Abstract
BACKGROUND Anopheles darlingi is the most efficient vector of malaria parasites in the Neotropics. Nevertheless, the specificities of its larval habitats are still poorly known.
OBJECTIVES Characterize permanent larval habitats, and population dynamics of An. darlingi and other potential vectors in relation to climate, physicochemical variables, insect fauna and malaria cases.
METHODS A 14-month longitudinal study was conducted in Porto Velho, Rondônia, western Brazilian Amazon. Monthly, 21 permanent water bodies were sampled. Immature anophelines and associated fauna were collected, physicochemical characteristics, and climate variables were recorded and analyzed.
FINDINGS Five types of habitats were identified: lagoon, stream, stream combined with lagoon, stream combined with dam, and fishpond. A total of 60,927 anophelines were collected. The most abundant species in all habitats were Anopheles braziliensis and An. darlingi. The highest density was found in the lagoon, while streams had the highest species richness. Abundance was higher during the transition period wet-dry season. There was a lag of respectively four and five months between the peak of rainfall and the Madeira River level and the highest abundance of An. darlingi larvae, which were positively correlated with habitats partially shaded, pH close to neutrality, increase dissolved oxygen and sulphates.
MAIN CONCLUSIONS The present study provides data on key factors defining permanent larval habitats for the surveillance of An. darlingi and other potential vectors as well as a log-linear Negative Binomial model based on immature mosquito abundance and climate variables to predict the increase in the number of malaria cases.
Key words: Anopheles spp.; climate; principal components; Poisson log-linear model; Negative Binomial log-linear model; malaria
Anopheles (Nyssorhynchus) darlingi Root is considered the most efficient vector of malaria parasites in the Neotropics,1,2 especially in the Amazon basin3,4 where over 90% of malaria cases in the Americas occur.5 The State of Rondônia (RO), in the southwestern Brazilian Amazon, has been historically a hot spot for malaria transmission, particularly in the municipality of Porto Velho (PVH).6,7 Between 2000 and 2021, 182,117 cases were reported in RO, 52% of which were reported for PVH.8 As in the rest of the Amazon, the causes for high malaria prevalence are diverse and complex, but notably deforestation for human settlements, agriculture, mining, and timbering together with human migration including cross-border malaria, parasite resistance to drugs, relapses of Plasmodium vivax, insecticide resistance and vector behavioral resistance are among those causes.9,10,11,12
Although An. darlingi has been associated with riverine forested areas,13 changes in land use have created appropriate conditions for the proliferation of this species throughout a vast area east of the Andes between 10º N and 20º S.14-22
Larval habitats of An. darlingi are diverse, and include natural water bodies such as streams, lagoons and lakes, and man created larval habitats such as fishponds, dams, abandoned gold mining dugouts, and brick pits.23,24 Despite the importance of identification and characterization of anopheline larval habitats for the design and implementation of control interventions, few studies have been conducted in the Amazon basin, particularly in Brazil.25-31 Moreover, longitudinal studies characterizing larval habitats and describing population dynamics of immature stages of An. darlingi, and other potential or secondary malaria vectors have not been conducted in the Brazilian Amazon.
The present study was conducted for 14 months with the goal of characterizing anopheline larval habitats and focusing on the population dynamics of An. darlingi in relation to climate, physicochemical variables, associated fauna and malaria cases in PVH.
MATERIALS AND METHODS
Study area - The municipality of PVH, RO, (08º 07’S, 63º 39’W), Brazil, with an area of 34,091 km2. At the time of the study, the municipality accounted for 65 neighborhoods in the urban area and 446 localities in peri-urban and rural areas, with a population of 314,525 people.32 This area is located over the following geological formations: Jamari Complex, Serra de la Providencia Granites, Jací-Paraná Formation33 and the Rio Madeira Formation, composed of fluvial sediments beginning with a basal conglomerate with sandy and calcareous matrix, frequent manganese, and carbonaceous material.34 The physical relief has been classified as flat and gently undulating.35 The study area is in the Madeira River basin. The water level of the Madeira River may vary up to 21 m between February and April, down to 9 m between September and November.36 The Madeira River runs across PVH and influences the formation of natural and permanent water collections that are suitable larval habitats for anophelines all year.
The climate in PVH is characterized by high temperature and humidity, with a mean annual temperature of 26ºC, average relative humidity of 86%, and annual rainfall of 2,300 mm,37,38 and two well defined seasons: the rainy season (October to April), and a dry season (May to September) [Supplementary data (Table I)].
Although rate of deforestation has increased, the study area is mostly covered by Amazonian Forest, with trees of 30 to 50 m high represented by genera such as Hevea, Bertholetia and Diniza.39
For decades, PVH has been classified as an area of high malaria risk.40 Of the total number of malaria cases reported in the municipality during the study period (17,856), 74% were reported from rural areas and the remaining 26% from urban/peri-urban areas.32 The most prevalent malaria parasite species is P. vivax (88.7%), followed by Plasmodium falciparum (10.4%) and mixed infections (0.9%).32 Although the situation has improved in recent years, PVH was still among the six municipalities with the highest number of malaria cases in the country in 2020 (~ 6,000 cases); although, a lower risk (Annual Parasite Index < 10) was reported for 2023.41
Climatological data - Monthly air temperature, relative humidity and cumulative rainfall were obtained from the government climatological station located in the city of PVH.37
Identification and selection of larval habitats - It has long been known that the larval habitats of An. darlingi in the Brazilian Amazon are essentially large, deep and permanent water bodies.3,23,42 Thus, the urban and peri-urban areas of PVH were surveyed for large, deep and permanent water bodies. All potential habitats were then identified, inventoried, and evaluated against the following inclusion criteria for use in a longitudinal study: 1) located in an area with recently reported malaria cases, 2) without vector control interventions during the last 12 months, and 3) free of evident domestic or industrial pollution.
Characterization of the aquatic environment - The following characteristics were recorded once a month, between 7:30 and 9:30 am, at each larval habitat for 14 months (April 1998 - May 1999): water turbidity, presence of floating detritus (twigs, leaves, flowers, and fruits), water movement (subjectively accessed as stagnant, moderate and strong), and associated vegetation as emergent, floating or submersed according to the classification of Hess and Hall.43 The physical characteristics included water surface area (km2), depth (m), temperature (ºC) and sun exposure (%); the later subjectively estimated as shaded, partial, or totally exposed to sunlight. The LaMotte Company kit (Chestertown, MD, USA) was used for determining water chemistry: dissolved oxygen (DO), dissolved carbon dioxide (DCO2), silica (SiO2), calcium carbonate (CaCO3), ammonia (NH4), sulphates (SO4 -2), nitrate (NO3 -), phosphorus (P), and chlorides (Cl-) expressed in mg/L; the pH was measured using a Lutron pH-meter (model pH 206). The chemical variables were estimated as the mean number of the results for three samples of water from the same larval habitat at each sampling event.
Larval sampling technique - The standard form for characterization of larval habitats of the Brazilian National Malaria Control Program was used. Briefly, immature mosquitoes and associated fauna were sampled with a 350 mL dipper, and at least 85 scoops taken at each sampled site per month. All collected Anopheles larvae were counted according to the instars (L1, L2, L3 and L4) and pupae; the L4 stage were fixed in 80% ethanol for species identification and the pupae were placed in plastic containers and transported to the laboratory and reared to adults for species identification. Larvae were collected from the margins of the habitats as well as among floating vegetation. An inflatable boat was used to collect samples in the large and deep habitats.
Species identification - Collected larvae were treated with a 10% potassium hydroxide for cleaning the specimens, then transferred to a microscope slide and examined under an Olympus CX4 microscope at 400X (eye piece 10X and objective 40X). The keys by Consoli & Lourenço-de-Oliveira42 and Faran44 and Linthicum45 were used for the identification of fourth stage larvae and adults that emerged from collected pupae. The other aquatic insects collected were preserved in 80% ethanol and identified at the family level using the keys of Stehr46 and Chu & Cutkomp.47
Data analysis - Cross-correlations and time series analyses were conducted between the monthly anopheline species abundance and rainfall, the level of the Madeira River, relative humidity (% RH), air temperature and the number of malaria cases. These analyses were also used to determine the time lag between each pair of environmental variables, mosquito species, and malaria cases using R Studio.48
A log-linear Poisson model was used to model the response variable (each anopheline species and total mosquitoes) and the explanatory (environmental) variables, with selection based on the highest correlation value obtained.
