Abstract
Sepsis is a life-threatening complication of pneumonia, including coronavirus disease-2019 (COVID-19)-induced pneumonia. Evidence of the benefits of vitamin C (VC) for the treatment of sepsis is accumulating. However, data revealing the targets and molecular mechanisms of VC action against sepsis are limited. In this report, a bioinformatics analysis of network pharmacology was conducted to demonstrate screening targets, biological functions, and the signaling pathways of VC action against sepsis. As shown in network assays, 63 primary causal targets for the VC action against sepsis were identified from the data, and four optimal core targets for the VC action against sepsis were identified. These core targets were epidermal growth factor receptor (EGFR), mitogen-activated protein kinase-1 (MAPK1), proto-oncogene c (JUN), and signal transducer and activator of transcription-3 (STAT3). In addition, all biological processes (including a top 20) and signaling pathways (including a top 20) potentially involved in the VC action against sepsis were identified. The hub genes potentially involved in the VC action against sepsis and interlaced networks from the Kyoto Encyclopedia of Genes and Genomes Mapper assays were highlighted. Considering all the bioinformatic findings, we conclude that VC antisepsis effects are mechanistically and pharmacologically implicated with suppression of immune dysfunction-related and inflammation-associated functional processes and other signaling pathways. These primary predictive biotargets may potentially be used to treat sepsis in future clinical practice.
Keywords: vitamin C, sepsis, network pharmacology, immune dysfunction, inflammation
Introduction
Sepsis results from pus-forming bacteria or virus, and post-inducing endotoxins, in the tissue or blood, and it is associated with an unacceptably high death rate [1]. The majority of sepsis cases are caused by pneumonia (itself a serious respiratory disorder), and cases can be community-acquired or hospital-acquired [2]. Although limited data are available regarding sepsis induced by COVID-19, pathogenic coronaviruses-induced sepsis is observed with Middle East respiratory syndrome [3]. Septic shock is associated with infection-driven systemic immune and organ dysfunction and is characterized by a severe inflammatory response to infection [4].
Common clinical treatments for sepsis aim to reverse dysfunctions of the organ and immune system and include immunomodulating therapies [5]. In clinical practice, an anti-infection strategy using antibacterials is prescribed to manage sepsis [6]. However, the chemotherapeutics (including antibacterial agents, antiviral drugs) used for antisepsis have been linked with adverse reactions [7, 8]. As a result, there is an express need for pharmacologically bioactive components that combat sepsis.
Vitamin C (VC) is a key trace element with known beneficial effects, including antioxidation effects, antiaging effects and the prevention of angiocardiopathy [9]. Interestingly, VC treatment was found to elevate the survival rate of septic patients in an intensive care unit [10]. Moreover, VC has been reported to be an alternative therapeutic agent against sepsis [11]. Clinical investigations have revealed that high doses of intravenous VC may reduce inflammation and vascular injury related to sepsis [12]. VC has several known pharmacological actions, and VC therapy may reduce inflammatory reaction, oxidative stress and ameliorate endovascular and immunologic dysfunctions [13].
Generally, data reporting the use of VC against sepsis have been collected from preliminary clinical investigations, and in-depth mechanistic preclinical studies are currently limited. In addition, details of biomarkers and the signaling pathways through which VC exerts its effects against sepsis remain unknown. A network pharmacology-based approach has previously proved successful in revealing treatable targets and mechanisms from bioinformatics assays [14]. Indeed, in our previous studies, we have identified the pharmacological biotargets of several bioactive agents used to manage clinical diseases [15, 16].
Accordingly, our present study was designed to use this emerging network pharmacology approach to reveal predictive targets and therapeutic mechanisms underlying the action of VC against sepsis. We hope that the predictive targets and therapeutic mechanisms may be subsequently verified in in vitro and in vivo experiments.
Methods
Screening of antiseptic targets of VC
Using the Traditional Chinese Medicine Systems Pharmacology (TCMSP) [17], Drugbank [18], SuperPred [19] and Swiss Target Prediction [20] databases, well-reported pharmacological targets of VC were obtained. Likewise, the DisGeNET [21], Drugbank, and Genecard [22] databases were used to screen pathological targets of sepsis. All primary targets of VC and sepsis were evaluated using Venn diagrams to identify potential targets for VC against sepsis [23].
