
Crotalaria is a genus of flowering plants in the family Fabaceae commonly known as rattlepods. The genus includes over 700 species of herbaceous plants and shrubs. Africa is the continent with the majority of Crotalaria species, which are mainly found in damp grassland, especially in floodplains, depressions and along edges of swamps and rivers, but also in deciduous bush land, roadsides and fields. Some species of Crotalaria are grown as ornamentals. The common name rattlepod or rattlebox is derived from the fact that the seeds become loose in the pod as they mature, and rattle when the pod is shaken. The name derives from the Ancient Greek κρόταλον, meaning "castanet", and is the same root as the name for the rattlesnakes (Crotalus).

Borage, also known as starflower, is an annual herb in the flowering plant family Boraginaceae native to the Mediterranean region. Although the plant contains small amounts of pyrrolizidine alkaloids, some parts are edible and its seeds provide oil.

Tussilago farfara, commonly known as coltsfoot, is a plant in the tribe Senecioneae in the family Asteraceae, native to Europe and parts of western and central Asia. The name "tussilago" is derived from the Latin tussis, meaning cough, and ago, meaning to cast or to act on. It has had uses in traditional medicine, but the discovery of toxic pyrrolizidine alkaloids in the plant has resulted in liver health concerns.

Borage seed oil is derived from the seeds of the plant, Borago officinalis (borage).

Ageratum conyzoides is native to Tropical America, especially Brazil, and is an invasive weed in many other regions. It is an herb that is 0.5–1 m. high, with ovate leaves 2–6 cm long, and flowers are white to mauve.

Pyrrolizidine is a heterocyclic organic compound that forms the central chemical structure of a variety of alkaloids known collectively as pyrrolizidine alkaloids. It is one of five classes of iminosugars. These are often synthesized from a carbohydrate.

Utetheisa ornatrix, also called the ornate bella moth, ornate moth, bella moth or rattlebox moth, is a moth of the subfamily Arctiinae. It is aposematically colored ranging from pink, red, orange and yellow to white coloration with black markings arranged in varying patterns on its wings. It has a wingspan of 33–46 mm. Moths reside in temperate midwestern and eastern North America as well as throughout Mexico and other parts of Central America. Unlike most moths, the bella moth is diurnal. Formerly, the bella moth or beautiful utetheisa of temperate eastern North America was separated as Utetheisa bella. Now it is united with the bella moth in Utetheisa ornatrix.

A loline alkaloid is a member of the 1-aminopyrrolizidines, which are bioactive natural products with several distinct biological and chemical features. The lolines are insecticidal and insect-deterrent compounds that are produced in grasses infected by endophytic fungal symbionts of the genus Epichloë. Lolines increase resistance of endophyte-infected grasses to insect herbivores, and may also protect the infected plants from environmental stresses such as drought and spatial competition. They are alkaloids, organic compounds containing basic nitrogen atoms. The basic chemical structure of the lolines comprises a saturated pyrrolizidine ring, a primary amine at the C-1 carbon, and an internal ether bridge—a hallmark feature of the lolines, which is uncommon in organic compounds—joining two distant ring carbons. Different substituents at the C-1 amine, such as methyl, formyl, and acetyl groups, yield loline species that have variable bioactivity against insects. Besides endophyte–grass symbionts, loline alkaloids have also been identified in some other plant species; namely, Adenocarpus species and Argyreia mollis.