To test the interaction between multiple factors such as time (months) and physicochemical water characteristics of the most common larval habitats (streams) on the response variables (taxa abundance data), the distance-based redundancy analysis (dbRDA) was used.49 The software CANOCO version 4.0 was used for all the analysis.50 The dbRDA is a non-parametric test used to determine the significance of individual factors in a multifactorial variance analysis for multiple variables using association measurements of the principal components analysis.51 The abundance and temporal distribution of aquatic insects (anopheline larvae and other insects) in relation to the larval habitat conditions (physicochemical variables) and the community structure were investigated by means of a partial ordination technique.51 The larval density of the most abundant anophelines species were related to environmental variables and natural predators (aquatics insects) using the dbRDA. The RDA is recommended for the analysis of sites representing short ecological gradients where a linear relationship between species and environmental factors is assumed.50,52 Additionally, the partial RDA was used to determine if variations in the response variables could be attributed to explicative variables. For performing the RDA analysis, three matrices were used: matrix Y (Anopheles species larvae density per month); matrix X (physicochemical water properties), and matrix W (families of aquatic insects collected). For each analysis, the statistical significance of the overall model (relationship between the set of variables and the taxa abundances) was tested using 999 Monte Carlo permutations tests and the value of the sum of all canonical eigenvalues was recorded.50,51
RESULTS
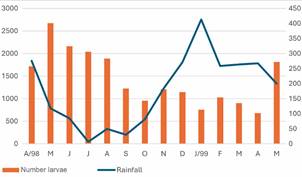
Climatological data - During the study period, the mean annual temperature was 26.4ºC, 68% mean annual relative humidity and 1,800 mm of rainfall.37 The level of the Madeira River varied from 3.32 m in September 1998 to 14.82 m in March 1999 [Fig. 1, Supplementary data (Table I)]. The time series data for the PVH climatological data for the period 1999-2021 reported a mean annual temperature of 26.1ºC, 84% relative humidity and annual rainfall of 2,216 mm,53 higher than that reported for the study period. This variation was mainly due to the strong El Niño Southern Oscillation event (ENSO) between June 1997 and June 1998.54
monthly rainfall (mm) and Madeira River level (m) between April 1998 and May 1999. Porto Velho, Rondônia, Brazil.
Characterization of the aquatic environment - A total of 137 potential larval habitats corresponding to diverse hydrological types were identified in the study area, such as streams (igarapés), lagoons, dams, rain pools, ponds, swamps, excavations for the extraction of sand, and drainage channels. However, only 21 (16%) of them met the inclusion criteria and included: streams (N = 15), lagoon (1), fishpond (1), stream combined with lagoon (2) and stream combined with dam (2) (Fig. 2).
relative location of permanent larval habitats of anophelines in the Municipality of Porto Velho, Rondônia, Brazil.
Streams: hydrological type most abundant in the study area; they are permanent with size varying from 3.45 ± 5.20 km2 and, 1.16 ± 0.84 m deep with moderate flow and shade produced by tall trees along the margins. The color of the water varies from dark to yellowish, while turbidity varies from slightly to completely turbid. Emergent vegetation such as Montrichardia spp. as well as floating plants like Eichhornia sp. and Pistia sp. were common in this type of larval habitat. The palm trees Mauritia vinifera (buriti) and Orbignya oleifera (babaçu) usually grew along the margins.
Lagoon: small (~ 800 m2) water body occurring in natural terrain depression originated from overflow of streams, 1.91 ± 0.61 m in depth; it was permanent with high diversity of emergent vegetation. The surrounding vegetation included tall trees and the banks were essentially covered by grass.
Fishpond: stagnant permanent water body created by construction of a dam in a stream for rearing the fish Colossoma macropomum; water is moderately turbid with a surface area of 3.4 km2, and 0.79 ± 0.23 m in depth. The pond margins were covered by grass.
Stream combined with lagoon: permanent and partially shaded water bodies, with a surface area of 1.05 ± 0.46 km2 and 1.05± 0.46 m in depth; moderate flow and turbidity varied from slightly turbid to clear.
Stream combined with dam: this permanent water body was created by damming a stream for domestic use. Throughout the study the water was slightly turbid, and with organic detritus present. The estimated surface area was 1.58 ± 0.32 km2 and 1.46 ± 0.89 m in depth; there was moderate water flow, and it was partially shaded.
Table I shows the physicochemical characteristics of each type of the 21 selected larval habitats.
Species and anopheline abundance - Between April 1998 and May 1999, 308 collections were performed in 21 habitats (Fig. 2). A total of 60,927 anopheline immatures (L1 to L4 and pupae) were collected (Table I). Although 66.7% were found in the 15 sampled streams, the highest mean number of larvae per scoop, regardless of developmental stage, was detected in the lagoon (19.5). Also, the higher mean number of identified immature forms (L3, L4 and pupae) collected per type of site was recorded for the lagoon (2,096) (Table II), followed by streams combined with lagoon (1,196.5), streams combined with dam (1,055), the fishpond (897) and stream (816.2). The number of larvae per scoop was higher for the lagoon (19.4/10), a slightly smaller number of immatures were collected in the streams combined with dam (17.5/10), followed by streams combined with lagoon, streams and the fishpond had the smaller frequency (8.7/10) [Table II, Supplementary data (Tables II-III)].
Number of Anopheles larvae identified in permanent aquatic habitats of Porto Velho, Rondônia, Brazil. April 1998 - May 1999
Due to the difficulties in identifying early stages, only L4 and some L3 could be identified to species; representing 32.4% (19,744) of all specimens collected and comprising 10 species, with eight belonging to the subgenus Nyssorhynchus and two to the subgenus Anopheles (i.e., An. mattogrosensis and An. peryassui). The most abundant species were An. braziliensis (50.7%) and An. darlingi (28.0%) (Table II). The streams were the type of habitat with higher species richness (10 species collected). Regarding the spatial distribution of species, An. braziliensis, An. darlingi, An. peryassui, An. triannulatus s.l., An. evansae and An. mattogrosensis are considered generalist since they were collected at least once in all types of larval habitats. In contrast, An. oswaldoi s.l. was only collected in streams, while An. nuneztovari s.l. and An. albitarsis s.l. were never collected from the fishpond (Table II).
Anopheles darlingi was considerably more common in the lagoon, where 2.8 larvae (L3 and L4 larvae) were collected per scoop. Frequency of An. darlingi larvae per scoop was similar in the streams and stream combined with lagoon or dam (1 - 1.1), and less frequent in the fishpond (0.8).
Anopheline abundance in relation to environmental variables - There was a major peak of the total identified immature stages of Anopheles spp. in May 1998, during the transition period from the rainy season to the dry season (Fig. 3).
total number of Anopheles spp. immature stages identified collected monthly in all type of habitats in relation to rainfall (mm) in Porto Velho, Rondônia, Brazil. April 1998 - May 1999.
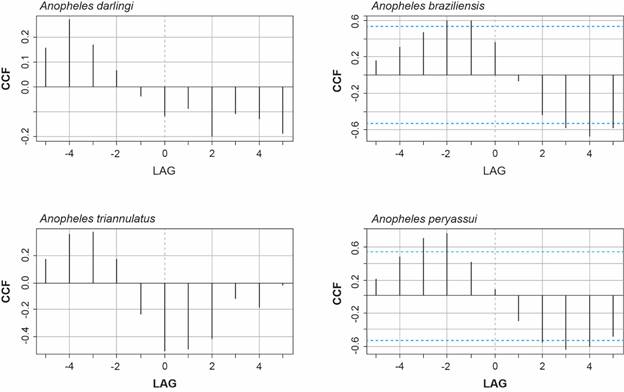
Regarding the temporal distribution of the four most abundant species (An. darlingi, An. braziliensis, An. peryassui and An. triannulatus s.l.) in relation to rainfall, temperature, relative humidity and river level, a cross-correlation analysis was conducted to determine the time lag between each pair of environmental variables and mosquito species. Fig. 4 illustrates the cross-correlation relationship between the variable “Rainfall” and the population of the most abundant mosquito species. The plot clearly indicates that the cross-correlation function (CCF) line for rainfall, with lags of one and two months, surpasses the threshold for statistical significance, indicating a significant association between the two variables, particularly for An. braziliensis and An. peryassui.
cross-correlation analysis between the variable rainfall and number of the four most abundant Anopheles species collected. Porto Velho, Rondônia, Brazil. April 1998 - May 1999.
Additionally, the ‘lag2.plot’ function from the Applied Statistical Time Series Analysis (astsa) R library48 was used to display the precise values of the correlations for the four most abundant species and the climate lagged variables.
To model the response of anopheline abundance to environmental variables, five Generalized Linear Models (GLM) function with a Poisson family were developed, each model focused on each of the four most abundant species (An. braziliensis, An. darlingi, An. triannulatus s.l. and An. peryassui) and the total number of mosquito immatures collected monthly and for the explanatory variables, three environmental variables were selected based on the highest correlation value obtained from the ‘lag2.plot’.
The log-linear Poisson model for the number of immature mosquitoes can be expressed as:
Where E(Y(t)) is the expected value of the response variable (number of mosquitoes for a given species) at time which is assumed to have a Poisson distribution; Rain, RH, and RL are respectively Rainfall (mm), Relative humidity (%) and River level (m) at lags t-k 1 , t-k 2 and t-k 3 which are the lagged predictors in the model. Model coefficients β i ,i = 0,…,3 are estimated by maximum likelihood (ML) using the R function glm. Lags t-k 1 , t-k 2 and t-k 3 vary with mosquito species.
Table III shows the significant predictors (climate lagged variables) for each Anopheles species. Rainfall (mm) and River level (m) are significant (p < 0.05) in all models. However, relative humidity (% RH) was found to be significant in all models except for An. darlingi and An. triannulatus s.l.
P-values for the lagged environmental predictor variables of the monthly abundance of the four most abundant Anopheles species in Porto Velho, Rondônia, Brazil. April 1998 - May 1999
Relation of anophelines abundance, environmental variables, and malaria cases - To model the malaria cases in response to anophelines abundance and rainfall, relative humidity and river level (explanatory variables), a log-linear Negative Binomial model was developed to address overdispersion.