Screening of best targets of VC against sepsis and construction of interrelated network
Using a STRING database, the mapped target of VC action against sepsis were reassayed to harvest a target-to-target function-related protein network, protein–protein interaction (PPI) network, and tsv. data. Furthermore, NetworkAnalyzer in Cytoscape_v3.7.1 was used to analyze topology parameters, such as median degrees of freedom and maximum degrees of freedom, in the network [24]. The best targets were selected based on degree values, with the upper limit of the filtering range taken as the maximum degree-value in the topology data and the lower limit taken as twice the median of freedom [25].
Assays of functional processes and molecular pathways
The R packages of ‘ClusterProfiler’, ‘ReactomePA’ and ‘AnnotationHub’ were used for enrichment analysis of Gene Ontology (GO) in biological function (BP) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway using the previously identified best targets. This data were used to generate a corresponding bubble chart and histogram [26]. GO and KEGG analyses were performed using clusterProfiler (version 3.14.3) [27] and ReactomePA (version 1.30.0) [28] from the R packages. GO data were acquired from org.Hs.eg.Db using a cut-off value adjusted to P < 0.05.
Visualization of network relationships
By using Cytoscape_v3.7.1 software, the targets of VC action against pneumonia and other best targets were enriched for KEGG-GENE visualization to facilitate further data analysis [29].
Results
A bioinformatics diagram of the target-based prioritization through systematic pharmacology approach that was used to help identify the VC action against sepsis is shown in Figure 1.
Figure 1.
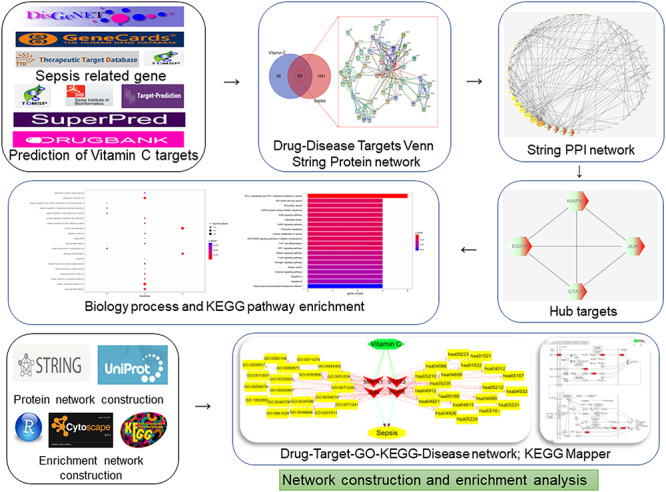
Investigative flowcharts for prioritization of biotargets and signaling pathways of VC action against sepsis through a systematic network pharmacology approach. All the reported genes/targets of VC and sepsis were screened and identified using databases available online prior to harvesting the antisepsis targets of VC. After the construction of a PPI network and identification of the hub targets of VC against sepsis, the biological processes and KEGG signaling pathways were determined. A network of VC-target-GO-KEGG-sepsis was visualized following bioinformatics analysis.
Detailed data of targets and PPI network
The inclusion criteria for sepsis screening through DisGeNET database was a gene-disease association score > 0.1 and for sepsis screening through GeneCards database was a gene score > 1. Using these criteria, 1704 genes associated with sepsis were identified. VC drug targets were screened and identified through both the TCMSP and Swiss Target Prediction databases, and 161 VC targets were identified (after removing duplicates) using Uniprot database. A Venn diagram of the two target sets was then used to obtain 63 intersection targets (Figure 2). Using a minimum required interaction score set to 0.700 (high confidence), a STRING database was used to collect function-related PPI data from the 63 targets (Supplementary Table 1). The pharmacological targets of VC action against sepsis and function-related PPI network was then constructed (Figure 2).
Figure 2.