Senecionine is a toxic pyrrolizidine alkaloid isolated from various botanical sources. It takes its name from the Senecio genus and is produced by many different plants in that genus, including Jacobaea vulgaris. It has also been isolated from several other plants, including Brachyglottis repanda, Emilia, Erechtites hieraciifolius, Petasites, Syneilesis, Crotalaria, Caltha leptosepala, and Castilleja.
Pyrrolizidine alkaloidosis is a disease caused by chronic poisoning found in humans and other animals caused by ingesting poisonous plants which contain the natural chemical compounds known as pyrrolizidine alkaloids. Pyrrolizidine alkaloidosis can result in damage to the liver, kidneys, heart, brain, smooth muscles, lungs, DNA, lesions all over the body, and could be a potential cause of cancer. Pyrrolizidine alkaloidosis is known by many other names such as "Pictou Disease" in Canada and "Winton Disease" in New Zealand. Cereal crops and forage crops can sometimes become polluted with pyrrolizidine-containing seeds, resulting in the alkaloids contaminating flour and other foods, including milk from cows feeding on these plants.

Crotalaria cunninghamii, also known as green birdflower, birdflower ratulpo, parrot pea or regal birdflower, is a plant of the legume family Fabaceae, named Crotalaria after the Greek word for rattle, because their seeds rattle, and cunninghamii after early 19th century botanist Allan Cunningham. Crotalaria cunninghamii is known as Mangarr to the Nyangumarta Warrarn Indigenous group.

Hair-pencils and coremata are pheromone signaling structures present in lepidopteran males. Males use hair-pencils in courtship behaviors with females. The pheromones they excrete serve as both aphrodisiacs and tranquilizers to females as well as repellents to conspecific males. Hair-pencil glands are stored inside the male until courtship begins, at which point they are forced out of the body by sclerotized levers present on the abdomen. Coremata are very similar structures. Their exact definition is confused by early descriptions but they are more specifically defined as the internal, glandular, eversible structures that bear the hair-pencils and can be voluntarily inflated with hemolymph or air.

Riddelliine is a chemical compound classified as a pyrrolizidine alkaloid. It was first isolated from Senecio riddellii and is also found in a variety of plants including Jacobaea vulgaris, Senecio vulgaris, and others plants in the genus Senecio.

Parasenecio hastatus is a flowering plant species in the genus Parasenecio found in China and East Asia. It contains toxic pyrrolizidine alkaloids.

Hydroxydanaidal is an insect pheromone synthesized by some species of moth from pyrrolizidine alkaloids found in their diet.

Amabiline is a pyrrolizidine alkaloid first isolated in 1967 from Cynoglossum amabile. It is also found in the seeds and flowers of borage and in borage seed oil.

Echites umbellatus is a flowering climber, belonging to subfamily Apocynoideae of the family Apocynaceae and has the English common name devil's potato. It was first described in 1760 by Dutch botanist, Nikolaus Joseph von Jacquin. The species grows in parts of Florida, Tabasco, Yucatán Peninsula, Belize, Honduras, Cayman Islands, Cuba, Hispaniola, Jamaica, Leeward Islands, Bahamas, Turks and Caicos Islands, and the Colombian islands in the Western Caribbean.

Thomas Hartmann,, was a German pharmaceutical biologist and ecologist who was professor in the Department of Pharmaceutical Biology at the Technische Universität Braunschweig. His research focused on the biosynthesis, intracellular transport, and action of quinolizidine and pyrrolizidine alkaloids in fungi and plants and the sequestration of these secondary natural products by insects.
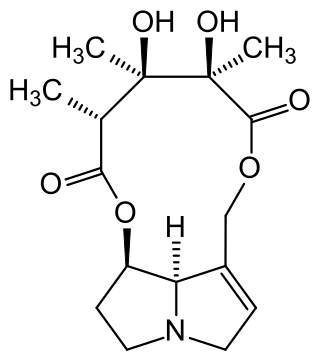
Monocrotaline (MCT) is a pyrrolizidine alkaloid that is present in plants of the Crotalaria genus. These species can synthesise MCT out of amino acids and can cause liver, lung and kidney damage in various organisms. Initial stress factors are released intracellular upon binding of MCT to BMPR2 receptors and elevated MAPK phosphorylation levels are induced, which can cause cancer in Homo sapiens. MCT can be detoxified in rats via oxidation, followed by glutathione-conjugation and hydrolysis.


