The number of malaria cases can be modeled as:
Where E(Z(t) is the expected number of malaria cases at time t; MN, Rain, RH, and RL are respectively Mosquito numbers, Rainfall (mm), Relative humidity (%) and River level (m) at lags at lag t-k, t-4, t-5 and t-4. In this case, a negative binomial distribution is assumed for the response variable Z(t). Model coefficients γ i ,i=0,…,4 are estimated by ML using the R function glm. The value of lag t-k varies with mosquito species.
In all malaria models, the variables “Rainfall” and “River level” lagged by four months, while “relative humidity” lagged by five months. The selection of lagged values for different mosquito species was based on the outcomes obtained from the cross-correlation functions.
The p-values for the explanatory variables indicate that “relative humidity” is significant (p < 0.01) when the species “darlingi” is present in the model. In the model with “total mosquitoes”, “river level” becomes significant (p = 0.0237). However, in other models, no significant variables were found.
We also conducted diagnostic tests for the model using “darlingi” and “total mosquitoes,” employing deviance-based goodness-of-fit tests and residual plots. As the p-values exceeded the significance level of 0.05, we can confidently conclude that the models fit the data well. Furthermore, the residual plots exhibited a random pattern in the distribution of data points, further supporting the adequacy of the model fit.
Associated fauna - Together with immature stages of mosquitos of the genus Anopheles, 13,779 aquatic insects belonging to the orders Coleoptera, Diptera, Ephemeroptera, Heteroptera and Odonata were collected (Table IV). The most abundant were immature stages of Diptera belonging to the subfamily Culicinae (6,617). When considering the density of aquatics insects by habitat type in terms of mean number collected per scoop, Coleoptera and Heteroptera showed the higher density in the lagoon; Diptera and Ephemeroptera in the stream combined with lagoon, while Odonata density was higher in the fishpond. The lowest density of all orders was found in the streams. Characidae fishes were frequently observed in all types of habitats. Among the aquatic insects collected there were important predators of mosquito larvae such as members of the families Dytiscidae (Coleoptera),55 Belostomatidae, Corixidae, Nepidae and Notonectidae (Heteroptera),56,57,58,59 Libellulidae y Coenagrionidae (Odonata).57,60,61,62,63 The higher density of aquatic insects besides Anopheles was also recorded in May 1998 during the transition period between rainy and dry season showing a similar pattern as anopheline larval abundance.
Interaction between physicochemical water parameters, time (months), abundance of Anopheles species and aquatic insects - Throughout the study, the water temperature within and between larval habitats did not show significant differences; thus, this variable was not considered in further analysis. The dbRDA of the Anopheles species in relation to environmental variables showed different temporal distributions and associations with the different physical and chemical variables in the streams, which were the most common larval habitats (Fig. 5A). The RDA ordination of variables in relation to Anopheles species abundance reflected the difference in species compositions among months (Fig. 5A). The first axis explains 13% of total variance in species distribution and abundance and the second axis explains 43%. Overall, An. braziliensis, An. darlingi, An. peryassui, An. albitarsis s.l., An. triannulatus s.l., An. evansae and An. nuneztovari s.l. were well-represented in the ordination diagram, with more than 12% of the variance accounted for the significance. Although this value might seem low, this type of analysis is highly informative.50 The diagram summarizes the relation of anopheline species with time (months) and indicates the estimated localization of maximum abundances and optimal physicochemical conditions for each species. The diagram shows two groups of species separated by the first axis: one group to the right comprising An. darlingi, An. evansae, An. triannulatus s.l., An. peryassui and An. braziliensis, and the other group to the left comprised by An. nuneztovari s.l. and An. albitarsis s.l.
(A) Correlation biplots based on distance-based redundancy analysis (dbRDA) ordination of environmental variables, months, and Anopheles species in streams. Porto Velho, Rondônia, Brazil. April 1998 - May 1999. Vectors (arrows) point in the direction of increasing values for the respective variables; longer vectors indicate stronger correlations between variables scores and axes. (B) Correlation biplots based on dbRDA ordination of aquatic insects and Anopheles species in streams. Porto Velho, Rondônia, Brazil. April 1998 - May 1999. Vectors (arrows) point in the direction of increasing values for the respective variables; longer vectors indicate stronger correlations between variables scores and axes.
Anopheles darlingi is positively correlated with shaded larval habitats, pH close to neutrality, increased DO and sulphates during May (rainy-dry transition period); a similar pattern was shown by An. evansae and An. triannulatus s.l., although in less shaded larval habitats. On the other hand, An. darlingi is negatively correlated with increased water temperature and depth, increased ammonia, and dissolved carbon dioxide.
Anopheles peryassui is correlated with the period of less rainfall in June and higher phosphates and chlorides. An. albitarsis s.l. and An. nuneztovari s.l. show contrasting behavior; An. albitarsis s.l. is correlated with October (dry-rainy transition period), less shaded habitats with higher dissolved carbon dioxide and ammonia, while An. nuneztovari s.l. is correlated with the months January, February, and March (rainy season), shallow and sun exposed larval habitats with lower dissolved carbon dioxide and ammonia, and pH closed to neutrality. An. braziliensis, the most abundant species, was positively correlated with the month of April (beginning of the rainy-dry transition period), deep larval habitats with increased concentration of nitrates and calcium carbonate, while this interaction decreased during August and September [Supplementary data (Table IV)]. Due to total loss of the primary data, it was not possible to conduct similar analyses among and between different types of larval habitats.
The ordination of Anopheles species in relation to aquatic insects is shown in Fig. 5B. The first axis explains 44% of the variance in species abundance and the second axis 22%. In the upper left-hand side of the figure, we found that An. darlingi is positively correlated with Nepidae (Heteroptera), and negatively correlated with Libellulidae (Odonata), Corixidae and Pleidae (Heteroptera); a similar association with these insect families was found for An. triannulatus s.l. and An. nuneztovari s.l. but significantly less strong than that observed for An. darlingi. Contrasting results were observed for An. albitarsis s.l., which is positively correlated with these families. An. braziliensis and An. peryassui are correlated with Notonectidae, Veliidae, Hydrometidae, Naucoridae and Gerridae (Heteroptera), and Dytiscidae and Noteridae (Coleoptera); although negatively associated with Belostomatidae (Heteroptera), Trycorithidae (Ephemeroptera), and Coenagrionidae (Odonata). An. evansae is the only species correlated positively with members of these predatory families [Supplementary data (Table V)].
DISCUSSION
Anopheles darlingi has a wide range of distribution in the Neotropics, from southern Mexico to northern Argentina13 and is the most efficient vector of malaria parasites in South America in terms of high anthropophilic habits and its capacity to transmit all three parasite species P. falciparum, P. vivax and P. malariae.3,64,65,66 The females of this species may oviposit in a wide variety of natural and man-made water bodies, permanent or temporal.4,13 However, its main larval sites are usually permanent, large and deep-water bodies like rivers, streams, lakes and lagoons. Human activities have also created water bodies that are colonized by An. darlingi and associated species, such as dams, fishponds, ditches, clay pits, ponds, abandoned gold mines and containers.3,4,13-15,18-20,23,24,27,30,42,67-71
Tremendous efforts have been made to reduce the incidence of malaria in the Brazilian Amazon, particularly in Rondônia.7,64 Nevertheless, the persistence of transmission might be due to resistance of the parasites to the available drugs,72,73 and the biting (partially exophagic) and resting behavior (exophilic) of populations of An. darlingi and other secondary vectors present in the area that escape interception either by long lasting insecticide treated nets (LLINs) and/or insecticide residual spraying (IRS).22,23 Studies conducted in urban and peri-urban areas of PVH have shown that An. darlingi females are the most abundant species with a biting peak outdoors between 1800 and 2000 hrs.22,74,75 These results suggest that traditional vector control measures are less effective, and alternative and/or combined interventions are necessary. Therefore, WHO76 has indicated that supplementary interventions based on immatures control are effective in reducing vector density and malaria transmission where larval habitats of the principal malaria vector are few, fixed and findable, and where larval control is both feasible and cost-effective. The design and implementation of cost-effective larval source management (LSM) demands a profound knowledge of the ecology and population dynamics of vector species in larval sites.