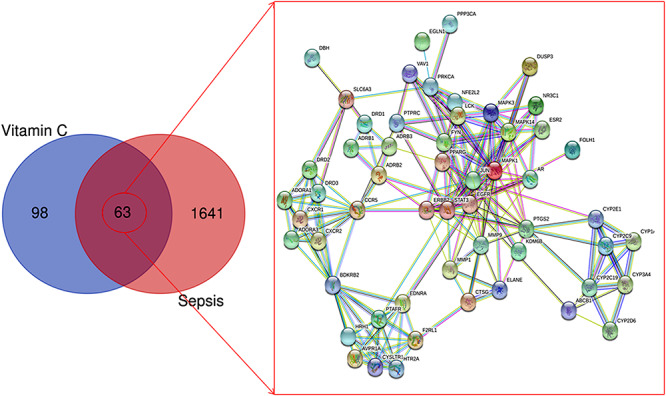
The total targets of VC, sepsis and 63 intersection targets of VC action against sepsis were identified via the Venn diagram and PPI network. In the analyses, the inclusion criteria for sepsis targets using DisGeNET and GeneCards databases were identified with gene-disease association score > 0.1 and gene score > 1. A PPI network of the 63 targets was plotted following a STRING database with the interaction score set to a high confidence at 0.700.
PPI network topology parameters and best core targets
The mapped proteins were imported into Cytoscape software to calculate the topological parameters of the interaction network of VC against sepsis targets and function-related proteins. The median degree of freedom of the target was 7.055 and the maximum degree of freedom was 18. The inclusion criteria for the core target screening conditions were set to 15 and 18 to obtain the optimal four core targets. These were epidermal growth factor receptor (EGFR), mitogen-activated protein kinase-1 (MAPK1), proto-oncogene c (JUN) and signal transducer and activator of transcription-3 (STAT3) (Figure 3).
Figure 3.
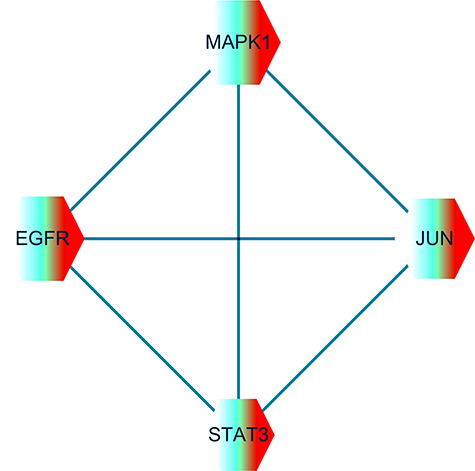
Hub genes/targets of VC action against sepsis were identified as EGFR, MAPK1, JUN and STAT3. In the algorithm using the Cytoscape software, the median and maximum degrees of freedom of the target were 7.055 and 18. The inclusion criteria for the screening of hub targets were set to 15 and 18 for harvesting the four important core targets.
Core targets in GO BPs and KEGG pathway enrichment
Using R language-related packages, the four core targets were analyzed for GO BP enrichment and by KEGG pathway analysis. The results of these analyses were visualized using a GO BP bubble diagram and histogram (Figure 4A–C), and an enriched in KEGG pathways bubble diagram and histogram (Figure 5A–C). According to the findings, GO BPs related to these targets included regulation of muscle cell proliferation; cellular response to cadmium ion; response to cadmium ion; epithelial cell proliferation; astrocyte differentiation; eyelid development in camera-type eye; cellular response to reactive oxygen species; cellular response to metal ion; positive regulation of epithelial cell proliferation; cellular response to inorganic substance; glial cell differentiation; positive regulation of DNA metabolic process; response to reactive oxygen species; positive regulation of gene silencing by micro RNA (miRNA); positive regulation of posttranscriptional gene silencing; learning or memory, gliogenesis; cognition; positive regulation of pre-miRNA transcription by RNA polymerase II; and cellular response to oxidative stress. Other related BPs are listed in Supplementary Table 2. The KEGG 90 pathways related to these targets (P < 0.05) included programmed death-ligand 1 (PD-L1) expression and PD-1 checkpoint pathway in cancer; non–small-cell lung cancer; pancreatic cancer; EGFR tyrosine kinase inhibitor resistance; ErbB signaling pathway; colorectal cancer; GnRH signaling pathway; endocrine resistance; choline metabolism in cancer; AGE-RAGE signaling pathway in diabetic complications; Th17 cell differentiation; HIF-1 signaling pathway; relaxin signaling pathway; FOXO signaling pathway; estrogen signaling pathway; breast cancer; oxytocin signaling pathway; Hepatitis C; Hepatitis B; and Kaposi sarcoma-associated herpesvirus infection. Other related pathways are listed in Supplementary Table 3.
