cellpainting-gallery/test-cpg0016-jump/source_4/workspace/load_data_csv/2021_08_23_Batch12/BR00126114/test_BR00126114_load_data_with_illum.parquet"
single_single_cell_input="s3://cellpainting-gallery/test-cpg0016-jump/source_4/workspace/backend/2021_08_23_Batch12/BR00126114/test_BR00126114.sqlite"
augmented_metadata_output="~/Desktop/load_data_with_illum_and_cell_location_subset.parquet"
python \
-m pycytominer.cyto_utils.cell_locations_cmd \
--metadata_input ${metadata_input} \
--single_cell_input ${single_single_cell_input} \
--augmented_metadata_output ${augmented_metadata_output} \
add_cell_location
# Check the output
python -c "import pandas as pd; print(pd.read_parquet('${augmented_metadata_output}').head())"
# It should look something like this (depends on the width of your terminal):
# Metadata_Plate Metadata_Well Metadata_Site ... PathName_OrigRNA ImageNumber CellCenters
# 0 BR00126114 A01 1 ... s3://cellpainting-gallery/cpg0016-jump/source_... 1 [{'Nuclei_Location_Center_X': 943.512129380054...
# 1 BR00126114 A01 2 ... s3://cellpainting-gallery/cpg0016-jump/source_... 2 [{'Nuclei_Location_Center_X': 29.9516027655562...Using pycytominer is simple and fun.
# Real world example
import pandas as pd
import pycytominer
commit = "da8ae6a3bc103346095d61b4ee02f08fc85a5d98"
url = f"https://media.githubusercontent.com/media/broadinstitute/lincs-cell-painting/{commit}/profiles/2016_04_01_a549_48hr_batch1/SQ00014812/SQ00014812_augmented.csv.gz"
df = pd.read_csv(url)
normalized_df = pycytominer.normalize(
profiles=df,
method="standardize",
samples="Metadata_broad_sample == 'DMSO'"
)Pycytominer was written with a goal of processing any high-throughput profiling data. However, the initial use case was developed for processing image-based profiling experiments specifically. And, more specifically than that, image-based profiling readouts from CellProfiler measurements from Cell Painting data.
Therefore, we have included some custom tools in pycytominer/cyto_utils.