The present study provides base line data to implement a surveillance program, an essential activity for vector control interventions and evaluation of their impact on malaria transmission. Five types of permanent anophelines larval habitats were identified and characterized: lagoon, stream, stream combine with lagoon, stream combine with dam, and fishpond. The species richness was higher in streams (10 species), and fishponds the least. An. darlingi, An. braziliensis, An. triannulatus s.l., An. peryassui, An. evansae, and An. mattogrosensis were collected, at least once, in all types of permanent water bodies; other species, although collected less frequently, were more specific. An. nuneztovari s.l. was not collected in the fishpond, while it was frequently collected (positivity index = 90.4%) in fishpond habitat in Amapá.30 These contrasting results might possibly be due to the fact that An. nuneztovari s.l. from Amapá may be a different species from the species present in PVH. As it has been shown, using morphological and molecular markers, An. nuneztovari is a complex of four species: An. nuneztovari s.s., An. goeldii, An. dunhami and An. jamariensis.77,78,79,80,81 Probably the species collected in Amapá in fishponds by Barbosa & Scarpassa30 is An. goeldii, while the species collected in the present study is the recently described species An. jamariensis collected in the municipality of Monte Negro, Rondônia.81 Similarly, An. albitarsis s.l. was never collected in the fishpond in PVH, but it was in Amapá30 and Amazonas.20An. albitarsis is a complex of at least 10 species of which only five have been formerly described: An. albitarsis sensu stricto, An. deaneorum, An. marajoara, An. oryzalimnetes, and An. janconnae.82,83,84,85An. deaneorum and An. oryzalimnetes have been reported from Rondônia;82,85,86 based on morphological characters, Meireles et al.22 and Rodrigues et al.87 identified adults collected in PVH, as An. deaneorum. It is not possible to establish the species identity of the mosquitoes of the Albitarsis complex collected during the present study. An. oswaldoi s.l., probably An. konderi22,87 was restricted to streams and with very low frequency. An. albitarsis s.l., An. nuneztovari s.l. and An. oswaldoi s.l. have been confirmed as vector of malaria parasites in different Amazonian states in Brazil [see Carlos et al.64 for a complete revision], as well as in Colombia88,89,90 and Venezuela,91,92,93,94 hence it is important that the surveillance program in PVH includes morphological and molecular identification of species belonging to these species complexes.
In general, the most productive larval habitat in terms of the mean number of larvae per scoop was the lagoon (Table I). The same was observed for An. darlingi which was more than twice as frequent there as in the other four habitat types (Table II). Similar results have been reported in Ameridian sites in Roraima and Amazonas,24 as well as in southern Venezuela.14,18,19,69 In terms of larval habitats, An. darlingi is a generalist species.4,13 The described physicochemical characteristics of water bodies within its range of distribution and types of most productive larval habitats vary. In the present study, the most distinctive characteristics of the lagoon in relation to the other types of water bodies is the higher percentage of shaded surface, depth, and DO. An. darlingi was frequently associated with the presence of An. braziliensis, An. triannulatus s.l. and An. albitarsis s.l. (Table II). An. braziliensis was the most abundant species in all larval habitats, particularly in the lagoon. These results highlight the heterogenous environmental conditions that determine the spatial distribution of anophelines species for the same type of larval habitat. In the Yanomami areas of Amazonas and Roraima, Sánchez-Ribas et al.24 reported that the most abundant species in lagoons was An. oswaldoi s.l. followed by An. darlingi; similar results were reported by Rubio-Palis et al.14 in a Yanomami area of southern Venezuela, while Moreno et al.18,69,95 reported that in lagoons in gold mining areas of southern Venezuela, An. triannulatus followed by An. albitarsis s.l. were the most abundant species.
Regardless of the type of water body, there are some physicochemical characteristics that might be important determinants for the development of An. darlingi such as partial shade, pH, depth, and DO. It has been reported that An. darlingi is not present in deep shaded forests where the most abundant mosquitoes belong to the genus Culex,96,97,98 nor in open savannahs in northern Roraima,99 Suriname4 and southern Venezuela.18An. darlingi is associated with sites of human interventions such as deforestation for agriculture, cattle ranching, urban development, forestry and mining, where the remaining forested areas and the transition zone (secondary forest-shrub) provides optimal ecological conditions for the colonization of permanent or temporal water bodies with shaded areas above 45% in Brazil, 27,70 Colombia (68), Peru,15 Suriname,100 Venezuela,14,18,19 and Belize.101 Furthermore, Barros & Honório70 have shown that An. darlingi larvae are found in areas with decreased luminosity compared to sunlit areas within the same water body.
The pH of water is an important determinant for the presence and abundance of mosquitoes as it has been demonstrated that it is directly related to cell functioning and acts on the permeability of the cell membrane.102 In general, anophelines are found in water bodies with a pH around 7, as reported in the present study and inclusive for An. darlingi. Indeed, An. darlingi and associated species were not present in fishponds and streams in Amapá at pH between 5.0 and 5.91.30 Curiously, Rufalco-Moutinho et al.27 collected An. darlingi in natural larval habitats 100 times more acidic (pH = 5.28) than man-made habitats (pH = 7.12), while this species has been found in natural water bodies like lagoons, swamps, and streams of southern Venezuela, at pH between 10 to 1,000 times more acidic (pH = 4.9-6.8).14,18,69 The pH in water bodies is related to the characteristics of the bedrock, soil composition, vegetation, debris, and other elements present and often due to human activities such as agricultural practices (slash and burn, fertilizers), and cattle ranching.103,104 These findings emphasize a need to characterize the larval habitats in particular environments where malaria is endemic.
Although anophelines depend on atmospheric oxygen to breathe, the concentration of DO in larval habitats is an indication of water quality and its potential for the development of immature stages. The reported values of DO in the present study are considered high and adequate for the development of anophelines, providing conditions for the proliferation of algae and other microorganisms that support the diet of mosquito larvae.105An. darlingi, An. triannulatus s.l., An. evansae, and An. peryassui are positively associated with high concentrations of DO in streams (Fig. 5A) and negatively associated with An. albitarsis s.l. and An. nuneztovari s.l. The values of DO have been considered a determinant factor for the presence of anopheline species in larval habitats showing either positive or negative relationship. Arcos et al.20 found a negative correlation between DO and An. darlingi in fishponds in Manaus, while in Amapá An. darlingi was not associated with DO but An. nuneztovari s.l. and An. triannulatus were most affected by it.30 In Ecuador, An. punctimacula was positively associated with DO, and no association was found for An. albimanus, An. pseudopunctipennis and An. oswaldoi s.l.,106 whereas An. albimanus is associated to low levels of DO in Mexico and Belize.107,108,109
Anopheles darlingi, as well as associated species, was less frequent in the fishpond, contrasting these results with more recent studies in Amazonas,20 Acre,27,67 Amapá30 and Roraima,70 as well as in the Peruvian Amazon15,110 where the proliferation of fish farms has created excellent conditions for the increased in abundance of An. darlingi and the risk for malaria transmission. It was not possible to analyze the significance of differences of physicochemical characteristics among water body types given the relatively low number of replicates.
An important characteristic of larval habitats is the presence of debris, floating and emergent vegetation as well as margins covered with grasses, providing food for larvae, refuges that protects from predators, and favorable conditions for oviposition.105,111,112,113 The dbRDA analysis showed that in general An. darlingi and the other anophelines species were negatively associated with various predacious aquatic insect families in the streams (Fig. 5B), except a positive relation between An. darlingi and the Nepidae family of Heteroptera, which might be due to only three specimens being collected.
The study of aquatic insects in the Neotropics as predators of mosquito larvae is scarce. There are some studies conducted under laboratory conditions as well as in micro-reservoirs such as artificial containers and phytothelmata showing that predation can control mosquito populations.114 Studies conducted in Mexico showed that the abundance of An. albimanus was lower in larval habitats with predators such as members of the order Heteroptera.115 During the present study, a rich fauna of aquatic insects representing 19 families and five orders were collected, including various predators of mosquito larvae.55-63,114 Nevertheless, the temporal distribution of anophelines is different from that of other aquatic insects. Anophelines are early colonizers of water bodies of the transition period from the rainy to dry season while predators are late colonizers, more abundant in the late rainy season.51 On the other hand, the development time of anopheline immature stages is much shorter than that of the predators.47 In relation to predators of Amazonian anophelines, it is suggested that only members of the families Noctonectidae and Veliidae (Heteroptera) could be of importance since they feed on small insects that rest on the water surface.116 The role of predators in the present study as determinants in the variation of anopheline larval abundance is doubtful. Other factors, such as climate, trophic and physicochemical characteristics of water bodies, might have a greater influence on the temporal and spatial abundance of An. darlingi and other anophelines in PVH.
Several studies have documented the influence of rainfall on mosquito larvae abundance.18,19,25,100,117 Depending on the Anopheles species and the type of larval habitat, this relation might be negative or positive. The temporal distribution of An. darlingi and associated species in PVH was significantly determined by rainfall and river level, independently of the type of permanent larval habitat. Furthermore, there was clearly a species succession which somehow reduces inter-specific competition: the abundance peak of An. braziliensis occurred two months after the peak of rainfall, whereas the peak for An. triannulatus s.l and An. peryassui occurred a month later (three months after the peak of rain), and An. darlingi peaks four months later. In general, the abundance of anophelines is higher in the transition period rainfall-drought and in the dry season (Fig. 3). Similar results were reported for An. darlingi larval abundance in Roraima,31 the Peruvian Amazon,15 southern Venezuela,18 and Belize;118 whereas Arcos et al.117 indicated an increase in the abundance of An. darlingi, An. triannulatus s.l. and An. nuneztovari s.l. during the rainy season in fishponds in Manaus. The increase in rainfall may produce several temporal larval habitats but also might have a negative effect producing the flush of larvae in habitats such as rivers and streams,71,100 resulting in the reduction of the adult population.25 The level of the Madeira River has a significant effect on anopheline’s abundance in PVH. The peak of precipitation precedes the peak river level (Fig. 1). The river level also showed a significant influence on the anopheline’s abundance in a successional manner where the peak of An. braziliensis occurred with a lag of one month, two months for An. peryassui, three for An. triannulatus, and five for An. darlingi (Table III).