Figure 4.
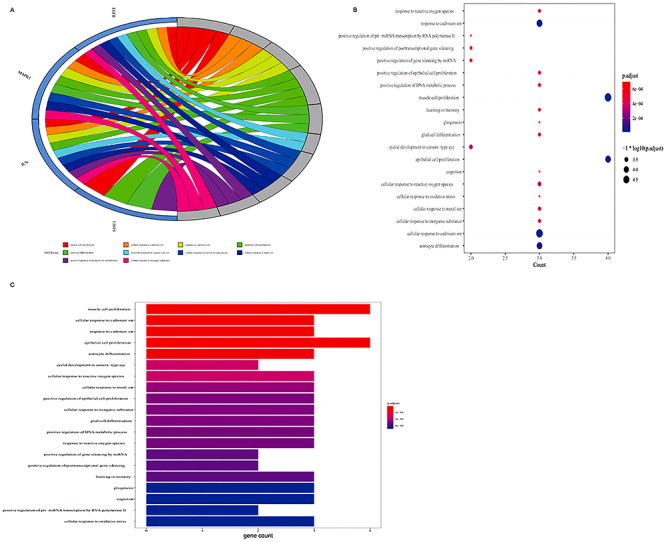
Biological process (BP) from bioinformatics data showing the top 20 BPs of VC action against sepsis. With the use of R-language packages, the four core targets were reassayed for GO-based biological function enrichment. (A) The blue Circro circles show the genes enriched in the top 10 items, and a total of top 10 BP enrichment items. (B) The bubble diagram shows the top 20 BP enrichment data. The x-axis represents the gene ratio and the intensities of different colors represent the adjusted P-value. (C) The histograms show the top 20 BP enrichment results. The x-axis represents the enriched gene count and the intensities of different colors represent the adjusted P-value.
Figure 5.
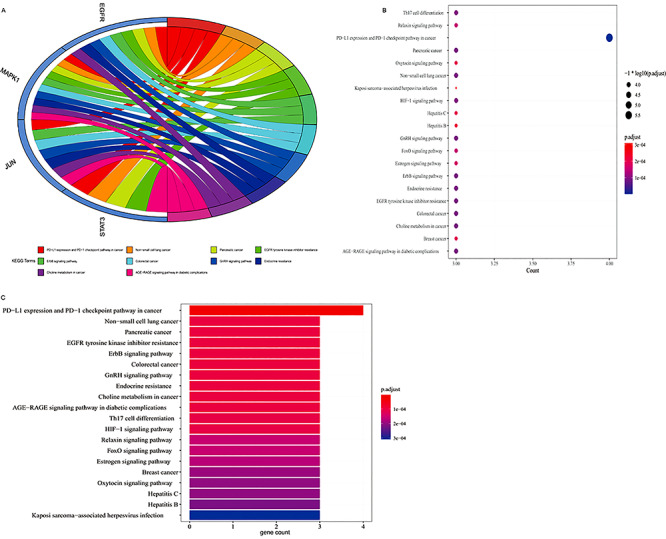
Detailed molecular pathways from bioinformatics findings exhibited the top 20 KEGG pathways of VC action against sepsis. Using the R-language packages, the four core targets were reanalyzed to reveal the KEGG molecular pathways. (A) The blue Circro circles show the genes enriched in the top 10 KEGG items. (B) The bubble diagram shows the top 20 KEGG enrichment pathways; the x-axis represents the gene ratio and the intensities of different colors represent the adjusted P-value. (C) The histograms highlight the top 20 KEGG enrichment pathways. The x-axis represents the enriched gene count and the intensities of different colors represent the adjusted P-value.
Construction of network diagram
Using Cytoscape software, a network visualization of VC against sepsis targets and an interaction diagram for core target-related pathways were generated (Figure 6). Additional hub genes from interlaced networks identified through KEGG Mapper assays were highlighted in red (Supplementary Figure S1).
Figure 6.
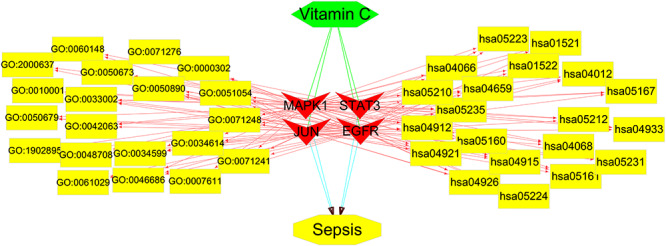
Network visualization from bioinformatics analysis highlighting the detailed interactions of VC-target-GO-KEGG-sepsis. The pink ovals represent the top 20 biological processes (pharmacological activities) in the action of VC against sepsis, and the yellow diamonds represent the top 20 KEGG signaling pathways (pharmacological mechanisms) in the action of VC against sepsis.
Discussion
Sepsis is the main cause of infection-associated death and therefore a healthcare priority [30]. The potential clinical pathogenesis of sepsis includes oxidative stress, immune dysfunction and inflammatory infiltration [31]. In the early stages of sepsis, an excessive immunological reaction leads to immune dysfunction, leaving the patient susceptible to secondary infections [32]. As the pathogenesis of sepsis is related to immune dysfunction and inflammatory stress, potential agents with antisepsis activities should target these processes. As mentioned above, clinical findings suggest that supplemental VC may reduce infection risks, including those associated with human sepsis [33]. However, the antisepsis therapeutic mechanism exerted by VC has not been characterized. The use of systematic network pharmacology for the identification and optimization of screening biotargets, functional processes, and molecular pathways of VC action against sepsis is an attractive research approach that can be completed before any future preclinical or clinical research. Using a promising bioinformatics assay, all potential biotargets of VC action against sepsis were methodically obtained, and the potential core pharmacological targets were identified as EGFR, MAPK1, JUN and STAT3. Functionally, EGFR activation can induce the proliferation of epithelial cells through regulation of the FOXO signaling pathway [34]. Some evidences indicate that suppression of the EGFR pathway via the reduction of eIF2α phosphorylation increases susceptibility to cellular oxidative stress [35]. It was also revealed in a clinical study that patients with EGFR mutation had increased PD-L1 expression and T cell infiltration [36]. The other identified core target, MAPK1, was responsible for the proliferation and survival of epithelial cells [37]. Increased activity of MAPK1 was reported to induce the survival of dendritic cells through the suppression of PD-1 expression [38]. In contrast, inhibition of Jun also reduced the proliferation and transition of epithelial cells [39]. In addition, activation of Jun expression could induce the activity of PD-L1 via glutamine depletion [40]. Other evidences also suggest that the activation of STAT3 is positively correlated with PD-L1 expression in inducing of the development of lymphoma [41]. Moreover, it has been reported that STAT3 promotes the proliferation of epithelial cells through the suppression of Krüppel-like factor 4 (KLF4) [42], and mediates oxidative stress in the lysosomal system [43]. Our bioinformatics analysis clearly showed that the four identified core targets might beneficially suppress sepsis-associated epithelial cell necrosis or apoptosis in the lung, peritoneum and bile duct. Furthermore, VC-mediated antioxidative effect might contribute to the reduction in the release of inflammatory cytokines and sepsis-related inflammatory reaction. In future investigations, we intend to elucidate the pharmacological mechanisms underlying the action of VC against sepsis through the modulation of EGFR, MAPK1, JUN and STAT3.