Several studies have demonstrated the influence of climate on the abundance of Anopheles mosquitoes and on malaria in Brazil,119,120,121,122 and around the world.65,123,124,125,126 Studies in Brazil,120,121,122 Peru127 and Venezuela65 confirm the influence of river level on the incidence of malaria, with peaks after river floods. Most studies on climate, mosquito abundance and malaria incidence focused on adult females, since no correspondence was found between the abundance of adults and immature stages, probably because each life stage has different factors that influence mortality besides the duration of the aquatic cycle. In the present study, several malaria models were developed. Accordingly, water temperature variation was not significant and did not influence the mosquito immature populations. Significant variables were found only for relative humidity when An. darlingi was included in the model, and river level when all mosquito species were included. The models proposed in the present study can be used to predict an increase in the number of malaria cases in PVH.
Based on the results of the present study, larval source management of permanent habitats seems a feasible intervention to reduce vector populations and reduce transmission in PVH. Some studies have been conducted in the Brazilian Amazon using biolarvicides for the control of anopheline populations with variable results.128,129,130 Galardo et al.130 showed promising results with the use of Bacillus sphaericus in mining sites in Amapá. The abundance of immatures of An. darlingi were reduced throughout the 52-weeks study and adult populations were reduced during the rainy season. Other studies in the region have shown the effectiveness of B. sphaericus in reducing the abundance of An. darlingi larvae in fishponds in Peru,131An. albimanus in ponds in Colombia132 and An. triannulatus, An. nuneztovari s.l., An. darlingi, and An. albitarsis s.l. in lagoons in southern Venezuela.133 Furthermore, Moreno et al.134 showed that there was also a significant reduction in the incidence of malaria cases in gold mining areas. All the reported studies show that the product must be applied every four-nine weeks, and that requirement might make it unsustainable for vector control programs.
A key characteristic of permanent larval habitats in PVH was the presence of marginal vegetation, which provides food and refuge to mosquito larvae. A community-based program for the regular removal of vegetation of habitats such as lagoons, dams and fishponds might be cost-effective and serve as a long-term solution for the elimination of larval habitats.76-135
In accordance with the objectives of the Brazilian National Plan for the Elimination of Malaria,12 the present study provides relevant data on key factors for permanent larval habitats in PVH for the surveillance of An. darlingi and other potential vectors, as well as a Negative Binomial model based on immature mosquito abundance and climate variables (Relative Humidity and River Level) for predicting increases in the numbers of malaria cases.
ACKOWLEDGEMENTS
To Alda Eunice FL da Cunha, Aldeir de Oliveira Maia, Marisa Ferreira Barroso and Osvaldo Lopes da Silva who participated in field studies. To Raquel Telles Moreira Sampaio from INPA/BR for their assistance in the identification of aquatic insects. Maria Eugenia Grillet (UCV/Venezuela) assisted in the dbRDA analysis. Alexandro Machado de Freitas elaborated the map. Special thanks to Jarbas Barbosa da Silva Jr and Fabiano Pimenta, who were instrumental in financial support from the Ministry of Health. We are grateful to Don Roberts for correcting the English language.
REFERENCES
- 1 Rubio-Palis Y, Zimmerman RH. Ecoregional classification of malaria vectors in the neotropics. J Med Entomol. 1997; 34: 499-510.
- 2 Manguin S, Carnevale P, Mouchet J, Coosemans M, Julvez J, Richard-Lenoble D, et al. Biodiversity of malaria in the world. Montrouge: John Libbey Eurotext; 2008.
- 3 Deane LM. Malaria vectors in Brazil. Mem Inst Oswaldo Cruz. 1986; 81(Suppl. 2): 5-14.
-
4 Hiwat H, Bretas G. Ecology of Anopheles darlingi Root with respect to vector importance: a review. Parasit Vectors. 2011; 4: 177. doi: 10.1186/1756-3305-4-177.
» https://doi.org/10.1186/1756-3305-4-177 - 5 WHO - World Health Organization. World Malaria Report 2023. Geneva: World Health Organization; 2023. Available from: https://www.mmv.org/newsroom/news-resources-search/world-malaria-report-2023.
- 6 Marques AC. Human migration and the spread of malaria in Brazil. Parasitol Today. 1987; 3: 166-70.
- 7 Marques RD, Angelo JR, Alves de Lima A, Fuller T, Barcellos C. Production of urban space and the occurrence of malaria in the Brazilian Amazon: the Porto Velho case. Cien Saude Colet. 2021; 26(9): 4263-74.
-
8 SVS - Secretaria de Vigilância em Saúde. SIVEP-Malária. Ministério da Saúde, Brasil. 2022. Available from: https://public.tableau.com/app/profile/malaria.brasil/vizzes#!/
» https://public.tableau.com/app/profile/malaria.brasil/vizzes#!/ -
9 Ferreira MU, Castro MC. Challenges for malaria elimination in Brazil. Malar J. 2016; 15(1): 284. doi: 10.1186/s12936-016-1335-1.
» https://doi.org/10.1186/s12936-016-1335-1 -
10 Baia-da-Silva DC, Brito-Sousa JD, Rodovalho SR, Peterka C, Moresco G, Lapouble OMM, et al. Current vector control challenges in the fight against malaria in Brazil. Rev Soc Bras Med Trop. 2019; 52: e20180542. doi: 10.1590/0037-8682-0542-2018.
» https://doi.org/10.1590/0037-8682-0542-2018 - 11 Wetzler EA, Marchesini P, Villegas L, Canavati S. Changing transmission dynamics among migrant, indigenous and mining populations in a malaria hotspot in Northern Brazil: 2016 to 2020. Malar J. 2022; 21(1): 127.
- 12 MS/SVS/DIDT - Ministério da Saúde/Secretaria de Vigilância em Saúde/Departamento de Imunização e Doenças Transmissíveis. Elimina malária Brasil: plano nacional de eliminação da malária. Brasília: Ministério da Saúde, Secretaria de Vigilância em Saúde, Departamento de Imunização e Doenças Transmissíveis; 2022. 60 pp.
- 13 Sinka ME, Rubio-Palis Y, Manguin S, Patil AP, Temperley WH, Gething PW, et al. The dominant Anopheles vectors of human malaria in the Americas: occurrence data, distribution maps and bionomic précis. Parasit Vectors. 2010; 3: 72.
- 14 Rubio-Palis Y, Menare C, Quinto A, Magris M, Amarista M. Caracterización de criaderos de anofelinos (Diptera: Culicidae) vectores de malaria en el Alto Orinoco, Amazonas, Venezuela. Entomotropica. 2005; 20: 29-38.
- 15 Vittor AY, Pan W, Gilman RH, Tielsch J, Glass G, Shields T, et al. Linking deforestation to malaria in the Amazon: characterization of the breeding habitat of the principal malaria vector, Anopheles darlingi. Am J Trop Med Hyg. 2009; 81: 5-12.
- 16 Harris A, Matias-Arnéz A, Hill N. Biting time of Anopheles darlingi in the Bolivian Amazon and implications for control of malaria. Trans R Soc Trop Med Hyg. 2006; 100(1): 45-7.
- 17 Montoya-Lerma J, Solarte YA, Giraldo-Calderón GI, Quiñones ML, Ruiz-López F, Wilkerson RC, et al. Malaria vector species in Colombia - A review. Mem Inst Oswaldo Cruz. 2011; 106(Suppl. 1): 223-38.
- 18 Moreno JE, Rubio-Palis Y, Sánchez V, Martínez A. Caracterización de hábitats larvales de anofelinos en el municipio Sifontes del estado Bolívar, Venezuela. Bol Mal Salud Amb. 2015; 55(2): 117-131.
- 19 Moreno JE, Rubio-Palis Y, Bevilacqua M, Sánchez V, Guzmán H. Caracterización de hábitats larvales de anofelinos en la cuenca baja del Río Caura, estado Bolívar, Venezuela. Bol Mal Salud Amb. 2018; 58: 32-45.
- 20 Arcos AN, Ferreira FA, Cunh HB, Tadei WP. Characterization of artificial larval habitats of Anopheles darlingi (Diptera: Culicidae) in the Brazilian Central Amazon. Rev Bras Entomol. 2018; 62: 267-74.
- 21 Prussing C, Saavedra MP, Bickersmith SA, Alava F, Guzmán M, Manrique E, et al. Malaria vector species in Amazonian Peru co-occur in larval habitats but have distinct larval microbia communities. PLoS Negl Trop Dis. 2019; 13(5): e0007412.
- 22 Meireles ACA, Silva LR, Simplício MF, Lima AA, Rios FGF, Menezes CA, et al. Anopheline diversity in urban and peri-urban malaria foci: comparison between alternative traps and seasonal effects in a city in the Western Brazilian Amazon. Malar J. 2022; 21: 258.
- 23 Deane LM, Causey OR, Deane MP. Notas sôbre a distribuição e a biologia dos anofelinos das regiões nordestina e amazônica do Brasil. Rev Serv Saude Publica. 1948; 1(4): 827-965.