In network pharmacology analysis, additional bioinformatics findings from GO annotation and KEGG analysis revealed that the potential VC antisepsis effects were mechanistically linked to pharmacological modulation of epithelial cell proliferation, cellular response to oxidative stress, immune dysfunction and inflammatory stress. From the detailed bioinformatics data, analysis of the molecular mechanisms correlated with VC action against sepsis suggests that VC action against sepsis is pharmacologically achieved via the regulation of EGFR tyrosine kinase inhibitor resistance pathway, and ErbB, GnRH and FOXO signaling pathways, as well as through the inactivation of intrapulmonary Th17 cell differentiation, the Th1 and Th2 cell differentiation pathways, the B cell receptor signaling pathway, the Toll-like receptor signaling pathway, IL-17 signaling pathways and TNF signaling pathways.
Immunological dysfunction is a disease condition where the immune system fails in its normally physiological action. Pathologically, immunological dysfunction can be associated with existing clinical diseases, such as allergy, acquired immune deficiency syndrome and rheumatism [44]. It has been reported that the changes to the innate and adaptive immune responses that occur during sepsis are a lasting development, and that immune-modulatory treatment is necessary [45]. The main pathogenesis of sepsis is a cytokine ‘storm’ induced by an autoamplifying inflammatory mediator such as IL-17 or TNF-α [46–47]. Together, the bioinformatics findings we have harvested hint that the specific inactivation of molecular pathways of immune dysfunction and inflammatory stress and targeted modulation of intrapulmonary EGFR, MAPK1, JUN and STAT3 expressions, may be potential pathways of VC action against sepsis. In future clinical practice, chemotherapeutics, including inflammation-based inhibitors and immunomodulators, may enhance the therapeutic effectiveness and reduce the side effects of VC supplementation during the management of sepsis.
Conclusion
Overall, our bioinformatics approach of using network pharmacology has effectively highlighted potential therapeutic targets, BPs and pharmacological pathways of VC action against sepsis. The bioinformatics data presented identify potential targets that may be validated in a preclinical study before being used for the clinical treatment of sepsis with VC.
Key Points
Network pharmacology analysis is a potent approach for untangling all potential biotargets and signaling pathways of agents used to treat clinical diseases.
All antisepsis biotargets exerted by vitamin C were identified by systematic pharmacology.
The most important biotargets of vitamin C against sepsis were identified accordingly.
The top biological processes and molecular pathways of vitamin C action against sepsis were screened and highlighted.
Vitamin C may be a potent agent for treating clinical sepsis according to our bioinformatics findings.
Supplementary Material
Wenjun Huang is a researcher at the Guangxi Key Laboratory of Tumor Immunology and Microenvironmental Regulation, Guilin Medical University. She carried out her work creating network pharmacology and assays for revealing the causes of clinical diseases.
Funding
National Natural Science Foundation of China (No. 81560134, 81860097) and National Natural Science Foundation of Guangxi (No. 2018GXNSFBA281025, 2019GXNSFBA185015).
Conflict of interest
The authors declare that they have no conflict of interest.
References
- [1]. Cecconi M, Evans L, Levy M. Rhodes. Sepsis and septic shock. Lancet 2018;392:75–87. [DOI] [PubMed] [Google Scholar]
- [2]. Giuliano KK, Baker D. Sepsis in the context of nonventilator hospital-acquired pneumonia. Am J Crit Care 2020;29:9–14. [DOI] [PubMed] [Google Scholar]
- [3]. Graham CA. Outbreaks, Middle East respiratory syndrome and sepsis in emergency care. Eur J Emerg Med 2015;22:297. [DOI] [PubMed] [Google Scholar]
- [4]. Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med 2013;369:840–51. [DOI] [PubMed] [Google Scholar]
- [5]. Gotts JE, Matthay MA. Sepsis:pathophysiology and clinical management. BMJ 2016;353:1585. [DOI] [PubMed] [Google Scholar]