- 24 Sánchez-Ribas J, Oliveira-Ferreira J, Gimnig JE, Pereira-Ribeiro C, Santos-Neves MSA, Silva-do-Nascimento TF. Environmental variables associated with anopheline larvae distribution and abundance in Yanomami villages within unaltered areas of the Brazilian Amazon. Parasit Vectors. 2017; 10(1): 571.
- 25 Barros FSM, Arruda ME, Gurgel HC, Honório NA. Spatial clustering and longitudinal variation of Anopheles darlingi (Diptera: Culicidae) larvae in a river of the Amazon: the importance of the forest fringe and obstructions to flow in frontier malaria. Bull Ent Res. 2011; 101: 643-58.
- 26 McKeon SN, Schlichting CD, Póvoa MM, Conn JE. Ecological suitability and spatial distribution of five Anopheles species in Amazonian Brazil. Am J Trop Med Hyg. 2013; 88(6): 1079-86.
- 27 Rufalco-Moutinho P, Schweigmann N, Bergamaschi DP, Sallum MAM. Larval habitats of Anopheles species in a rural settlement on the malaria frontier of Southwest Amazon, Brazil. Acta Trop. 2016; 164: 243-58.
- 28 Sánchez-Ribas J, Oliveira-Ferreira J, Rosa-Freitas MG, Trilla L, Silva-do-Nascimento TF. New classification of natural breeding habitats for Neotropical anophelines in the Yanomami Indian Reserve, Amazon Region, Brazil and a new larval sampling methodology. Mem Inst Oswaldo Cruz. 2015; 110(6): 760-70.
- 29 Lima GR, Arcos AN, Santos EV, Simões RC, Lima CAP, Tadei WP. Abundância larval de Anopheles em criadouros artificiais na zona leste de Manaus, Amazonas. SAJEBTT. 2021; 8(1): 35-47.
- 30 Barbosa LMC, Scarpassa VM. Bionomics and population dynamics of anopheline larvae from an area dominated by fish farming tanks in northern Brazilian Amazon. PLoS One. 2023; 18(8): e0288983.
- 31 Almeida NCV, Louzada J, Neves MSAS, Carvalho TM, Castro-Alves J, Silva-do-Nascimento TF, et al. Larval habitats, species composition and distribution of malaria vectors in regions with autochthonous and imported malaria in Roraima State, Brazil. Malar J. 2022; 21: 13.
- 32 MS - Ministério da Saúde. Vigilância Ambiental em Saúde. Brasília: FUNASA; 1999.
- 33 Adamy A, Romanini S. Programa levantamentos geológicos básicos do Brasil. Geología da Região Porto Velho - Abuna, estados de Rondônia e Amazonas. Brasilia: DNPM/CRPM; 1990. 273 pp.
- 34 Rizzotto GJ, Oliveira JGF, Quadros MLES, Castro JMR, Cordeiro A, Adamy A, et al. Projeto Rio Madeira - levantamento de informações para subsidiar o estudo de viabilidade do aproveitamento hidroelétrico (AHE) do Rio Madeira. Relatório Final. CPRM-Serviço Geológico do Brasil, Porto Velho, Brasil; 2005. 423 pp.
- 35 Melo DP, Costa RCR, Natali Filho T. Geomorfologia. In: Brasil DNPM - Projeto RADAM/ Brasil, Levantamento de Recursos Naturais, MME; 1978. p. 185-250.
- 36 MMB - Ministério da Marinha do Brasil. Informe sobre flutuação do nível do Rio Madeira. Porto Velho: Ministério da Marinha do Brasil; 2002.
- 37 EMBRAPA/RO - Empresa Brasileira de Pesquisa Agropecuária/Rondônia. Boletim climatológico - 1998 e 1999. Documento mimeografado. Porto Velho: Empresa Brasileira de Pesquisa Agropecuária; 2000.
- 38 SEDAM/RO - Secretaria de Estado do Desenvolvimento Ambiental/Rondônia. Boletim climatológico de Rondônia - 1999. Porto Velho: Secretaria de Estado do Desenvolvimento Ambiental; 2000.
-
39 IBGE - Instituto Brasileiro de Geografia e Estatística. Vegetação. 2023. Available from: https://www.ibge.gov.br/geociencias/informacoes-ambientais/vegetacao/ [accessed 20 May 2024].
» https://www.ibge.gov.br/geociencias/informacoes-ambientais/vegetacao/ - 40 Marques AC, Gutierrez HC. Combate à malária no Brasil: evolução, situação atual e perspectivas. Rev Soc Bras Med Trop. 1994; 27(Suppl. 3): 91-108.
- 41 MS/SVS - Ministério da Saúde/Secretaria de Vigilância em Saúde. SIVEP-Malária. Brasília: Ministério da Saúde/Secretaria de Vigilância em Saúde; 2023.
- 42 Consoli RAGB, Lourenço-de-Oliveira R. Principais mosquitos de importância sanitária no Brasil. Rio de Janeiro: Fiocruz; 1994. 228 pp.
- 43 Hess AD, Hall TF. The relation of plants to malaria control on impounded waters with a suggested classification. J Natl Malar Soc. 1945; 4: 20-45.
- 44 Faran ME. Mosquito studies (Diptera: Culicidae) XXXIV: a revision of the albimanus section of the subgenus Nyssorhynchus of Anopheles. Contr Am Entomol Inst. 1980; 15: 1-215.
- 45 Linthicum KJ. A revision of the Argyritarsis section of the subgenus Nyssorhynchus of Anopheles (Diptera: Culicidae). Mosq Syst. 1988; 20: 98-271.
- 46 Stehr FW. Immature insects. Vol. 1. Dubuque: Kendall/Hunt Pub Co; 1987. 754 pp.
- 47 Chu HF. How to know. The immature insects. Dubuque: WC Brown Publishers; 1992. 234 pp.
-
48 RStudio Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna: 2023. Available from: http://www.rstudio.com/
» http://www.rstudio.com/ - 49 Legendre P, Anderson J. Distance-based redundancy analysis: testing multispecies responses in multifactorial ecological experiments. Ecol Monographs. 1999; 69: 1-24.
- 50 Ter Braak CJF, Smilauer P. CANOCO reference manual and user's guide to CANOCO for Windows. Software for canonical community ordination. Ithaca: Microcomputer Power; 1998. 325 pp.
- 51 Grillet ME, Legendre P, Bocard D. Community structure of Neotropical wetland insects in northern Venezuela. I. Temporal and environmental factors. Arch Hydrobiol. 2002; 155: 413-36.
- 52 Legendre P, Legendre L. Numerical ecology. 2nd ed. Vol. 24. Amsterdam: Elsevier; 1998. 853 pp.
-
53 Serviço Geológico do Brasil - CPRM. 1999-2021. Available from: https://cprm.gov.br/sace/boletins/Madeira
» https://cprm.gov.br/sace/boletins/Madeira -
54 NOAA - National Oceanic and Atmospheric Administration. ENSO. Available from: https://nooa/gov/magazine/enso/el-nino.html
» https://nooa/gov/magazine/enso/el-nino.html - 55 Becker N, Petric D, Zgomba M, Boase C, Madon M, Dahl C, et al. Mosquitoes and their control. Berlin: Springer; 2010. 403 pp.
- 56 Nam VS, Yen NT, Holynska M, Reid JW, Kay BH. National progress in dengue vector control in Vietnam: survey for Mesocyclops (Copepoda), Micronecta (Corixidae) and fish as biological control agents, Am J Trop. Med Hyg. 2000; 62(1): 5-10.
- 57 Shaalan EA, Canyon DV. Aquatic insect predators and mosquito control. Trop Biomed. 2009; 26(3): 223-61.
- 58 Nurushev MZ, Esenbekova PA, Temreshev I. Aquatic hemipteran bioregulators of blood-sucking Diptera of the Ile-Balkash region. Vestn Omsk Gos Agrar Univ. 2015; 1(17): 41-61.
- 59 Dalal A, Cuthbert RN, Dick JTA, Gupta S. Prey preferences of notonectids towards larval mosquitoes across prey ontogeny and search area. Pest Manage Sci. 2020; 76(2): 609-16.
- 60 Saidzhafarova AO, Perevozkin VP, Smirnov GV. Peculiarities of feeding behavior of dragonfly nymphs and their influence on the structure of larval populations of blood-sucking mosquitoes in the experimental system "predator-prey". Dokl Tomsk Gos Univ Sistem Upravl Radioelektr. 2006; 5(13): 14-20.
- 61 Kumar R, Muhid P, Dahms HU, Tseng LC, Hwang JS. Potential of three aquatic predators to control mosquitoes in the presence of alternative prey: a comparative experimental assessment. Mar Freshwater Res. 2008; 59(9): 817-35.
- 62 Barry MJ, Roberts DM. Indirect interactions limit the efficiency of Odonata as natural control agents for mosquito larvae. J Insect Behav. 2014; 27(5): 626-38.
- 63 Jacob S, Thomas AP, Manju EK. Bio control efficiency of Odonata nymphs on Aedes aegypti larvae. IOSR-JESTFT. 2017; 11(9): 1-4.
- 64 Carlos BC, Rona LDP, Christophides GK, Souza-Neto JA. A comprehensive analysis of malaria transmission in Brazil. Path Glob Health. 2019; 113(1): 1-13.