- [6]. Minasyan H. Sepsis: mechanisms of bacterial injury to the patient. Scand J Trauma Resusc Emerg Med 2019;27:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7]. Domagala JM. Structure-activity and structure-side-effect relationships for the quinolone antibacterials. J Antimicrob Chemother 1994;33:685–706. [DOI] [PubMed] [Google Scholar]
- [8]. Lin GL, McGinley JP, Drysdale SB, et al. Epidemiology and immune pathogenesis of viral sepsis. Front Immunol 2018;9:2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9]. Ingles DP, Cruz Rodriguez JB, Garcia H. Supplemental vitamins and minerals for cardiovascular disease prevention and treatment. Curr Cardiol Rep 2020;22:22. [DOI] [PubMed] [Google Scholar]
- [10]. Byerly S, Parreco J, Soe-Lin H, et al. Vitamin C and thiamine are associated with lower mortality in sepsis. J Trauma Acute Care Surg 2020. [DOI] [PubMed] [Google Scholar]
- [11]. Marik PE. Vitamin C for the treatment of sepsis: the scientific rationale. Pharmacol Ther 2018;189:63–70. [DOI] [PubMed] [Google Scholar]
- [12]. Brant EB, Angus DC. Is high-dose vitamin C beneficial for patients with sepsis? JAMA 2019;322:1257–8. [DOI] [PubMed] [Google Scholar]
- [13]. Kashiouris MG, L'Heureux M, Cable CA, et al. The emerging role of vitamin C as a treatment for sepsis. Nutrients 2020;12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14]. Li R, Ma X, Song Y, et al. Anti-colorectal cancer targets of resveratrol and biological molecular mechanism: analyses of network pharmacology, human and experimental data. J Cell Biochem 2019;120:11265–73. [DOI] [PubMed] [Google Scholar]
- [15]. Pan Q, Zhou R, Su M, et al. The effects of plumbagin on pancreatic cancer: a mechanistic network pharmacology approach. Med Sci Monit 2019;25:4648–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16]. Xiao H, Qin X, Wan J, et al. Pharmacological targets and the biological mechanisms of formononetin for Alzheimer's disease: a network analysis. Med Sci Monit 2019;25:4273–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17]. Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Chem 2014;6:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18]. Wishart DS, Feunang YD, Guo AC, et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res 2018;46:1074–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19]. Nickel J, Gohlke BO, Erehman J, et al. SuperPred: update on drug classification and target prediction. Nucleic Acids Res 2014;42:26–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20]. Gfeller D, Grosdidier A, Wirth M, et al. SwissTargetPrediction: a web server for target prediction of bioactive small molecules. Nucleic Acids Res 2014;42:32–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21]. Piñero J, Queralt-Rosinach N, Bravo À, et al. DisGeNET: a discovery platform for the dynamical exploration of human diseases and their genes. Database (Oxford) 2015;2015:028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22]. Fishilevich S, Nudel R, Rappaport N, et al. GeneHancer: genome-wide integration of enhancers and target genes in GeneCards. Database (Oxford) 2017;2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23]. Zhou R, Wu K, Su M, et al. Bioinformatic and experimental data decipher the pharmacological targets and mechanisms of plumbagin against hepatocellular carcinoma. Environ Toxicol Pharmacol 2019;70:103200. [DOI] [PubMed] [Google Scholar]
- [24]. Su G, Morris JH, Demchak B, et al. Biological network exploration with Cytoscape 3. Curr Protoc Bioinform 2014;47:1–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25]. Li J, Guo C, Lu X, et al. Anti-colorectal cancer biotargets and biological mechanisms of puerarin: study of molecular networks. Eur J Pharmacol 2019;858:172483. [DOI] [PubMed] [Google Scholar]
- [26]. Ge B, Guo C, Liang Y, et al. Network analysis, and human and animal studies disclose the anticystitis glandularis effects of vitamin C. Biofactors 2019;45:912–9. [DOI] [PubMed] [Google Scholar]