- 65 Magris M, Rubio-Palis Y, Menares C, Villegas L. Vector bionomics and malaria transmission in the Upper Orinoco River, southern Venezuela. Mem Inst Oswaldo Cruz. 2007; 102(3): 303-11.
- 66 Sinka ME, Bangs MJ, Manguin S, Rubio-Palis Y, Chareonviriyaphap T, Coetzee M, et al. A global map of dominant malaria vectors. Parasit Vectors. 2012; 5: 69.
- 67 Reis IC, Codoço CT, Degener CM, Keppeler EC, Muniz MM, Oliveira FGS, et al. Contribution of fish farming ponds to the production of immature Anopheles spp. in a malaria - endemic Amazonian town. Malar J. 2015; 14: 452.
- 68 Brochero H, Pareja PX, Ortiz G, Olano VA. Breeding places and biting activity of Anopheles species in the municipality of Cimitarra, Santander, Colombia. Biomedica. 2006; 26(2): 269-77.
- 69 Moreno J, Rubio-Palis Y, Acevedo P. Identificación de criaderos de Anofelinos en un área endémica del estado Bolívar, Venezuela. Bol Mal Salud Amb. 2000; 40: 21-30.
- 70 Barros FSM, Honório NA. Deforestation and Malaria on the Amazon Frontier: larval clustering of Anopheles darlingi (Diptera: Culicidae) determines focal distribution of malaria. Am J Trop Med Hyg. 2015; 93: 939-53.
- 71 Villarreal-Treviño C, Penilla-Navarro RP, Vázquez-Martínez MG, Moo-Llanes DA, Ríos-Delgado C, Fernández-Salas I, et al. Larval habitat characterization of Anopheles darlingi from its northernmost geographical distribution in Chiapas, Mexico. Malar J. 2015; 14: 517.
- 72 Inoue J, Lopes D, do Rosário V, Machado M, Hristov AD, Lima GFMC, et al. Analysis of polymorphisms in Plasmodium falciparum genes related to drug resistance: a survey over four decades under different treatment policies in Brazil. Malar J. 2014; 13: 372.
- 73 Costa GL, Amaral LC, Fontes CJF, Carvalho LH, Alves de Brito CF, Nóbrega de Sousa T. Assessment of copy number variation in genes related to drug resistance in Plasmodium vivax and Plasmodium falciparum isolates from the Brazilian Amazon and a systematic review of the literature. Malar J. 2017; 16: 152.
- 74 Lourenço-de-Oliveira R, Guimarães AEG, Arlé M, Silva TF, Castro MG, Motta MA, et al. Anopheline species, some of their habits and relation to malaria in endemic areas of Rondônia State, Amazon Region of Brazil. Mem Inst Oswaldo Cruz. 1989; 84(4): 501-14.
- 75 Charlwood JD. Biological variation in Anopheles darlingi Root. Mem Inst Oswaldo Cruz. 1996; 91(4): 391-8.
- 76 WHO - World Health Organization. Larval source management: a supplementary measure for malaria vector control: an operational manual. Geneva: 2013; 128 pp.
- 77 Trindade DB, Scarpassa VM. Genetic differentiation and diagnostic loci among Anopheles (Nyssorhynchus) rangeli, An. (Nys.) nuneztovari, and An. (Nys.) dunhami (Diptera: Culicidae) in the Brazilian Amazon. J Med Entomol. 2002; 39(4): 613-20.
- 78 Foster PG, Bergo ES, Bourke BP, Oliveira TMP, Nagaki SS, Sant'Ana DC, et al. Phylogenetic analysis and DNA-based species confirmation in Anopheles (Nyssorhynchus). PLoS One. 2013; 8: e54063.
- 79 Sant'Ana DC, Bergo ES, Sallum MAM. Anopheles goeldii Rozeboom & Gabaldón (Diptera, Culicidae): a species of the Nuneztovari Complex of Anopheles Meigen. Rev Bras Entomol. 2015; 59(1): 68-76.
- 80 Scarpassa VM, Cunha-Machado AS, Saraiva JF. Evidence of new species for malaria vector Anopheles nuneztovari sensu lato in the Brazilian Amazon region. Malar J. 2016; 15: 205.
- 81 Sant'Ana DC, Sallum MAM. A new species of the Nuneztovari Complex of Nyssorhynchus (Diptera: Culicidae) from the western Brazilian Amazon. Zootaxa. 2022; 5134(2): 275-85.
- 82 Rosa-Freitas MG. Anopheles (Nyssorhynchus) deaneorum: a new species in the Albitarsis complex (Diptera: Culicidae). Mem Inst Oswaldo Cruz. 1989; 84(4): 535-43.
- 83 Brochero HL, Li C, Wilkerson RC. A newly recognized species in the Anopheles (Nyssorhynchus) albitarsis complex (Diptera: Culicidae) from Puerto Carreño, Colombia. Am J Trop Med Hyg. 2007; 76(6): 1113-7.
- 84 Motoki MT, Wilkerson RC, Sallum MAM. The Anopheles albitarsis complex with the recognition of Anopheles oryzalimnetes Wilkerson and Motoki, n. sp. and Anopheles janconnae Wilkerson and Sallum, n. sp. (Diptera: Culicidae). Mem Inst Oswaldo Cruz. 2009; 104: 823-50.
- 85 Ruíz-López F, Wilkerson RC, Conn JE, McKeon SN, Levin DM, Quiñones ML, et al. DNA barcoding reveals both known and novel taxa in the albitarsis Group (Anopheles: Nyssorhynchus) of Neotropical malaria vectors. Parasit Vectors. 2012; 5: 44.
- 86 Motoki MT, Linton YM, Conn JE, Ruíz-López F, Wilkerson RC. Phylogenetic network of mitochondrial COI gene sequences distinguishes 10 taxa within the Neotropical albitarsis Group (Diptera: Culicidae), confirming the separate species status of Anopheles albitarsis H (Diptera: Culicidae) and revealing a novel lineage, Anopheles albitarsis J. J Med Entomol. 2021; 58(2): 599-607.
- 87 Rodrigues MS, Batista EP, Silva AA, Costa FM, Neto AS, Gil LHS. Change in Anopheles rishness and composition in response to artificial flooding during the creation of the Jirau hydroelectric dam in Porto Velho, Brazil. Malar J. 2017; 16: 87.
- 88 Herrera S, Suárez M, Sánchez G, Quiñones M, Herrera M. Uso de la técnica inmuno853 radiométrica (IRMA) en Anopheles de Colombia para la identificación de esporozoitos de Plasmodium. Colomb Med. 1987; 18: 2-6.
- 89 Quiñones ML, Ruiz F, Calle DA, Harbach RE, Erazo HF, Linton Y-M. Incrimination of Anopheles (Nyssorhynchus) rangeli and An. (Nys.) oswaldoi as natural vectors of Plasmodium vivax in Southern Colombia. Mem Inst Oswaldo Cruz. 2006; 101(6): 617-23.
- 90 Jiménez P, Conn JE, Wirtz R, Brochero H. Anopheles (Diptera: Culicidae) vectores de malaria en el municipio de Puerto Carreño, Vichada, Colombia. Biomédica. 2012; 32(Suppl. 1): 13-21.
- 91 Rubio-Palis Y, Wirtz RA, Curtis CF. Malaria entomological inoculation rates in western Venezuela. Acta Trop. 1992; 52(2-3): 167-74.
- 92 Moreno JE, Rubio-Palis Y, Páez E, Pérez E, Sánchez V, Vaccari E. Malaria entomological inoculation rates in gold mining areas of Southern Venezuela. Mem Inst Oswaldo Cruz. 2009; 104(5): 764-68.
- 93 Rubio-Palis Y. Prevalencia de Plasmodium spp. en anofelinos de Venezuela. Talleres. 2009; 12: 79-84.
- 94 Abou Orm S, Moreno JE, Carrozza M, Acevedo P, Herrera F. Plasmodium spp. infection rates for some Anopheles spp. from Sifontes Municipality, Bolivar State, Venezuela. Bol Mal Salud Amb. 2017; 57(1): 17-25.
- 95 Moreno JE, Rubio-Palis Y, Sánchez V, Martínez A. Fluctuación poblacional y hábitat larval de anofelinos en el municipio Sifontes, estado Bolívar, Venezuela. Bol Mal Salud Amb. 2015; 55(1): 52-68.
- 96 Hutchings RSG, Sallum MAM, Ferreira RLM, Hutchings RW. Mosquitoes of the Jaú National Park and their potential importance in Brazilian Amazonia. Med Vet Entomol. 2005; 19(4): 428-41.
- 97 Hutchings RSG, Hutchings RW, Sallum MAM. Culicidae (Diptera: culicomorpha) from the central Brazilian Amazon: Nhamundá and Abacaxis rivers. Zoologia. 2013; 30(1): 1-14.
- 98 Hutchings RSG, Hutchings RW, Menezes IS, Motta MA, Sallum MAM. Mosquitoes (Diptera: Culicidae) from the northwestern Brazilian Amazon: Padauari River. J Med Entomol. 2016; 53(6): 1-18.
- 99 Rosa-Freitas MG, Tsouris P, Peterson AT, Honorio NA, de Barros FSM, de Aguiar DB, et al. An ecoregional classification for the State of Roraima, Brazil. The importance of landscape in malaria biology. Mem Inst Oswaldo Cruz. 2007; 102(3): 349-57.
- 100 Rozendaal JA. Epidemiology and control of malaria in Suriname: with special reference to Anopheles darlingi. Dordrecht: 1990; 172 pp.
- 101 Manguin S, Roberts DR, Andre RG, Rejmánková E, Hakre S. Characterization of Anopheles darlingi (Diptera: Culicidae) larval habitats in Belize, Central America. J Med Entomol. 1996; 33: 205-11.
- 102 Clark TM, Flis BJ, Remold SK. Differences in the effects of salinity on larval growth and developmental programs of a freshwater and a euryhaline mosquito species (Insecta: Diptera, Culicidae). J Exp Biol. 2004; 207: 2289-95.
- 103 Scorza JV, Rodríguez M, Moreno G. Ecología poblacional de Anopheles nuñeztovari Gabaldon, 1940 en el Occidente de Venezuela. Bol Dir Malariol San Amb. 1981; 21:1-27.
- 104 Mosher JJ, Findlay RH. Direct and indirect influence of parental bedrock on streambed microbial community structure in forested streams. Appl Environ Microbiol. 2011; 77(21): 7681-8.
- 105 Rejmánková E, Grieco J, Achee N, Roberts DR. Ecology of larval habitats. In: Manguin S, editor. Anopheles mosquitoes - New insights into malaria vectors. Rijeka: Intech Open; 2013. p. 397-446.
- 106 Pinault LL, Hunter FF. Characterization of larval habitats of Anopheles albimanus, Anopheles pseudopunctipennis, Anopheles punctimacula, and Anopheles oswaldoi s.l. populations in lowland and highland Ecuador. J Vector Ecol. 2012; 37(1): 124-36.
- 107 Rejmánková E, Pope KO, Pohl MD, Rey-Benayas JM. Freshwater wetland plant communities of northern Belize: implications for paleoecological studies of Maya wetland agriculture. Biotropica. 1995; 27: 28-36.
- 108 Rejmánková E, Pope KO, Post R, Maltby E. Herbaceous wetlands of the Yucatan Peninsula: communities at extreme ends of environmental gradients. Int Rev Hydrobiol. 1996; 81: 223-52.
- 109 Greico JP, Johnson S, Achee NL, Masuoka P, Pope K, Rejmánková E, et al. Distribution of Anopheles albimanus, Anopheles vestitipennis, and Anopheles crucians associated with land use in northern Belize. J Med Entomol. 2006; 43: 614-6.
- 110 Maheu-Giroux M, Casapia M, Soto-Calle VE, Ford LB, Buckeridge DL, Coomes OT, et al. Risk of malaria transmission from fishponds in the Peruvian Amazon. Acta Trop. 2010; 115(1-2): 112-8.
- 111 Orr BK, Resh VH. Experimental test of the influence of aquatic macrophyte cover on the survival of Anopheles larvae. J Am Mosq Control Assoc. 1989; 5: 579-85.
- 112 Merritt RW, Dadd RH, Walker ED. Feeding behavior, natural food, and nutritional relationships of larval mosquitoes. Ann Rev Entomol. 1992; 37: 349-76.
- 113 Rejmánková E, Rubio-Palis Y, Villegas L. Larval habitats of Anopheline mosquitoes in the upper Orinoco Venezuela. J Vector Ecol. 1999; 24(2): 130-7.
- 114 Vinogradov DD, Sinev AY, Tiunov AV. Predators as control agents of mosquito larvae in micro-reservoirs (Review). Inland Water Biol. 2022; 15(1): 39-53.
- 115 Hernández JE, Epstein LD, Rodríguez MH, Rodríguez AD, Rejmánková E, Roberts DR. Use of generalized regression tree model to characterize vegetation favoring Anopheles albimanus breeding. J Am Mosq Control Assoc. 1997; 13(1): 28-34.
- 116 Osuna E. Entomología del Parque Nacional Henri Pittier. Fundación Polar; 2000. 199 pp.
- 117 Arcos AN, Valente-Neto F, Ferreira FAS, Bolzan FP, da Cunha HB, Tadei WP, et al. Seasonality modulates the direct and indirect influences of forest cover on larval anopheline assemblages in western Amazônia. Sci Rep. 2021; 11: 12721.
- 118 Achee NL, Grieco JP, Andre RG, Roberts DR, Rejmánková E. Experimental evaluation of overhanging bamboo in Anopheles darlingi larval habitat selection in Belize, Central America. J Vector Ecol. 2006; 31: 145-51.
- 119 Olson SH, Gagnon R, Elguero E, Durieux L, Guégan J, Foley JA, et al. Links between climate, malaria, and wetlands in the Amazon Basin. Emerg Infect Dis. 2009; 15: 659-62.
- 120 Wolfarth-Couto B, Filizola N, Tadei WP, Durieux L. Epidemiological analysis of malaria and its relations with environmental variables in four municipalities of the State of Amazonas - Brazil. Hydrol Sci J. 2013; 58: 1495-504.
- 121 Wolfarth-Cuoto B, da Silva RA, Filizola N. Variability in malaria cases and the association with rainfall and rivers water level in Amazonas State, Brazil. Cad Saude Publica. 2019; 35(2): e00020218.
- 122 Fonseca F, Worfarth-Couto B, Santos A, Marinho R, Martinez JM, Filizola N. Hydrological scenarios and malaria incidence in the Amazonian context. Water. 2022; 14: 1283.
- 123 Hay SI, Sinka ME, Okara RM, Kabaria CW, Mbithi PM, Tago CC, et al. Developing global maps of the dominant Anopheles vectors of human malaria. PLoS Med. 2010; 7(2): e1000209.
- 124 Alemu A, Abebe G, Tsegaye W, Golassa L. Climatic variables and malaria transmission dynamics in Jimma town, southwest Ethiopia. Parasit Vectors. 2011; 4: 30.
- 125 Smith MW, Macklin MG, Thomas CJ. Hydrological and geomorphological controls of malaria transmission. Earth-Sci Rev. 2013; 116: 109-27.
- 126 Mafwele BJ, Lee JW. Relationships between transmission of malaria in Africa and climate factors. Sci Rep. 2022; 12: 14392.
- 127 Rama C, Vásquez J, Magallanes J, Carey C. Variables climáticas y transmisión de malaria en Loreto, Perú: 1995-2007. Rev Peru Med Exp Salud Publica. 2009; 26: 9-14.
- 128 Rodrigues IB, Tadei WP, Dias JMCS. Larvicidal activity Bacillus sphaericus 2362 against Anopheles nuneztovari, Anopheles darlingi and Anopheles braziliensis (Diptera, Culicidae). Rev Inst Med Trop São Paulo. 1999; 41(2): 101-5.
- 129 Rodrigues BI, Tadei WP, Dias JMSD, Lima CAP. Atividade larvicida de Bacillus sphaericus 2362 contra Anopheles sp (Diptera, Culicidae) em rios do Amazonas, Brasil. Bio Assay. 2013; 8(2): 1-7.
- 130 Galardo AKR, Zimmerman R, Galardo CD. Larval control of Anopheles (Nyssorrhynchus) darlingi using granular formulation of Bacillus sphaericus in abandoned gold-miners excavation pools in the Brazilian Amazon rainforest. Rev Soc Bras Med Trop. 2013; 46(2): 172-7.
- 131 Berrocal E, Carey C, Rodríguez L, Calampa C, Valdivia L. Residual effect of Bacillus sphaericus-VectoLex(r) for the control of Anopheles darlingi in Iquitos, Peru. J Am Mosq Control Assoc. 2000; 15: 305-6.
- 132 Suárez MF, Morales CA. Impact of Bacillus sphaericus (VectoLex(r) CG) in the control of Anopheles albimanus and Culex spp. in Buenaventura, Colombia. J Am Mosq Control Assoc. 1999; 15: 407.
- 133 Moreno JE, Acevedo P, Martínez A, Sánchez V, Petterson L. Evaluación de la persistencia de una formulación comercial de Bacillus sphaericus en criaderos naturales de anofelinos vectores de malaria en el estado Bolívar, Venezuela. Bol Mal Salud Amb. 2010; 50(1): 109-17.
- 134 Moreno JE, Martínez A, Acevedo P, Sánchez V, Amaya W. Evaluación preliminar de la eficiencia de Bacillus sphaericus en un área endémica a malaria del estado Bolívar, Venezuela. Bol Mal Salud Amb. 2014; 54(1): 47-57.
- 135 Tusting LS, Thwing J, Sinclair D, Fillinger U, Gimnig J, Bonner KE, et al. Mosquito larval source management for controlling malaria. Cochrane Database Syst Rev. 2013; 2013(8): CD008923.
Publication Dates
-
Publication in this collection
07 Oct 2024 -
Date of issue
2024
History
-
Received
07 June 2024 -
Accepted
04 Sept 2024

 Characterization of larval habitats of Anopheles (Nyssorhynchus) darlingi and associated species in malaria areas in western Brazilian Amazon
Characterization of larval habitats of Anopheles (Nyssorhynchus) darlingi and associated species in malaria areas in western Brazilian Amazon