- [27]. Yu G, Wang LG, Han Y, et al. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 2012;16:284–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28]. Yu G, He QY. ReactomePA: an R/bioconductor package for reactome pathway analysis and visualization. Mol Biosyst 2016;12:477–9. [DOI] [PubMed] [Google Scholar]
- [29]. Li R, Song Y, Ji Z, et al. Pharmacological biotargets and the molecular mechanisms of oxyresveratrol treating colorectalcancer: network and experimental analyses. Biofactors 2020;46:158–67. [DOI] [PubMed] [Google Scholar]
- [30]. Napolitano LM. Sepsis 2018: definitions and guideline changes. Surg Infect (Larchmt) 2018;19:117–25. [DOI] [PubMed] [Google Scholar]
- [31]. Uhle F, Lichtenstern C, Brenner T, et al. Pathophysiology of sepsis. Anasthesiol Intensivmed Notfallmed Schmerzther 2015;50:114–22. [DOI] [PubMed] [Google Scholar]
- [32]. Reyes M, Filbin MR, Bhattacharyya RP, et al. An immune-cell signature of bacterial sepsis. Nat Med 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33]. Briegel J. High-dose vitamin C administration in patients with sepsis and acute respiratory distress syndrome: comments on the CITRIS-ALI study. Anaesthesist 2019;68:852–3. [DOI] [PubMed] [Google Scholar]
- [34]. Saxena R, Chandra V, Manohar M, et al. Chemotherapeutic potential of 2-[piperidinoethoxyphenyl]-3-phenyl-2H-benzo(b)pyran in estrogen receptor- negative breast cancer cells: action via prevention of EGFR activation and combined inhibition of PI-3-K/Akt/FOXO and MEK/Erk/AP-1 pathways. PLoS One 2013;8:e66246. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- [35]. Kim MJ, Choi WG, Ahn KJ, et al. Reduced EGFR level in eIF2α phosphorylation deficient hepatocytes is responsible for susceptibility to oxidative stress. Mol Cells 2020;43:264–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36]. Chen K, Cheng G, Zhang F, et al. PD-L1 expression and T cells infiltration in patients with uncommon EGFR-mutant non-small cell lung cancer and the response to immunotherapy. Lung Cancer 2020;142:98–105. [DOI] [PubMed] [Google Scholar]
- [37]. Upadhya D, Ogata M, Reneker LW. MAPK1 is required for establishing the pattern of cell proliferation and for cell survival during lens development. Development 2013;140:1573–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38]. Park SJ, Namkoong H, Doh J, et al. Negative role of inducible PD-1 on survival of activated dendritic cells. J Leukoc Biol 2014;95:621–9. [DOI] [PubMed] [Google Scholar]
- [39]. Liu F, Shang YX. Sirtuin 6 attenuates epithelial-mesenchymal transition by suppressing the TGF-β1/Smad3 pathway and c-Jun in asthma models. Int Immunopharmacol 2020;82:106333. [DOI] [PubMed] [Google Scholar]
- [40]. Ma G, Liang Y, Chen Y, et al. Glutamine deprivation induces PD-L1 expression via activation of EGFR/ERK/c-Jun signaling in renal cancer. Mol Cancer Res 2020;18:324–39. [DOI] [PubMed] [Google Scholar]
- [41]. Estrada CC, Paladugu P, Guo Y, et al. Krüppel-like factor 4 is a negative regulator of STAT3-induced glomerular epithelial cell proliferation. JCI Insight 2018;3:e98214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42]. Martínez-Fábregas J, Prescott A, van Kasteren S, et al. Lysosomal protease deficiency or substrate overload induces an oxidative-stress mediated STAT3-dependent pathway of lysosomal homeostasis. Nat Commun 2018;9:5343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43]. Shen J, Li S, Medeiros LJ, et al. PD-L1 expression is associated with ALK positivity and STAT3 activation, but not outcome in patients with systemic anaplastic large cell lymphoma. Mod Pathol 2020;33:324–33. [DOI] [PubMed] [Google Scholar]
- [44]. Fuentes E, Fuentes M, Alarcón M, et al. Immune system dysfunction in the elderly. An Acad Bras Cienc 2017;89:285–99. [DOI] [PubMed] [Google Scholar]
- [45]. Delano MJ, Ward PA. Sepsis-induced immune dysfunction: can immune therapies reduce mortality? J Clin Invest 2016;126:23–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46]. Faix JD. Biomarkers of sepsis. Crit Rev Clin Lab Sci 2013;50:23–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47]. Chousterman BG, Swirski FK, Weber GF. Cytokine storm and sepsis disease pathogenesis. Semin Immunopathol 2017;39:517–28. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


